acid catalysed hydrolysis of ester experiment
|
IV SEMMESTER
EXPERIMENT Page No 1 Kinetics of Acid hydrolysis of an ester 2 2 Estimation of mixture of acids conductometrically 4 3 Estimation of Copper (II) by |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
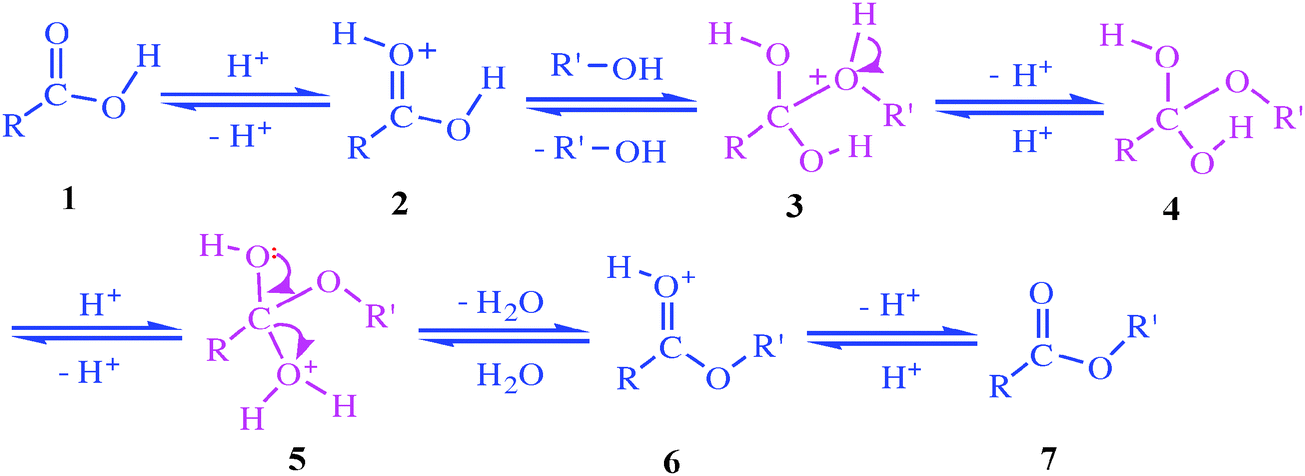
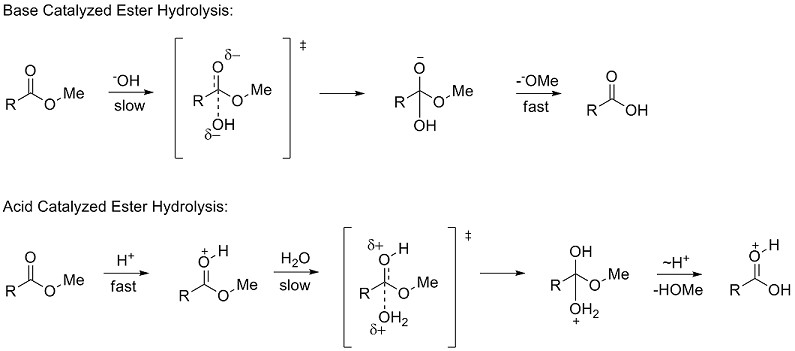
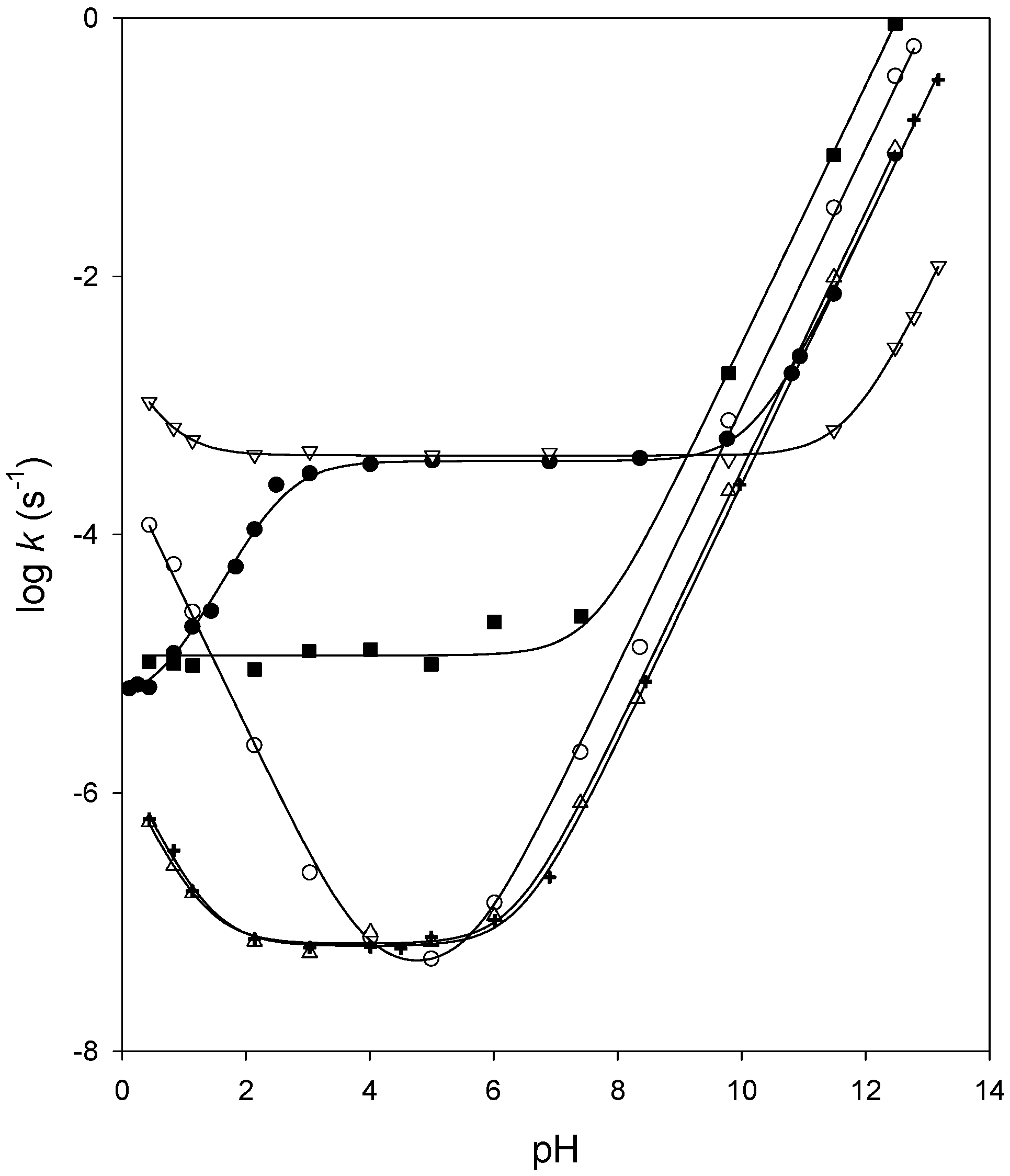
The hydrolysis of a carboxylic acid ester can proceed via three distinct bimolecular mechanisms; acid catalyzed, neutral and base enhanced Acid catalyzed rate |
|
Experiment 5
To determine the rate constant for the acid-catalyzed hydrolysis of methyl acetate The progress of the reaction (hydrolysis of ester) is followed by removing a |
|
Page 1 of 12 CHEM 100L Lab 7: Ester Hydrolysis Purpose: In the
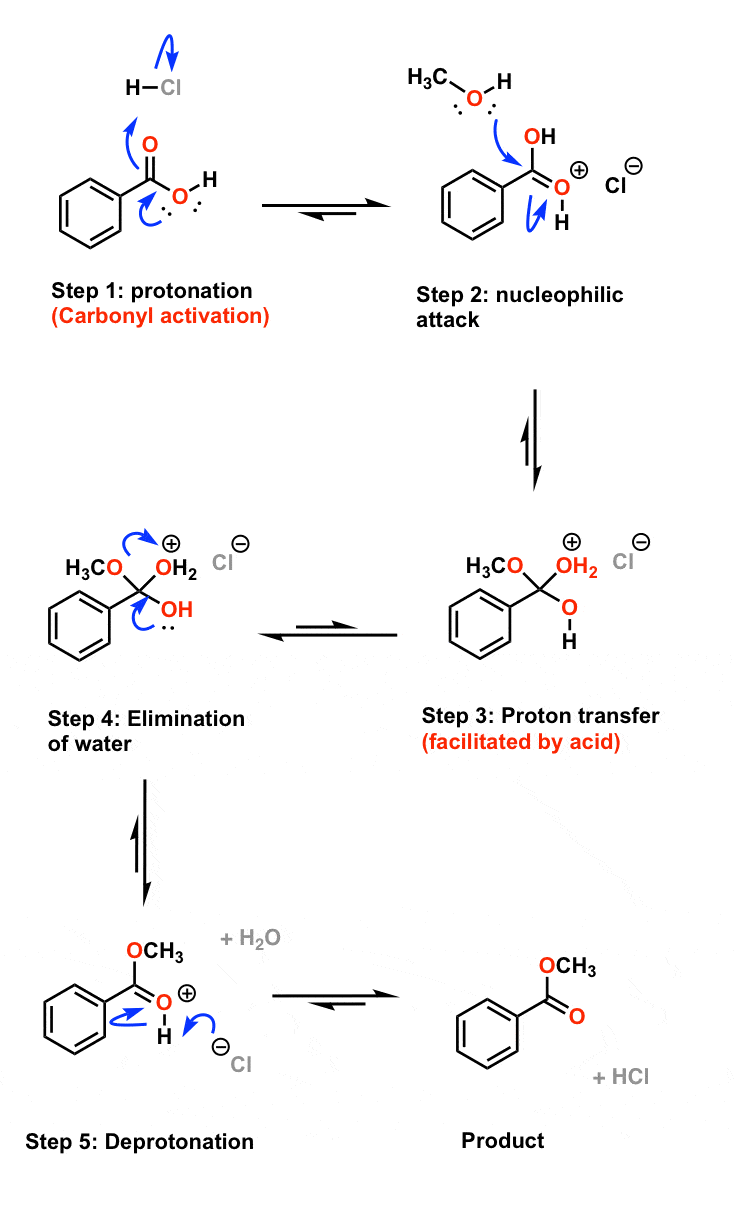
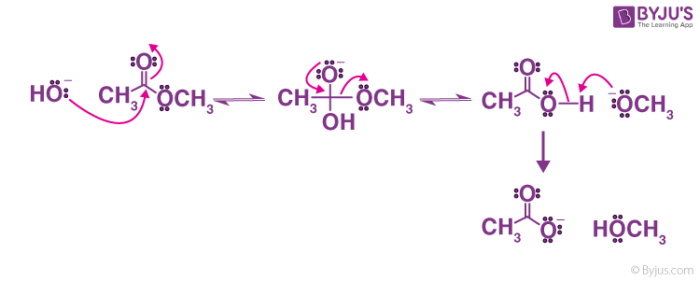
The addition of an acid catalyst can increase the rate of the reaction In this virtual experiment, you will be hydrolyzing methyl propionate by refluxing the ester in aqueous solution, using sulfuric acid as the catalyst A general mechanism for an acidic ester hydrolysis is shown below in figure 7 2 |
|
Hydrolysis of ester lab report - Squarespace
We will study catalyzed acid (HCl) ester hydrolysis (ethyl acetate, etAc), form alcohol (ethanol, EtOH) and acid (acetic acid, HAc): CH3CH2CO2CH3 + H2O |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide), HCl was used as catalyst to the presence of a mineral acid gives acetic acid and ethyl alcohol final titre value at the end of the experiment and Vt = titre value at |
|
Student 3: results of 22-9-06 experiment (velocity constant):
To Determine the Velocity Constant for the Hydrolysis of Methylacetate Using an Theory: Methylacetate hydrolyses in presence of an acid (acts as catalyst) and produces Let, a = initial concentration of ester (at t = 0) of the above reaction |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions form new esters by base- or acid-catalysed transesterification mechanisms; |

















