acetic anhydride hydrolysis mechanism
|
PicoSpin 45: Hydrolysis of Acetic Anhydride with Heavy Water
Acetic anhydride rapidly hydrolyzes in the presence of water alcohol and catalyzing acid in this case water We can monitor the evolution of the reaction using NMR by a modified in situ reaction monitoring whereby a single aliquot of the reaction mixture is injected into the RF coil of the NMR probe |
Which spectroscopic techniques are used to monitor acetic anhydride hydrolysis?
Two spectroscopic techniques (NIR and UV–vis) were compared for the in-situ, real-time monitoring of the reaction. Hydrolysis of acetic anhydride is a fast and very exothermic reaction that has been studied with different analytical techniques to account for the reaction progress.
What are the kinetic and mechanistic similarities of acetic anhydride?
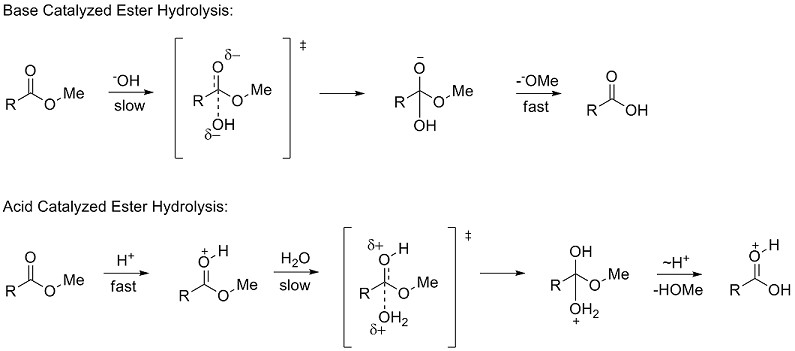
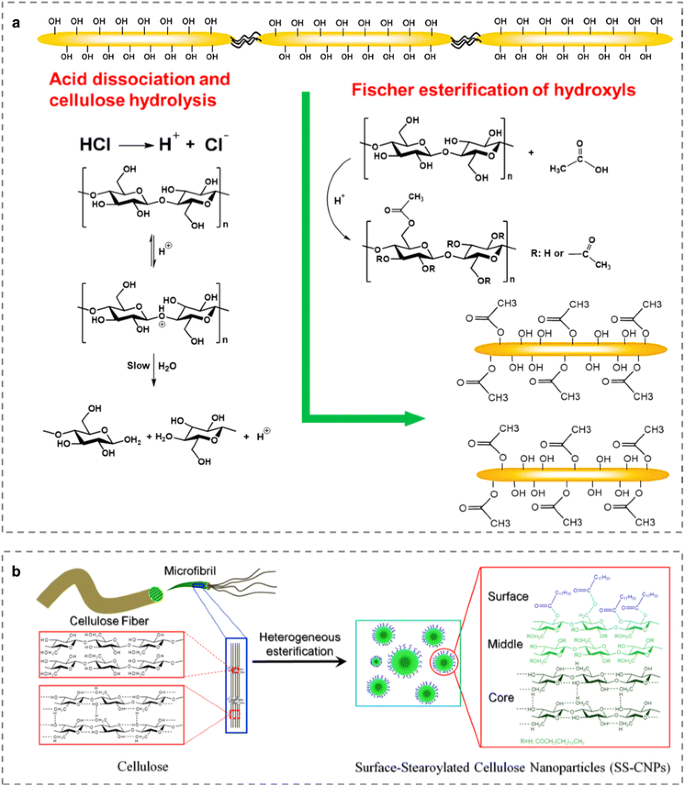
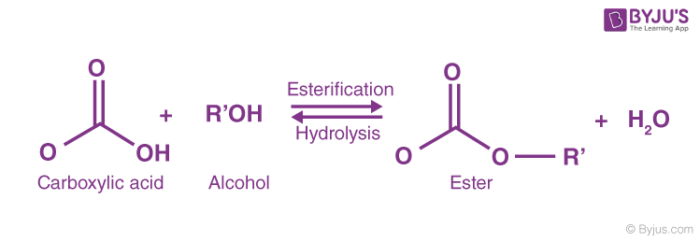
The reaction has also kinetic and mechanistic similarities with other important industrial reactions of acetic anhydride, such as the acetylation of hydroxyl groups to form acetates, and with other different industrial processes, such as hydrolysis of esters and esterification of different alcohols using anhydrides.
How can acetic anhydride be monitored using NMR?
