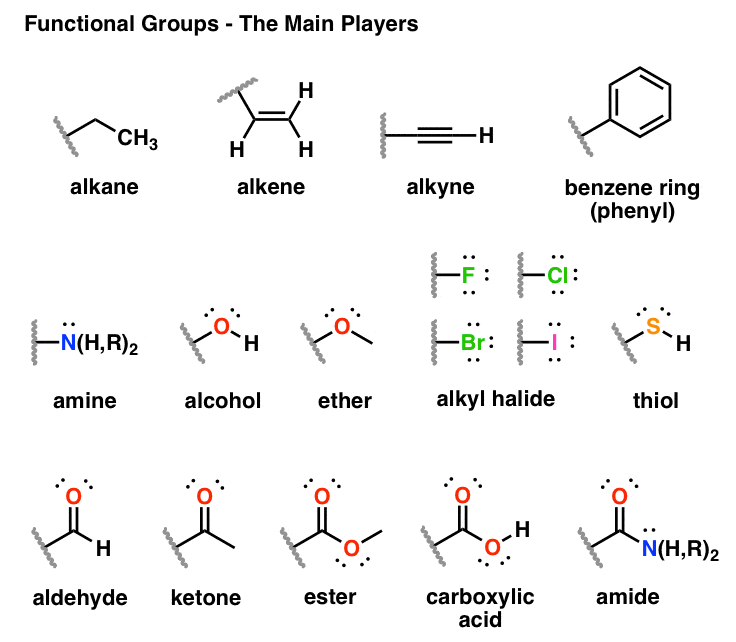
alcohol ether aldehyde ketone carboxylic acid ester amine
What are carboxylic acids & esters?
The names for carboxylic acids and esters include prefixes that denote the lengths of the carbon chains in the molecules and are derived following nomenclature rules similar to those for inorganic acids and salts (see these examples): The functional groups for an acid and for an ester are shown in red in these formulas.
Can ethanol be oxidized to aldehydes?
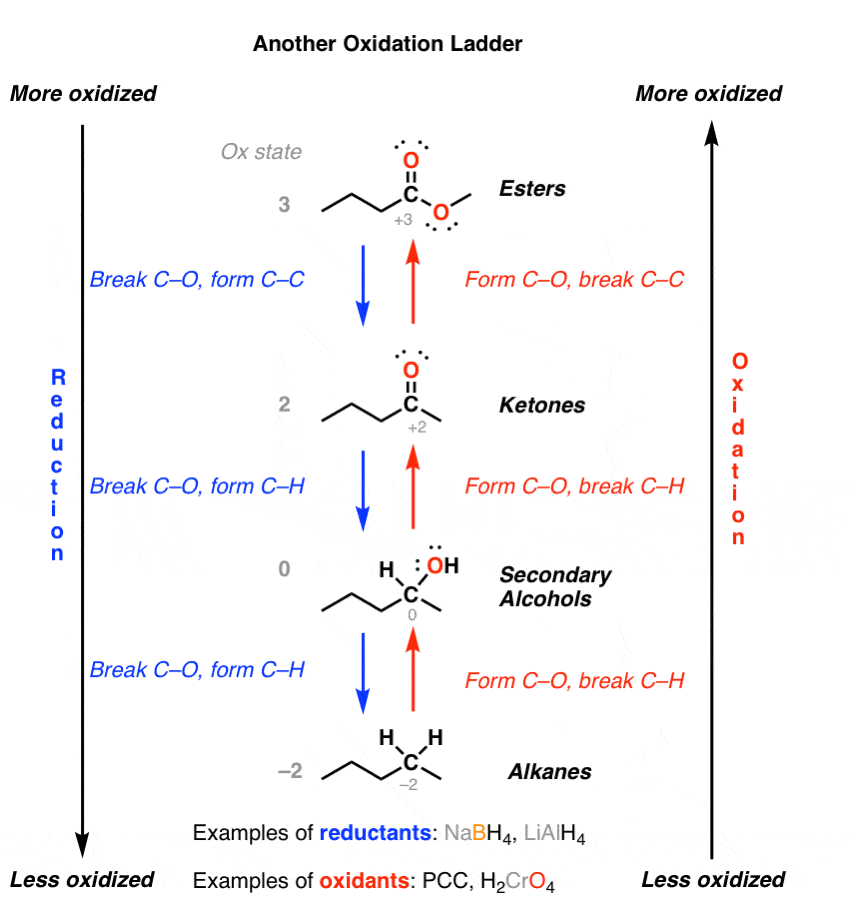
It turns out that primary alcohols may be oxidized to aldehydes which, in turn, may be oxidized to carboxylic acids. Figure 12.7.1 12.7. 1 shows how ethanol, a primary alcohol that has two carbon atoms, is eventually oxidized to ethanoic acid, a two-carbon carboxylic acid.
What is the difference between aldehyde and ketone?
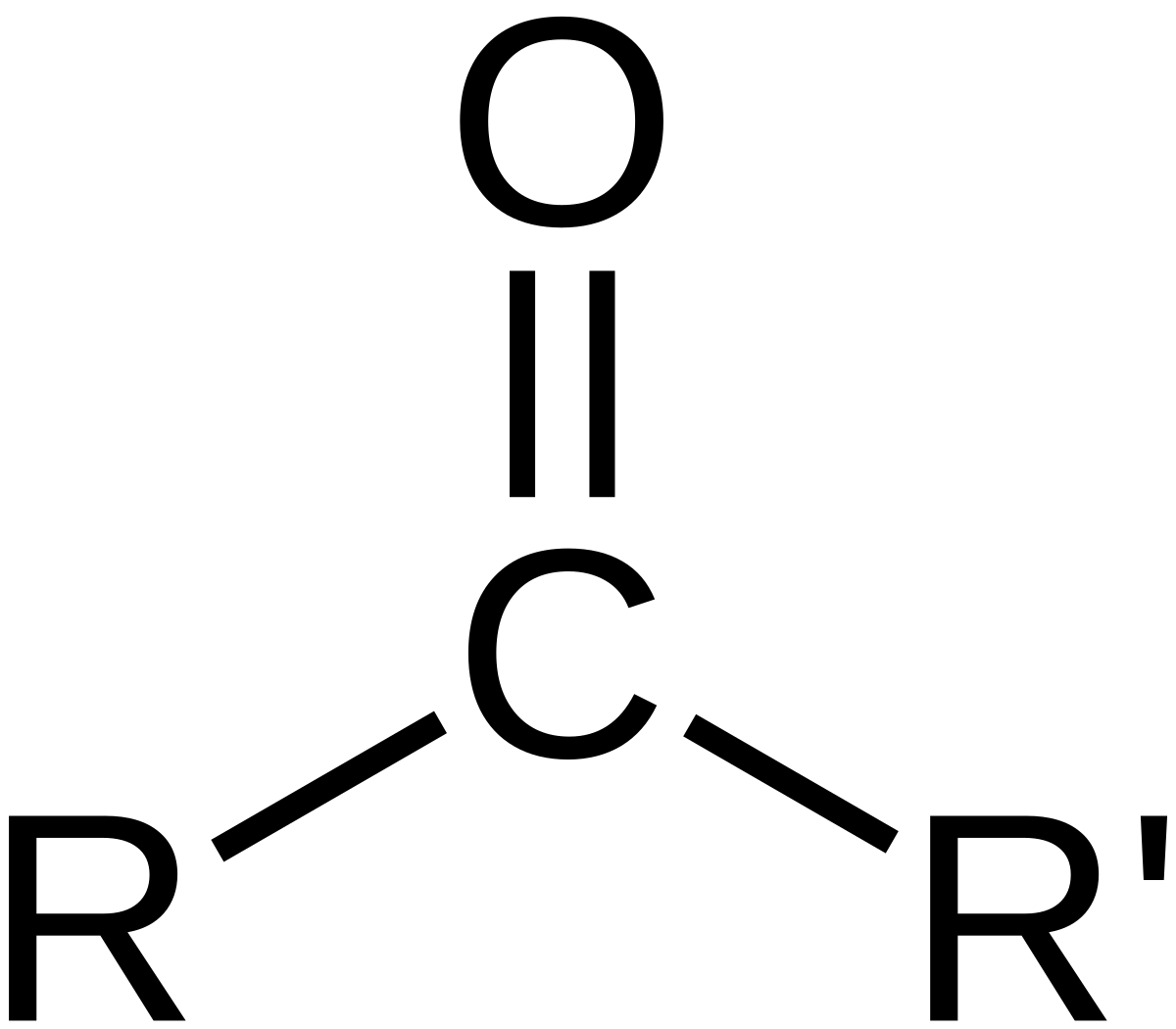
The names for aldehyde and ketone compounds are derived using similar nomenclature rules as for alkanes and alcohols, and include the class-identifying suffixes –al and –one, respectively: In an aldehyde, the carbonyl group is bonded to at least one hydrogen atom. In a ketone, the carbonyl group is bonded to two carbon atoms:
Aldehydes and Ketones
Both aldehydes and ketones contain a carbonyl group, a functional group with a carbon-oxygen double bond. The names for aldehyde and ketone compounds are derived using similar nomenclature rules as for alkanes and alcohols, and include the class-identifying suffixes –al and –one, respectively: In an aldehyde, the carbonyl group is bonded to at leas
Carboxylic Acids and Esters
The odor of vinegar is caused by the presence of acetic acid, a carboxylic acid, in the vinegar. The odor of ripe bananas and many other fruits is due to the presence of esters, compounds that can be prepared by the reaction of a carboxylic acid with an alcohol. Because esters do not have hydrogen bonds between molecules, they have lower vapor pres
Glossary
aldehyde: organic compound containing a carbonyl group bonded to two hydrogen atoms or a hydrogen atom and a carbon substituent carbonyl group: carbon atom double bonded to an oxygen atom carboxylic acid: organic compound containing a carbonyl group with an attached hydroxyl group ester: organic compound containing a carbonyl group with an attached