![PDF] Kinetic studies on acid catalysed hydrolysis of starch PDF] Kinetic studies on acid catalysed hydrolysis of starch](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/b4afe119-dd2d-4434-b749-548bf9a123dc/gr1_lrg.jpg)

![PDF] Kinetic studies on acid catalysed hydrolysis of starch PDF] Kinetic studies on acid catalysed hydrolysis of starch](https://pubs.rsc.org/image/article/2018/RA/c7ra11824d/c7ra11824d-f7_hi-res.gif)





![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://doubtnut-static.s.llnwi.net/static/q-thumbnail/12775017.png)




![PDF] Kinetic studies on acid catalysed hydrolysis of starch PDF] Kinetic studies on acid catalysed hydrolysis of starch](https://imgv2-2-f.scribdassets.com/img/document/269192528/149x198/c19d41bd9d/1606817532?v\u003d1)







![PDF] Kinetic studies on acid catalysed hydrolysis of starch PDF] Kinetic studies on acid catalysed hydrolysis of starch](https://demo.vdocuments.mx/img/378x509/reader018/reader/2020011320/55a14cab1a28ab4e398b45bd/r-1.jpg)
![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://i.ytimg.com/vi/YAIwkLv1zac/hq720.jpg?sqp\u003d-oaymwEhCK4FEIIDSFryq4qpAxMIARUAAAAAGAElAADIQj0AgKJD\u0026rs\u003dAOn4CLCcxIamRrv8ok95ED0oGIsNvpyGnQ)
