alkaline ester hydrolysis mechanism
How ester hydrolysis is different from hydrolysis of alkene?
Esters susceptible to hydrolysis from aqueous acids and aqueous strong alkalis. But given products are different. Ester is broken to two parts, carboxylic acid and alcohol. Ethyl part will become ethanol. How hydrolysis of an ester is different from hydrolysis of alkene? Esters and alkenes give different products in hydrolysis.
Which acid is used for acidic ester hydrolysis?
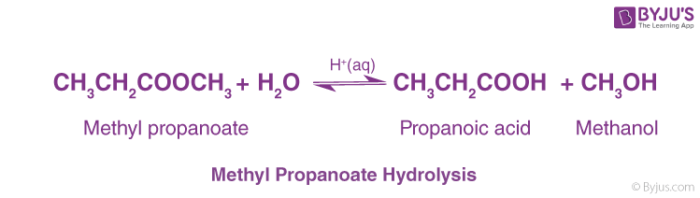
Concentrated sulfuric acid is used for acidic ester hydrolysis. This reaction is done on heating. A carboxylic acid compound and an alcohol compound are given as products. And this reaction is a reversible one. Therefore we cannot get products completely from this reaction.

Ester Hydrolysis Reaction Mechanism

Esters 6. Alkaline hydrolysis of esters (inc. saponification)

s=
|
MECHANISM OF ALKALINE HYDROLYSIS OF HINDERED
MECHANISM OF ALKALINE HYDROLYSIS OF HINDERED. AROMATIC ESTERS THE BAL2 MECHANISM. L. R. C. BARCLAY |
|
An E2 Mechanism for the Alkaline Hydrolysis of Toluene -a
09?/03?/2022 a the formation of phenyl sulphene in alkali from toluene- a-sulphonate esters of phenols with pK < 6 because the corresponding anions are ... |
|
Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in Alkaline
09?/06?/2021 The classification of ester hydrolysis mechanisms proposed by Ingold [14] was adopted: the capital letters A and B stand for acid- and base- ... |
|
Kinetics of alkaline hydrolysis of the monomethyl ester of
The alkaline hydrolysis of the monoesters of dicarboxylic acids represents a group of model reactions convenient for studying the influence of reaction. |
|
Mechanism of nucleophilic displacement at phosphorus in the
alkaline hydrolysis of phosphinate esters7 including hydrolyses in oxygen-18 enriched media which enable us to determine whether hydroxide attacks at alkyl |
|
The Effect of Structure on Kinetics and Mechanism of the Alkaline
Contribution of two types of stericeSects to the rate is indicated one of which closely parallels steric effects in ester hydrolysis rates. |
|
Carbanion mechanism of ester hydrolysis. II. o-Nitrophenyl .alpha
The second-order rate constant for alkaline hydrolysis of p-nitrophenyl acetate is within reactions of ester types la and lb with both lyate species. |
|
Rates of Alkaline Hydrolysis of a Series of Primary and Secondary
drance to ester hydrolysis because of possible ap- It seemed likely that such side reactions might be ... Alkaline. Hydrolysis of Alkyl Acetates. |
|
Alkaline Hydrolysis of Normal and Pseudo Methyl Esters of o
The rates of alkaline hydrolysis in 70% dioxane-30% water at 30° of a series of normal and pseudo addition mechanism (1) similar to that involved in. |
|
Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions
11 jui 2013 · The hydrolysis of carboxylic acid esters is one of the best- studied reactions in chemistry It is very often encountered both in the laboratory and in |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
draw the mechanism of ester hydrolysis under acidic and basic reaction conditions form new esters by base- or acid-catalysed transesterification mechanisms; |
|
The Alkaline Hydrolysis of Sulfonate Esters - CORE
26 nov 2013 · terms of both stepwise and concerted mechanisms In particular, a recent study of the alkaline hydrolysis of a series of benzene arylsulfonates |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
Hydrolysis reactions are normally sensitive to a variety a catalytic influences that include specific acid and base catalysis, general acid and base catalysis, |
|
Facile Hydrolysis of Esters with KOH-Methanol at Ambient
27 nov 2003 · Both acidic and alkaline hydrolysis are equilibrium reactions [1] However for preparative purposes, ester hydrolysis is invariably performed in |
|
Kinetics of alkaline hydrolysis of the monomethyl ester of
The alkaline hydrolysis of the monoesters of dicarboxylic acids represents a group of model reactions convenient for studying the influence of reaction medium |





![PDF] The Alkaline Hydrolysis of Sulfonate Esters: Challenges in PDF] The Alkaline Hydrolysis of Sulfonate Esters: Challenges in](https://cdn1.byjus.com/wp-content/uploads/2017/09/Ester-Hydrolysis-3-700x281.png)