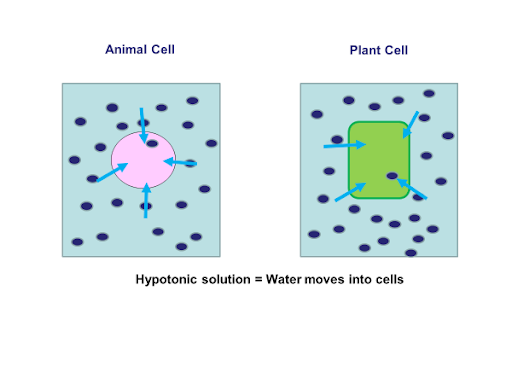
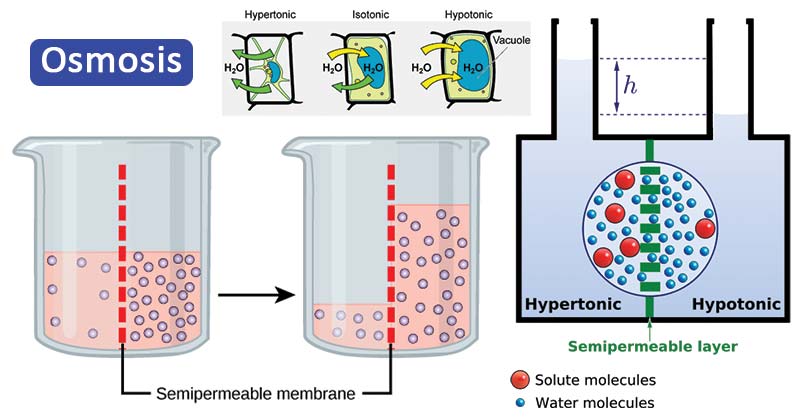
animal cell in hypertonic solution
What is a hypotonic solution?
The opposite solution with a lower concentration is known as the hypotonic solution. Scientists must describe cell contents compared to the environment. If a cell is placed in a hypertonic solution, the cell is considered hypotonic. as the water moves between the two solutions
What happens if you put an animal cell in a hypertonic solution?
The cell would then expand and eventually lyse or burst. What happens when you place an animal cell in a hypertonic solution? In this case, water will leave the cell since the cell has a lower osmolarity than the extracellular fluid. As a result the cell would shrink in what is called plasmolysis.
Why do animals swell and burst in a hypotonic solution?
Animal cells swell and burst in a hypotonic solution. Animal cells in hypertonic solutions (with higher solute concentrations than the cytosol) shrivel up as water leaves the cell. From this brief description, you should conclude that water crosses from the hypotonic to the hypertonic side of a membrane.
What happens if a cytosol becomes hypertonic?
Too many solutes and the cytosol will become a hypertonic solution compared to the environment. Cells without cell walls can burst in this condition. Too few solutes in the environment will become the hypertonic solution. In this case, the opposite will happen, as water moves out of the cell.
Overview
Osmosis and tonicity. Hypertonic, isotonic, and hypotonic solutions and their effect on cells. khanacademy.org
Introduction
Have you ever forgotten to water a plant for a few days, then come back to find your once-perky arugula a wilted mess? If so, you already know that water balance is very important for plants. When a plant wilts, it does so because water moves out of its cells, causing them to lose the internal pressure—called turgor pressure—that normally supports the plant. Why does water leave the cells? The amount of water outside the cells drops as the plant loses water, but the same quantity of ions and other particles remains in the space outside the cells. This increase in solute, or dissolved particle, concentration pulls the water out of the cells and into the extracellular spaces in a process known as osmosis. khanacademy.org
How it works
Why does water move from areas where solutes are less concentrated to areas where they are more concentrated? This is actually a complicated question. To answer it, let’s take a step back and refresh our memory on why diffusion happens. In diffusion, molecules move from a region of higher concentration to one of lower concentration—not because they’re aware of their surroundings, but simply as a result of probabilities. When a substance is in gas or liquid form, its molecules will be in constant, random motion, bouncing or sliding around one another. If there are lots of molecules of a substance in compartment A and no molecules of that substance in compartment B, it’s very unlikely—impossible, actually—that