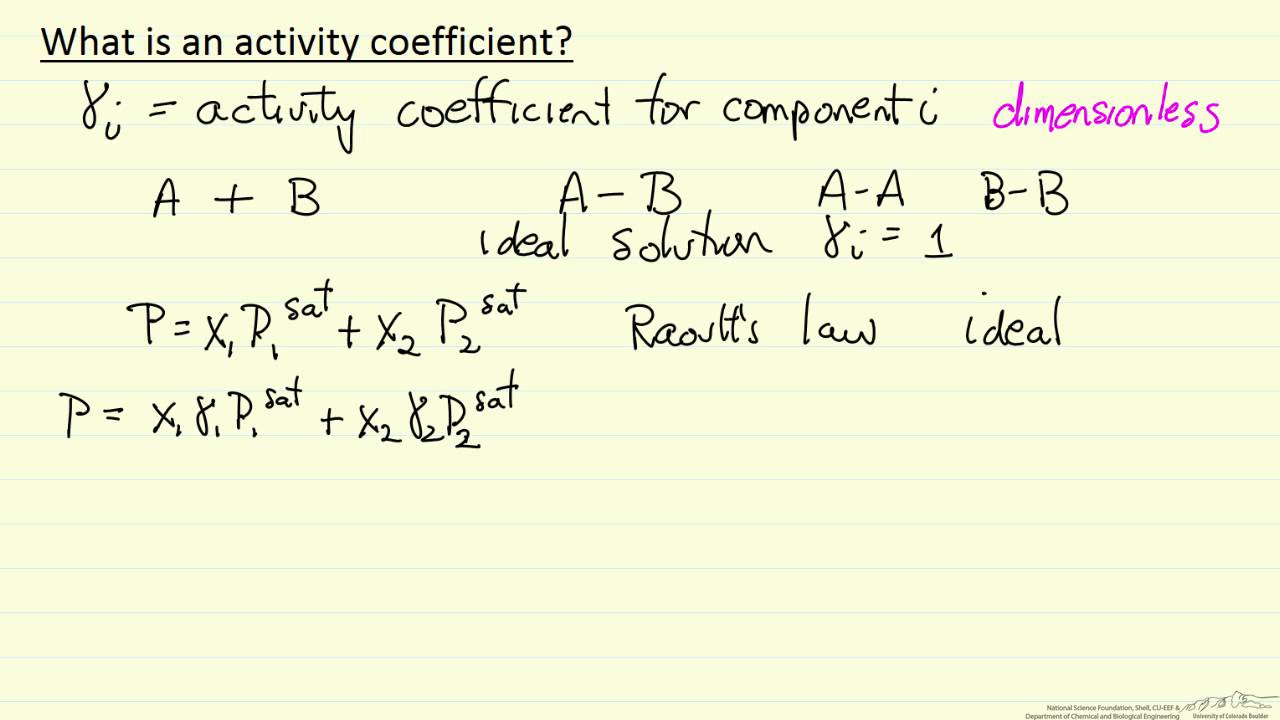activity coefficient equation
|
Chem 355 Activity Coefficients
allows calculation of activity coefficients ϒχ for ions from knowledge of charge Z the ionic strength of the solution μ and average diameter of hydrated ion |
1Activity is the effective concentration of any solution. 2a = ¥*C where a is activity, ¥ is activity coefficient (pronounced as gamma) and C is its concentration.
3) For ideal solution and solid phase, ¥ is 1 i.e. activity= concentration.
What is activity coefficient formula?
In the 1920s, the Dutch-American chemist Peter Debye and the German chemist Erich Hückel developed a foundational theory and equation that relates ionic strength and activity coefficients for relatively dilute electrolyte solutions.
The Debye-Hückel activity coefficient equation is: log γ ± = − 0.509 z + z − I.
How do you find the mean activity coefficient?
The mean activity coefficient is defiend as follows:
1γ±=(γ+γ−)1/2.2−logγi=0.5z2iμ1/2.3−logγ±=0.5z+z−μ1/2,What is the activity coefficient γ?
However, the mean activity coefficient γl can be measured for any salt.