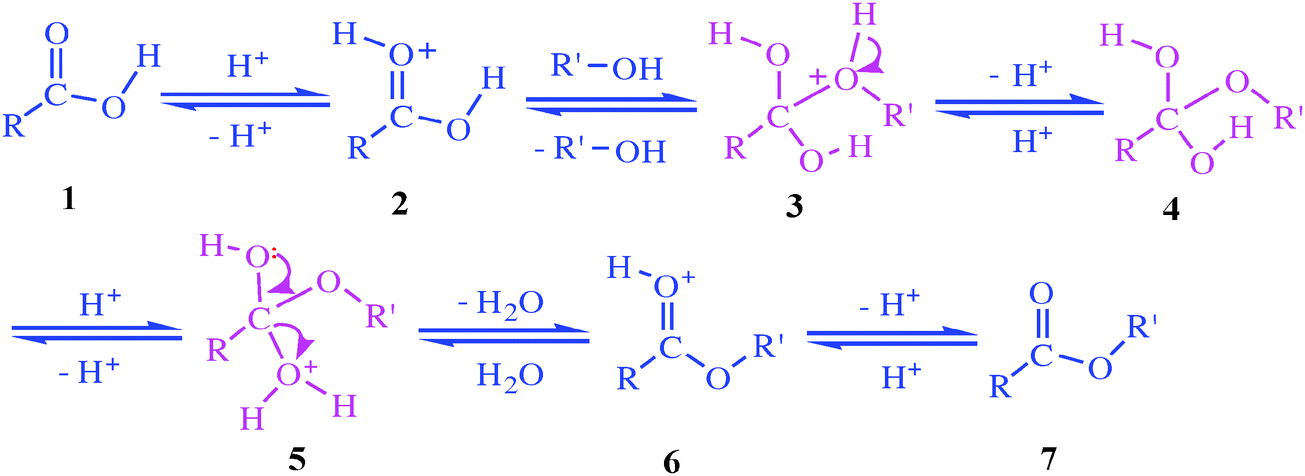
Acetic anhydride rapidly hydrolyzes in the presence of water, alcohol and catalyzing acid, in this case water. We can monitor the evolution of the reaction using NMR by a modified in situ reaction monitoring whereby a single aliquot of the reaction mixture is injected into the RF coil of the NMR probe.
What is the hydrolysis of acetic anhydride?
The hydrolysis of acetic anhydride is an exothermic reaction that can be described as: (1) A + B → k 2 C where A is acetic anhydride (the limiting reactant), B is water (present in excess) and C is acetic acid.
|
PicoSpin 45: Hydrolysis of Acetic Anhydride with Heavy Water (D2O)
The hydrolysis of acetic anhydride (Ac2O) to acetic acid (AcOH) serves as a Lowry T.H.; Richardson |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: V
The acid-catalyzed hydrolysis of acetic anhydride has been studied by Kilpatrick (I) and by Gold and I-Iilton (23). It is i~sually accepted that the first step |
|
Kinetics and Activation Energy Parameters for Hydrolysis of Acetic
The hydrolysis reaction of acetic anhydride in a water/acetone cosolvent systems forming acetic is pseudo-first order. The reaction mechanism begins by acetic |
|
Solvent Isotope Effects in Catalyzed Hydrolysis of Carboxylic Acid
The latter mechanism is discarded for carboxylic acid derivatives with good leaving groups pyridine-catalyzed hydrolysis of acetic anhydride. |
|
The Hydrolysis of Carboxylic Anhydrides. V.12 The Acid Hydrolysis
The acid hydrolysis of trimethylacetic anhydride follows an A-2 mechanism in both water and aqueous di- oxane and is slower than that of acetic anhydride. |
|
2305 446. The Hydrolysis of Acetic Anhydride. Part VI.* Kinetics in
acetate ions in this reaction. The rate-controlling step in this mechanism is a proton-transfer from hydrogen ion or acetic acid to the oxygen atom. |
|
The Mechanism of Hydrolysis of Acetyl Dihydrogen Phosphate
was first employed to study the alkaline hydrolysis of «-amyl acetate.6 In case. (I) O18 would be found in the acetic acid produced |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VIII
The hydrolysis of acetamide was followed by ailalyziilg for the acetic acid produced. About 5 in1 of reaction solutioil (initially either 0.2992 M in |
|
[ 19551 Gold and Hiltmi. 843 Th c H?/droZysi.s of Acetic Anhydride
mechanism in which the rate-determining step is the formation of an acetylium The hydrolysis of acetic anh3dride i?t nqueoids perchloric acid at 0". |
|
Acetylpyridinium ion intermediate in pyridine-catalyzed hydrolysis
Hydrolysis and Acyl Transfer Reactions of Acetic Anhydride. involve the same mechanism with another nucleophile |
|
Kinetic Modeling of the Hydrolysis of Acetic Anhydride at Higher
14 oct 2013 · mechanisms pathways for the production of acetic acid Keywords: Kinetic modeling; Thermos flask; Hydrolysis; Esterification; Exothermic |
|
Reactions of Anhydrides Mechanism of Anhydride Substitution
excess alcohol under acidic conditions • The mechanism is similar to acid- catalyzed esterification/hydrolysis • Esters can also undergo trans-esterification |
|
Carboxylic acid deri
acetic anhydride benzoic The mechanism of nucleophilic acyl substitution involves Hydrolysis of esters: esters can be hydrolyzed to the carboxylic |