a molecule will randomly move from B to A. On the other hand, it’s extremely likely that a molecule will move from A to B. You can picture all of those molecules bouncing around in compartment A and some of them making the leap over to compartment B. So, the net movement of molecules will be from A to B, and this will be the case until the concentrations become equal. In the case of osmosis, you can once again think of molecules—this time, water molecules—in two compartments separated by a membrane. If neither compartment contains any solute, the water molecules will be equally likely to move in either direction between the compartments. But if we add solute to one compartment, it will affect the likelihood of water molecules moving out of that compartment and into the other—specifically, it will reduce this likelihood. Why should that be? There are some different explanations out there. The one that seems to have the best scientific support involves the solute molecules actually bouncing off the membrane and physically knocking the water molecules backwards and away from it, making them less likely to cross1,2 . Regardless of the exact mechanisms involved, the key point is that the more solute water contains, the less apt it will be to move across a membrane into an adjacent compartment. This results in the net flow of water from regions of lower solute concentration to regions of higher solute concentration. This process is illustrated in the beaker example above, where there will be a net flow of water from the compartment on the left to the compartment on the right until the solute concentrations are nearly balanced. Note that they will not become perfectly equal in this case because the hydrostatic pressure exerted by the rising water column on the right will oppose the osmotic driving force, creating an equilibrium that stops short of equal concentrations. khanacademy.org
Osmolarity
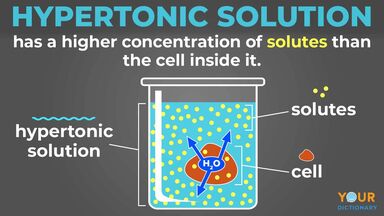
Osmolarity describes the total concentration of solutes in a solution. A solution with a low osmolarity has fewer solute particles per liter of solution, while a solution with a high osmolarity has more solute particles per liter of solution. When solutions of different osmolarities are separated by a membrane permeable to water, but not to solute, water will move from the side with lower osmolarity to the side with higher osmolarity. Three terms—hyperosmotic, hypoosmotic, and isoosmotic—are used to describe relative osmolarities between solutions. For example, when comparing two solution that have different osmolarities, the solution with the higher osmolarity is said to be hyperosmotic to the other, and the solution with lower osmolarity is said to be hypoosmotic. If two solutions have the same osmolarity, they are said to be isoosmotic. khanacademy.org
Tonicity
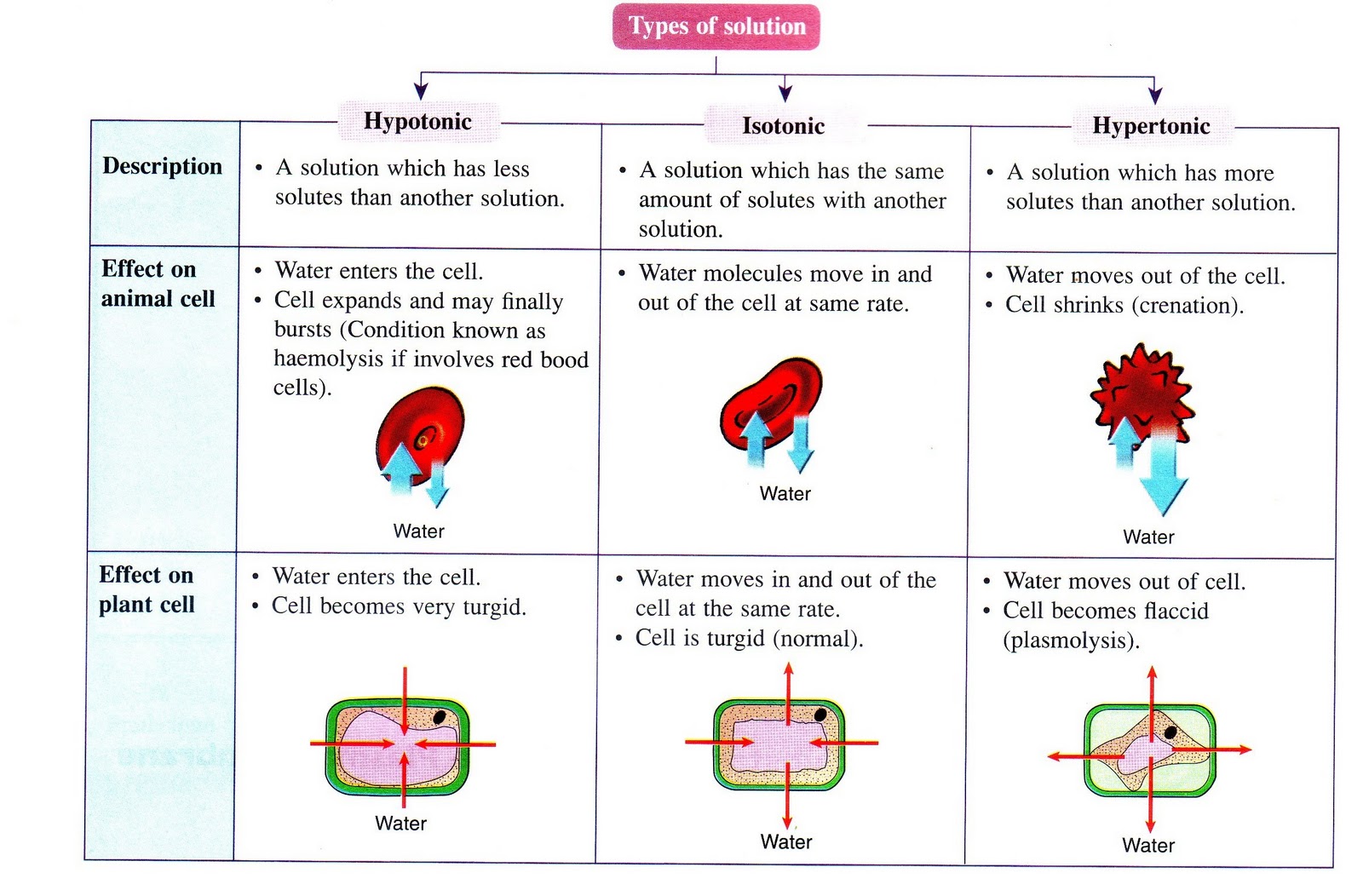
