amine reactions organic chemistry
How do you make an amine?
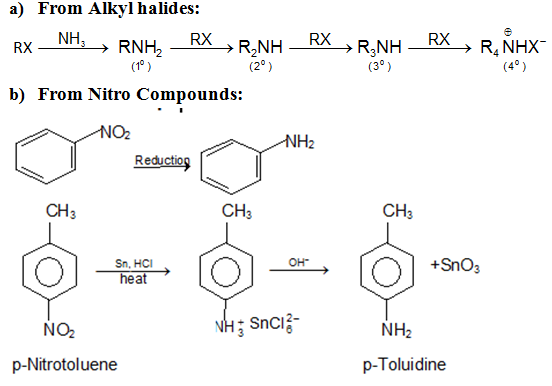
There are many ways how you can make an amine. Here, I want to go over the most common ones going from roughly the least useful to the most useful. The direct substitution of alkyl halides is a simple S N 2 reaction. In this reaction you will react primary or secondary alkyl halide with ammonia or an amine.
When should you use amine synthesis?
If you’re planning an amine synthesis, you should always try using this reaction. Especially if you’re trying to make some sort of highly functionalized amine with complex substituents on it. This reaction is very versatile and can yield a vast range of amine products.
Can amines be alkylated by acylation?
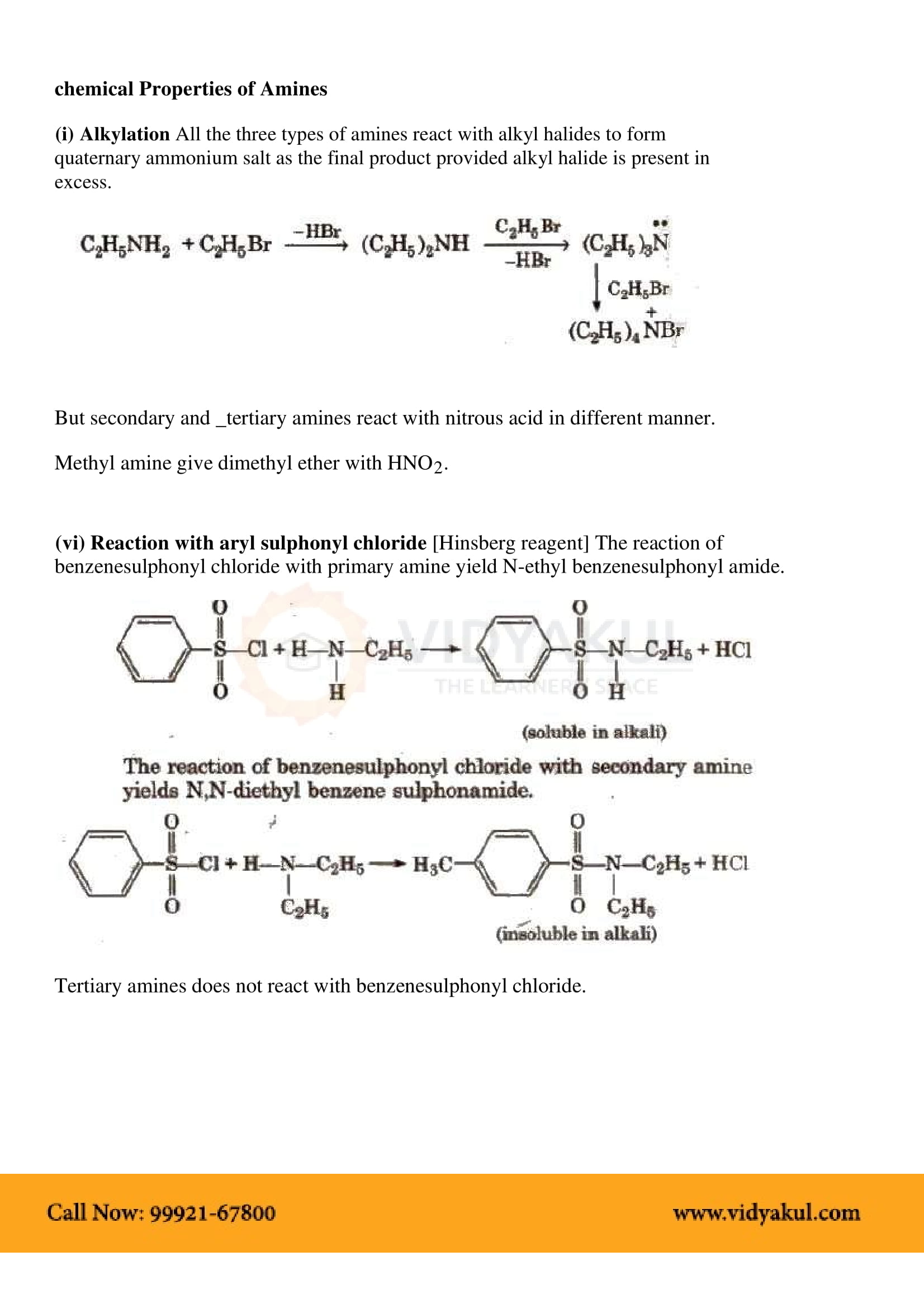
We’ve already studied the two most general reactions of amines—alkylation and acylation. As we saw earlier in this chapter, primary, secondary, and tertiary amines can be alkylated by reaction with a primary alkyl halide.