The mean activity coefficient is determined from measurements of such properties of solutions as freezing point, vapor pressure, solubility, and electrical potential.
|
Application of several activity coefficient models to water-organic
22 de set. de 2005 The equilibrium liquid phase concentrations can be calcu- lated by solving the water concentration so that the Kelvin equation (Eq. 1) is ... |
|
Calculation of drug-like molecules solubility using predictive activity
26 de out. de 2018 The aim of this work is to evaluate different activity coefficient models. UNIFAC |
|
Determination of Vapor-Liquid Equilibrium Parameters Using
1 de set. de 2011 The Wilson equation is applied as the activity coefficient model. 3. Component 1. Component 2. Condition. Azeotropic type. |
|
Calculation of Activities of Ions in Molten Salts with Potential
These differences may be expressed as ratios of activity coefficients which depend on the identity of the solvent and other factors. Here |
|
Measurement and Modeling of Mean Ionic Activity Coefficient in
Knowledge of activity coefficients of the constituent components in aqueous solutions is essential in calculation of phase equilibrium. A number of elaborate. |
|
3. Activity Coefficients of Aqueous Species 3.1. Introduction
The Davies equation predicts a unit activity coefficient for all neutral solute species. This is known to be inaccurate. In general the activity coefficients |
|
Generalization of Guggenheims combinatorial activity coefficient
15 de set. de 2018 Subsequently we obtain a general formula for the combinatorial activity coefficient for mixtures of molecules differing in size and shape. |
|
CALCULATION OF ACTIVITY COEFFICIENT AND SOLUBILITY FOR
30 de set. de 2019 One of the basic equations for these calculations is presented in equation 1 which is the calculation of the molality for an aqueous solution. |
|
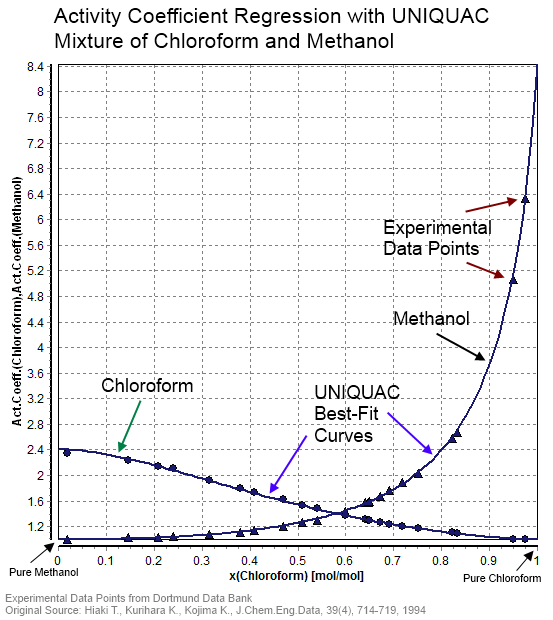
Comparison of Activity Coefficient in ABE with MEP with UNIQUAC
UNIQUAC equations in calculating the activity coefficient Acetone Butanol Ethanol (ABE) ternary system using the Mat-lab programming language then verified |
|
Vacancy Solution Theory of Adsorption Using Flory-Huggins Activity
16 de jan. de 2022 Flory-Huggins activity coefficient equations. The isotherm equation contains three regression parameters: a Henry's law constant ... |
|
Chem 321 Lecture 11 - Chemical Activities - CSUN
3 oct 2013 · In 1923 Peter Debye and Erich Hückel developed an expression that allows one to calculate activity coefficients The extended Debeye-Hückel equation indicates that γ depends on three factors where z is the charge on the ion; α is the hydrated ion radius (in pm); μ is the ionic strength of the solution |
|
3 Activity Coefficients of Aqueous Species 31 Introduction
Mole fraction based activities and activity coefficients (λi), are occasionally The Davies equation predicts a unit activity coefficient for all neutral solute species |
|
Chapter 6 Activity Scales and Activity Corrections
11 oct 2004 · The single ion activity coefficients by the mean salt method are compared with values calculated from the extended Debye-Huckel equation in Fig |
|
Concept of Activity
mean activity coef is defined as Mean activity coefficient of an electrolyte AmBn; The Debye-Huckel equation actually breaks down for large μ values Individual |
|
TEMPERATURE EFFECTS ON THE ACTIVITY COEFFICIENT OF
Therefore, the Ion Interaction Model is a simple and accurate technique to calculate the equilibrium properties of mixed electrolyte solutions The above equations |
|
Ionic strength
•calculation of ionic strength •activity coefficients •effect of ionic strength and size /charge of ions on activity coefficients •the real definition of pH--- •charge and |
|
Measurement and Modeling of Mean Ionic Activity Coefficient in
Knowledge of activity coefficients of the constituent components in aqueous solutions is essential in calculation of phase equilibrium A number of elaborate |
|
Activity and Concentration
activity coefficient γ, where [a] = γ (c) Until now we Calculation of Ionic Strength equation • Davies Equation for higher ionic strengths ( |