and purification of an ester pre lab answers
|
Synthesis Isolation and Purification of an Ester
An ester is a chemical compound that is f o r med when an organic acid reacts with an alcohol E s t e r s fre q u e n t l y have distinctive odors and are na t u ra l l y occurring f l a vor and fragrance c h e m i c a l s in many fruits and plants In this e x p e ri m e n t the ester ethyl acetate (ethyl ethanoate) is pre p a re d |
How do you prepare a fruity ester?
Provide a selection of carboxylic acids (acetic acid, propionic acid, benzoic acid, salicylic acid, and anthranilic acid) as well as alcohols (methyl alcohol, ethyl alcohol, propyl alcohol, isoamyl alcohol, and octyl alcohol). Ask students to research natural fruity esters and select appropriate combinations to prepare them.
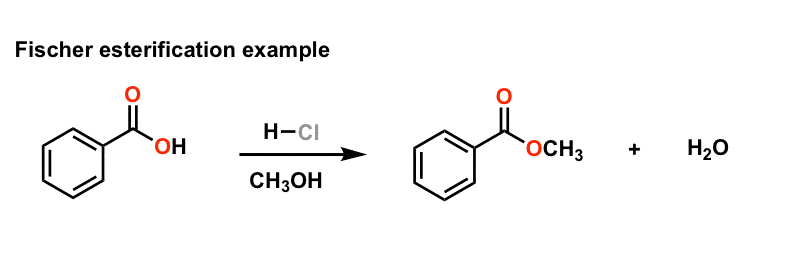
How is ester produced from Fisher esterification?
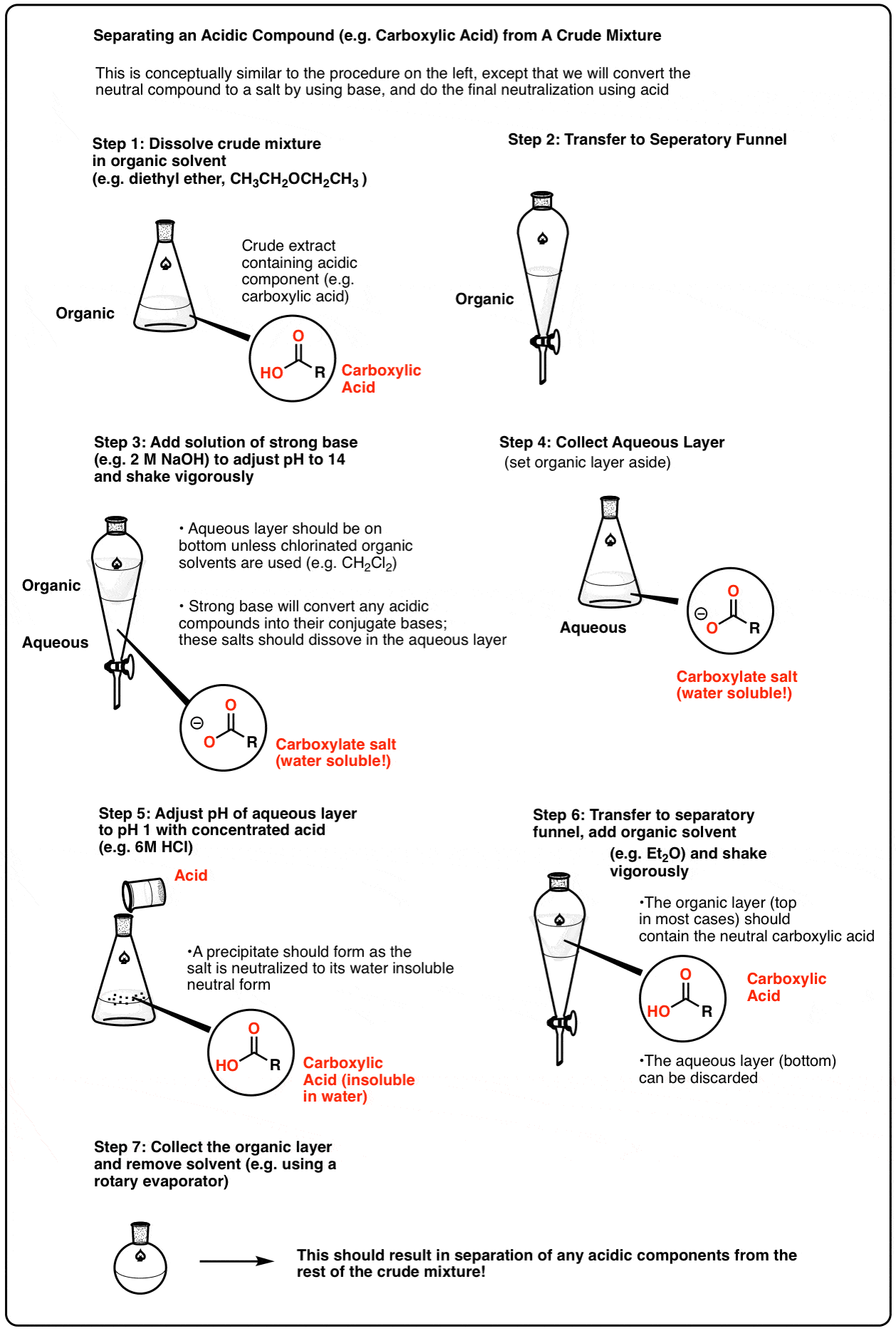
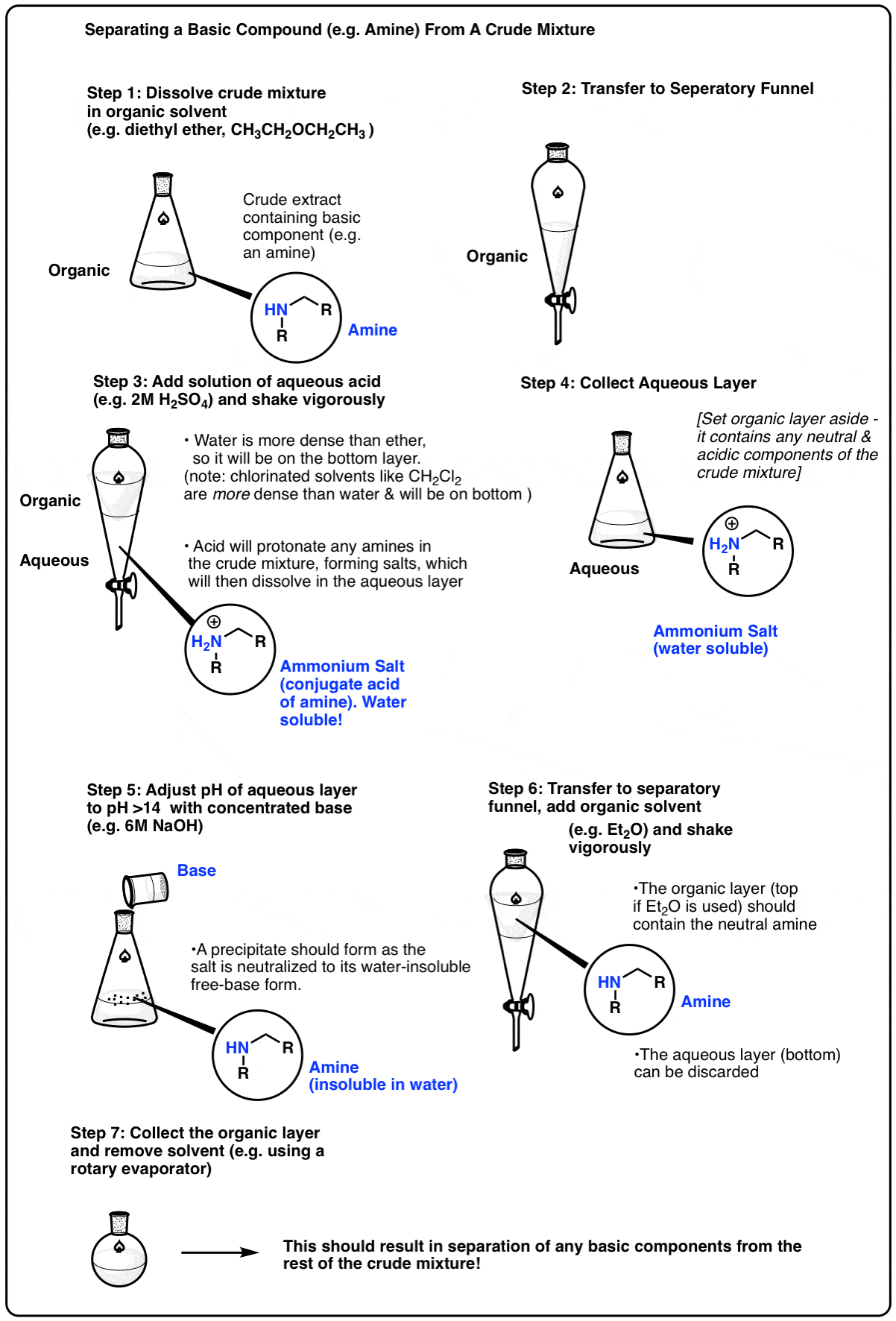
The ester produced from Fisher esterification will be in an equilibrium mixture with unreacted alcohol and carboxylic acid. Sulfuric acid is also present as catalysts are not consumed in chemical reactions. Ester is isolated from the mixture by using a separating funnel.
What happens if an ester is separated from an aqueous layer?
After the ester-containing organic layer is separated from the aqueous layer, purification should be carried out. This is because unreacted alcohol and carboxylic acid can be found in the organic layer (particularly those with lower solubility in water).
