acetic anhydride reaction with primary amine
Which anhydride is used in a carboxylic acid reaction?
Because many anhydrides are made from the coupling of two carboxylic acids, reactions using anyhydrides waste one equivalent of the carboxylic acid as a leaving group. As a consequence, reactions are only commonly performed with inexpensive, readily available anhydrides, such as acetic anhydride or benzoic anhydride.
Are anhydrides reactive to nucleophilic attack?
Anhydrides are highly reactive to nucleophilic attack and undergo many of the same reactions as acid chlorides that were explored in section 21.4. Although slower reacting than acid chlorides, anhydrides react with water to form carboxylic acids, with alcohols to form esters, and with amines to form amides.
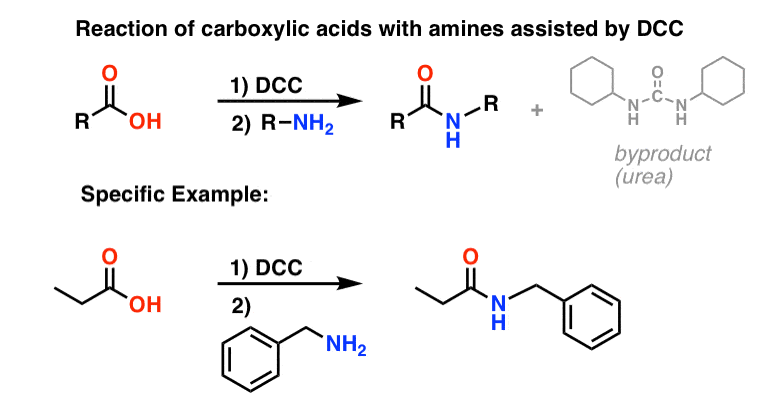
How do acid anhydrides react with amines to form amides?
Acid Anhydrides react with amines to form amides General Reaction Example 1: Mechanism 1) Nucleophilic Attack by the Amine 2) Deprotonation by the amine 3) Leaving group removal Contributors Prof. Steven Farmer (Sonoma State University) Back to top Acid Anhydrides react with alcohols to form esters
How do acid anhydrides react with water?
The chemistry of acid anhydrides is similar to that of acid chlorides, although anhydrides react more slowly. Thus, acid anhydrides react with water to form acids, with alcohols to form esters, with amines to form amides, and with LiAlH 4 to form primary alcohols. Only the ester- and amide-forming reactions are commonly used, however.

Imine and Enamine Formation Reactions With Reductive Amination

Nomenclature and properties of amides Organic chemistry Khan Academy

Nomenclature and properties of acyl (acid) halides and acid anhydrides Khan Academy
|
Analytical Procedures Employing Karl Fischer Reagent. XI. The
the system acetic acid-ammonia has been com- used in the determination of tertiary amines6·7 ... tive reaction of acetic anhydride with the primary. |
|
THE PREPARATION OF SOME PRIMARY AMINES BY THE
as primary and the former may frequently constitute the chief reaction products. Acetic Acid Solutions. Nitrile. Solvent. %. Primary amine. oSecondary. |
|
Formylation of Amines
10 jui. 2014 The amine and formic acid were heated to 80 °C until the reaction ... generated in situ from excess formic acid and acetic anhydride at ?20 ... |
|
Aminolysis of acid anhydrides in water. II. Nonlinear structure
Abstract: The reactions in water of 21 primary and secondary amines with phthalic and Each reaction was first order in both anhydride and amine neutral ... |
|
Mild and eco-friendly chemoselective acylation of amines in
9 We thought to add sodium bicarbonate to an aqueous solution of amine hydrochloride which will liberate free amine and react with acetic anhydride and convert |
|
Determination of Tertiary Aliphatic Amines in the Presence of
The Analysis of Aliphatic Amine Mixtures; Determination of Tertiary Aliphatic of acetic acid in the reaction mixture cer- tain tertiary amines are ... |
|
Nitrosation Reactions of Primary Vinylamines. 3-Amino-2
addition of acetic anhydride water |
|
Testsforfunctionalgroups - inorganiccompounds
Both the above reactions are used as tests for unsaturation. Primary amine e.g. aniline reacts with nitrous acid generated in situ by the. |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the products of amide synthesis and hydrolysis reactions. |
|
Efficient acetylation of primary amines and amino acids - Indian
The hydro- gen chloride (HCl) liberated during the reaction reacts with sodium acetate to form acetic acid in situ and thus the reaction medium is buffered due to |
|
Acetylation of amines using catalytic amount of zinc acetate - NOPR
structurally diverse amines, and the reaction is chemoselective with respect to phenols, Keywords: Microwave irradiation, amines, acetic acid, zinc acetate, N- acetylation acetylation of primary and secondary amines, where a variety of |