Amine Synthesis Reactions Organic Chemistry

Amine Synthesis Reactions

Amines: Crash Course Organic Chemistry #46
|
Reactions of Amines
Key: An organic will move into the organic phase if it is ionized. Why: Amine D is protonated in acid to give a water soluble ammonium ion. The oxygen compounds |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the Amines are organic derivatives of ammonia |
|
UNIT II A. Phenols* B. Aromatic Amines* C. Aromatic Acids* 10
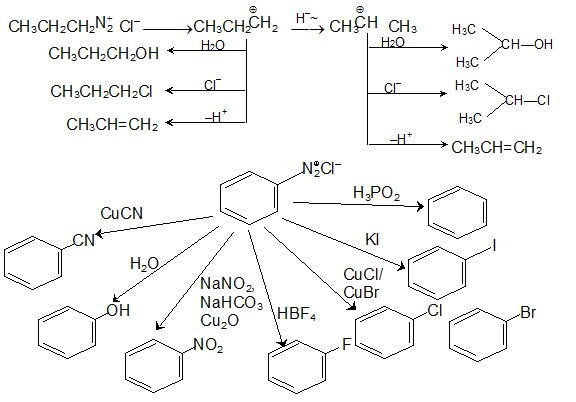
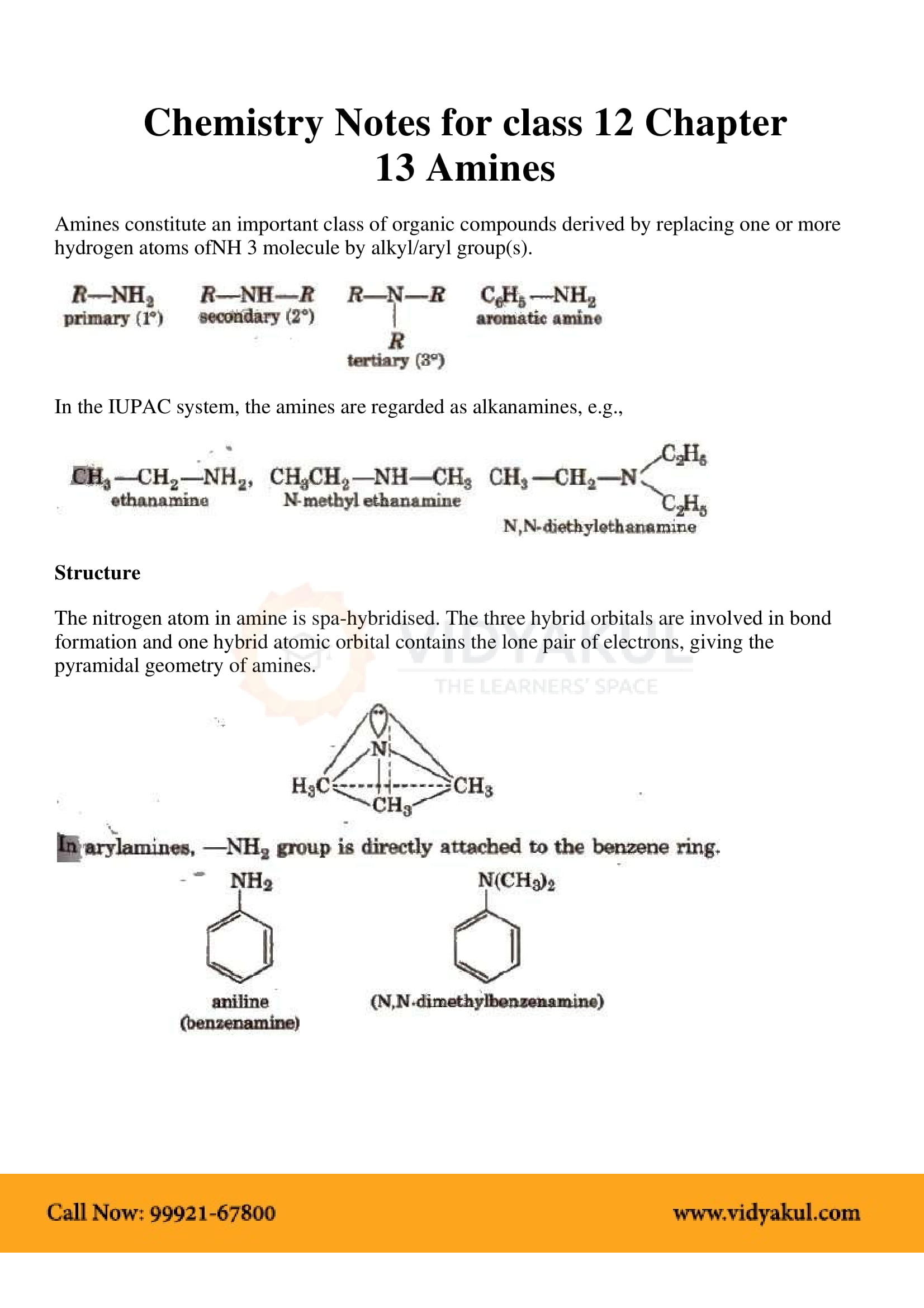
Amines constitute an important class of organic compounds derived by replacing The equilibrium constant for this reaction is called base-dissociation. |
|
Amines Amines
Amines constitute an important class of organic compounds derived by replacing one or more Write chemical equations for the following reactions:. |
|
Primary Amine Catalyzed Activation of Carbonyl Compounds: A
an understanding of the mechanisms of reaction and catalytic European Journal of Organic Chemistry published by Wiley-VCH GmbH. |
|
Reaction of Orthoesters with Amine Hydrochlorides: An Introductory
01-Mar-2016 We describe a second-semester organic chemistry laboratory experiment in which students acetylate unknown amine hydrochloride salts with ... |
|
Testsforfunctionalgroups - inorganiccompounds
Organic compounds containing > C = C < and / or — C ? C – bonds are called Both the above reactions are used as tests for unsaturation. |
|
Organic Reaction Workup Formulas for Specific Reagents
For use with acid stable target compounds only. 2. Wash the organic layer several times with 10% copper sulfate (aqueous and blue). The copper-complexed amine. |
|
145 Chapter 24: Amines Amines: Nitrogen containing organic
There are many naturally occurring organic compounds that contain nitrogen (alkaloids) Synthesis of primary amines from the reaction of alkyl halides. |
|
Organic Chemistry Specific Name Reactions - Meritnation