Oxidation of Alcohols to Aldehyde Ketone and Carboxylic Acid

Aldehydes and Ketones

Aldehyde Ketone and Carboxylic Acid: Structure Properties and Reactions // HSC Chemistry
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Carbonyl compounds have substantial dipole moments and are polar than ethers. The high polarity of the carbonyl group is explained on the basis of resonance |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to Boiling Point: Carboxylic acid. Alcohols. Aldehydes/Ketones. Ethers. |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
carboxylic acids and their derivatives (e.g. esters |
|
Systematic optimization of a fragment-based force field against
19 juil. 2021 functional groups involving oxygen and/or nitrogen atoms namely: ether |
|
CHEM-109 SLO Assessment - SLOA Committee - Ohlone College
22 févr. 2012 alcohol ether |
|
Reducing-agents.pdf
It will reduce aldehydes ketones |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to Boiling Point: Carboxylic acid. Alcohols. 1°/2° Amines. 3° Amines/Alkanes ... |
|
SIMPOL.1: a simple group contribution method for predicting vapor
ether alkyl ring ether |
|
Lab 14: Qualitative Organic Analysis
one major functional group (alcohol ketone |
|
Alcohols, Ethers, Aldehydes, and Ketones
ether, thiol, sulfide, disulfide, aldehyde, and ketone families, and describe how they are named Alcohols, Ethers, and Related Disulfides ▫ S-S bonds between two cysteines (amino acids) ketones, carboxylic acids, esters, and amides |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
carboxylic acids, and their derivatives (e g esters, Aldehydes, ketones and carboxylic acids are widespread in plants polar than ethers This method is suitable for volatile alcohols and is of industrial 12 2: Some N-Substituted Derivatives of Aldehydes and Ketones (>C=N-Z) -H Ammonia Imine -R Amine —OH |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Alcohols are oxidized to aldehydes and ketones (example: biological oxidation of ethanol to acetaldehyde) • The carbonyl group may be further oxidized to carboxylic acids Reactions of Aldehydes and Ketones with Amines prepared in diethyl ether Treating a carboxylic acid with an excess of an alcohol gives esters |
|
Lab 14: Qualitative Organic Analysis - California State University
one major functional group (alcohol, ketone, aldehyde, amide, amine, carboxylic acid, or ester) Additionally Both strong and weak acids (Carboxylic acids and phenols) will be Recrystallize the precipitate from petroleum ether or heptane |
|
Unit One Part 2: naming and functional groups
Suffix Prefix General form acid R OH O –oic acid –carboxylic acid carboxy ester R OR O –oate –carboxylate alkoxycarbonyl R-COOR amide R NH2 O –amide ketone R R O –one oxo R-CO-R alcohol R OH –ol hydroxy R-OH amine alcohols ethers amines alkyl halides thiols aldehydes ketones carboxylic |
|
Twelve To Remember: The Functional Groups
-"D" for C=O double bond 12 Amine -one nitrogen, no C=O -"N" for No C=O double Alcohol Ether -NH2 Aldehyde Ketone Carboxylic Acid Ester Amide |
|
Functional Groups and Infrared Spectroscopy - CORE
31 mai 2019 · C=O compounds: aldehydes, ketones, carboxylic acids, esters, acyl ether c) 1° amine d) 3° alcohol e) aldehyde f) 1° alkyl halide g) thiol h) |
|
Chapter 17 An Introduction to Organic Chemistry, Biochemistry, and
Alcohols • Carboxylic Acids • Ethers • Aldehydes • Ketones • Esters • Amines • Amides • Organic Compounds with More Than One Functional Group |
|
Alcohols Phenols and Ethers
7 avr 2020 · By Aliphatic Primary Amines: RCH2NH2 + HNO2 → RCH2OH + N2 + H2O Reaction with carboxylic acid (Esterification) : R-OH +R'-COOH +H Reaction with aldehyde and ketones: Glycol reacts with aldehyde and ketones |




![Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry Alcohol Reactions [Reaction Map PDF] – Master Organic Chemistry](https://imgv2-1-f.scribdassets.com/img/document/251222736/original/62a495914f/1613337934?v\u003d1)


![2 aldehydes ketones - carboxylic acids - [PDF Document] 2 aldehydes ketones - carboxylic acids - [PDF Document]](https://upload.wikimedia.org/wikipedia/commons/thumb/7/70/Acetal-protection-example.png/1200px-Acetal-protection-example.png)