In healthcare settings and biology labs, it’s often helpful to think about how solutions will affect water movement into and out of cells. The ability of an extracellular solution to make water move into or out of a cell by osmosis is known as its tonicity. Tonicity is a bit different from osmolarity because it takes into account both relative solute concentrations and the cell membrane’s permeability to those solutes. Three terms—hypertonic, hypotonic, and isotonic—are used to describe whether a solution will cause water to move into or out of a cell: If a cell is placed in a hypertonic solution, there will be a net flow of water out of the cell, and the cell will lose volume. A solution will be hypertonic to a cell if its solute concentration is higher than that inside the cell, and the solutes cannot cross the membrane. If a cell is placed in a hypotonic solution, there will be a net flow of water into the cell, and the cell will gain volume. If the solute concentration outside the cell is lower than inside the cell, and the solutes cannot cross the membrane, then that solution is hypotonic to the cell. khanacademy.org
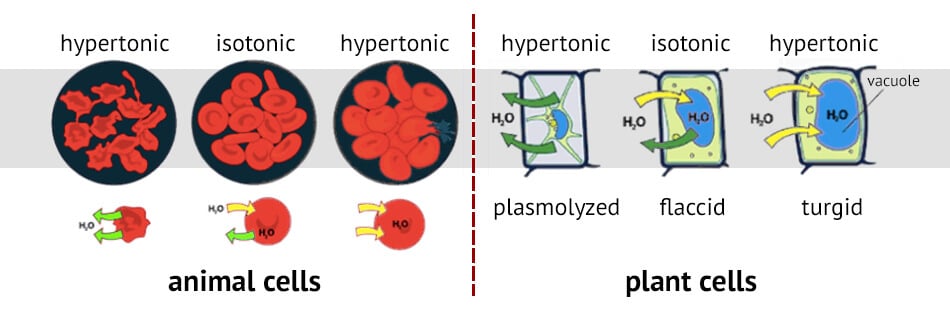
Tonicity in living systems
If a cell is placed in a hypertonic solution, water will leave the cell, and the cell will shrink. In an isotonic environment, there is no net water movement, so there is no change in the size of the cell. When a cell is placed in a hypotonic environment, water will enter the cell, and the cell will swell. In the case of a red blood cell, isotonic conditions are ideal, and your body has homeostatic (stability-maintaining) systems to ensure these conditions stay constant. If placed in a hypotonic solution, a red blood cell will bloat up and may explode, while in a hypertonic solution, it will shrivel—making the cytoplasm dense and its contents concentrated—and may die. In the case of a plant cell, however, a hypotonic extracellular solution is actually ideal. The plasma membrane can only expand to the limit of the rigid cell wall, so the cell won't burst, or lyse. In fact, the cytoplasm in plants is generally a bit hypertonic to the cellular environment, and water will enter a cell until its internal pressure—turgor pressure—prevents further influx. Maintaining this balance of water and solutes is very important to the health of the plant. If a plant is not watered, the extracellular fluid will become isotonic or hypertonic, causing water to leave the plant's cells. This results in a loss of turgor pressure, which you have likely seen as wilting. Under hypertonic conditions, the cell membrane may actually detach from the wall and constrict the cytoplasm, a state called plasmolysis (left panel below). Tonicity is a concern for all living things, particularly those that lack rigid cell walls and live in hyper- or hypotonic environments. For example, paramecia—pictured below—and amoebas, which are protists that lack cell walls, may have specialized structures called contractile vacuoles. A contractile vacuole collects excess water from the cell and pumps it out, keeping the cell from lysing as it takes on water from its hypotonic environment. [Attribution and references] khanacademy.org
Want to join the conversation?