Organic Chemistry – Specific Name Reactions. Class XII. Sandmeyer Reaction Gabriel synthesis is used for the preparation of pure primary amines. |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Organic Chemistry II / CHEM 252 by designating the organic groups separately in front of the word amine Reaction of ammonia with an alkyl halide |
|
145 Chapter 24: Amines Amines: Nitrogen containing organic
There are many naturally occurring organic compounds that contain nitrogen Synthesis of primary amines from the reaction of alkyl halides or tosylates with |
|
Reactions of Amines
Key: An organic will move into the organic phase if it is ionized Why: Amine D is protonated in acid to give a water soluble ammonium ion The oxygen compounds |
|
Amines Amines - NCERT
Amines constitute an important class of organic compounds derived by replacing one or more hydrogen atoms of ammonia molecule by alkyl/aryl group(s) In nature, they occur among proteins, vitamins, alkaloids and hormones Synthetic examples include polymers, dyestuffs and drugs |
|
CHEM 2211 Organic Chemistry I: Chapter 7 Summary of Reactions
X • Best to prep 1o amines and quaternary ammonium salts Page 4 Direct nucleophilic substitution Page 5 • S N 2 mechanism • Best for CH 3 X or RCH |
|
Chapter 17: Amines and Amides
17 1 Bonding Characteristics of Nitrogen Atoms in Organic Compounds Nitrogen atoms aromatic amines and the simplest aromatic amine is the aniline Alkanamines organic a) Reaction of ammonia with alkyl halides b) Reduction of |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Predict the products for the reactions of amines with water or with strong acids 6 Organic compounds are categorized into various ______ by the presence of |
|
Lecture 10 Carbon-Nitrogen Bonds Formation I - NPTEL
NPTEL – Chemistry – Principles of Organic Synthesis adduct depends on the structure of the aldehydes, amine, and the reaction conditions For examples |
|
Chapter 19 – Amines
Amines are classified as 1°, 2°, or 3° based on how many R groups are attached to the o Mr Baker likes to ask questions about what would be left in the organic or aqueous o The problem with this reaction is that it's difficult to control how many alkyl groups add Synthesis of amines by acylation-reduction o Just what |