Fisher Esterification
Fisher esterification refers to the reaction between an alcohol and a carboxylic acid which produces an ester and water. The reaction is considered a condensation reactiondue to the production of a small molecule i.e. water alongside the main organic product. Condensation reactions can produce molecules other than water. 1. slow 2. reversible 3. ex
Role of Concentrated H2SO4
Concentrated sulfuric acid acts as acatalyst: increases reaction rate by lowering the activation energy. As a result the reaction rate is increased. scienceready.com.au
Reaction Conditions For Esterification
Esterification is conducted under heat with refluxat 140 – 180ºC. Heat is required to meet the activation energy of the reaction and increase reaction rate. Heat should be supplied using a heating mantle (hotplate). Bunsen burner should notbe used in esterification because alcohol is flammable. Reflux is the process of condensing gaseous products b
Why Is Reflux Important?
Reflux is important for several reasons: 1. Allows heat to be used and therefore increases the reaction rate. 1. Prevents the loss of volatile substances e.g. ester,alcohol, carboxylic acid by returning them into liquid states (condensation). As a result, the yield of ester is increased. 1. Allows esterification to be carried out in an open chemica
Isolation of Ester
The ester produced from Fisher esterification will be in an equilibrium mixture with unreacted alcohol and carboxylic acid. Sulfuric acid is also present as catalysts are not consumed in chemical reactions. Ester is isolated from the mixture by using a separating funnel. scienceready.com.au
Purification of Ester
After the ester-containing organic layer is separated from the aqueous layer, purification should be carried out. This is because unreacted alcohol and carboxylic acid can be found in the organic layer (particularly those with lower solubility in water). scienceready.com.au

Esters 4. Organic Preparation & Purification of an Ester.

Saponification

Nomenclature and properties of esters Organic chemistry Khan Academy
| A SN1 Reaction: Synthesis of tert-Butyl Chloride |
| A SN1 Reaction: Synthesis of tert-Butyl Chloride |
| Lab 14: Qualitative Organic Analysis |
|
Ester Synthesis Lab (Student Handout)
Ester Synthesis Lab (Student Handout). Lab Report Components: Esters are produced by the reaction between alcohols and carboxylic acids. For example. |
|
Synthesis of paracetamol by acetylation
As a pre-lab assignment students should make a table showing the Any residual alcohol can interfere with the purification steps and the corresponding. |
|
Lab 12: Synthesis of an Ester
Purify a compound via microscale fractional distillation. In this experiment you will react a carboxylic acid and an alcohol under ... Lab Report”. |
| Separation purification and identification of the components of a |
|
5.310 F17 Experiment 5: Fischer Esterification
EXPERIMENT #5. Fischer Esterification: The Organic Synthesis Isolation |
| Chemistry |
|
Protein Purification by Affinity Chromatography
5. Carboxylic acid ester derivatives of various ligands have also been linked to agarose by the proce- dures described in this report. Such bonds can be cleaved. |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
In this experiment you will react a carboxylic acid and an alcohol under acidic brine and anhydrous sodium sulfate, the ester will be purified by fractional distillation For solutions, include the concentration and volume 4 Lab Report” |
|
5310 F17 Experiment 5: Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and This angular momentum results in a technique for you during the pre-lab lecture |
|
Synthesis, Isolation, and Purification of an Ester
In this experiment the ester ethyl acetate (ethyl ethanoate) is prepared and purified by distillation Concepts • Esters • Theoretical yield • Distillation In the pre-lab Question #6, part c, the theoretical yield of ethyl acetate was calculated |
|
5310 F17 Experiment 5: Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and This angular momentum results in a technique for you during the pre-lab lecture |
|
PREPARATION OF AN ESTER EXPERIMENT 27
You can synthesize an ester in the lab by heating a carboxylic acid in an alcohol solution containing a small amount of strong acid as a catalyst R—COOH +1 R' — |
|
Experiment 18 — Synthesis of Esters
Be sure to leave enough time for the pre-lab work; you will not be able to of your reactants is water-soluble, that can be removed from the ester by extraction |
|
Synthesis, Isolation and Purification of an Ester - Flinn Scientific
Low molecular weight esters are responsible for the pleasant odor or fragrance of many fruits and The inquiry process begins with pre-lab preparation to determine the quantities of just one of the variables to obtain the “best” results |
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification, wherein a experiment, lauric acid (dodecanoic acid) is converted to ethyl laureate The aqueous layer from ether extraction: let the solution sit in the hood to allow ether to |
|
Esters An Introduction
mixture – 7 esters, 3 carboxylic acids, and 7 essential oils (other natural extracts) In some In practice often a 10:1 or 1:10 ratio is used, which results in even An extraction process removes sulfuric acid and a ESSENTIAL: Prelab Work |