Log in khanacademy.org

Hypertonic Hypotonic and Isotonic Solutions!

Types of Solutions-Isotonic-Hypertonic-Hypotonic-Animation

Hypertonic Hypotonic and Isotonic Solutions and Their Effects on Cells
|
Cellular Transport Review
When water leaves a plant cell the osmotic pressure will . increase decrease. The shrinking of ANIMAL cells that are placed in a HYPERTONIC solution is called |
|
Principles of Biology BIOL001 Exam I I
hypotonic solution and the plant cell is in an isotonic solution. b. The animal cell is in an isotonic solution |
|
Principles of Biology BIOL001 Exam I I
The animal cell is in a hypertonic solution and the plant cell is in a hypotonic solution. 18. Celery stalks that are immersed in fresh water for several |
|
IB Questionbank
Outline the effects of putting plant tissue in a hypertonic solution. Deduce with a reason |
|
TRANSPORT IN CELLS
20. What is different about a plant cell placed in a hypotonic solution and an animal cell? 21. Considering the difference in the structure of plant & |
|
A Cell Placed In A Hypotonic Solution Will (2023) ; www1
Look” to see plant and animal cells in hypotonic environments on the bottom the onion cells in hypertonic solution: WebHypotonic solution has less solute ... |
|
Phospholipid bilayer
Sep 17 2012 Osmosis! Osmosis. More free water. Less free water. Hypotonic solution. Isotonic solution. Hypertonic solution. Animal cell. ... Plant cell. Plant ... |
|
Untitled
solution the cell has been placed into (isotonic hypotonic or hypertonic). ... What keeps plant cells from bursting when they are placed in a hypotonic solution? |
|
UGA Extension
In this case water moves equally into the cell and out of the cell. Figure 3. Three cases of water movement. EXPLANATION hypotonic solution hypertonic. |
|
Untitled
Placing an animal cell in a hypotonic solution will cause water to The shrinking of a plant cell membrane away from the cell wall when placed in a hypertonic ... |
|
Q1.A student investigated the effect of different sugar solutions on
Diagram B shows the same cell in a hypertonic solution. Diagram A. Diagram B (b) What word is used to describe plant cells placed in: (i) a hypotonic ... |
|
What Happens to Plant & Animal Cells When Placed in Hypertonic
The diffusion of water is referred to as osmosis. HYPERTONIC SOLUTIONS. Page 2. A solution is hypertonic to a cell if it has a |
|
Cellular Transport Review-kEY.pdf
When water leaves a plant cell the osmotic pressure will . increase decrease. The shrinking of ANIMAL cells that are placed in a HYPERTONIC solution is called |
|
Cell Membrane & Tonicity Worksheet
__A __ solution in which the solute concentration is the same. B. Hypertonic. __ A__ condition plant cells require. __ A__ condition that animal cells |
|
Untitled
Animal cells contain a nucleus cytoplasm |
|
TRANSPORT IN CELLS
Transport in Cells B1YvM2. 4. MODEL 3: Osmosis in Plant and Animal Cells. External solution: Hypertonic. Isotonic. Hypotonic. Animal. Cell. |
|
Determining the Water Potential of Potato Cells
If a plant cell has a lower water potential than its surrounding environment and if pressure is equal to zero is the cell hypertonic (in terms of solute |
|
Identification of Biology Students Misconception in Makassar State
plant cell walls with 3 forms of misconceptions including (1) Animal cells and plant cells placed in a solution with hypotonic osmosis causing normal plant |
|
Untitled
In a hypotonic solution animal cells experience osmosis and a pressure builds up in the cell. What causes the pressure? (Be specific). Hypotonic solution =. |
|
Untitled
Label the type of solution hypertonic hypotonic |
|
Biology osmosis wspdf - Crestwood Local Schools
In a hypotonic solution, animal cells experience osmosis and a pressure builds up in the cell What causes the pressure? (Be specific) Hypotonic solution |
|
Cellular Transport Review
When water leaves a plant cell, the osmotic pressure will increase decrease The shrinking of ANIMAL cells that are placed in a HYPERTONIC solution is called |
|
TRANSPORT IN CELLS
What is different about a plant cell placed in a hypotonic solution and an animal cell? 21 Considering the difference in the structure of plant animal cells, do you |
|
Part 1 Osmosis Practice Key - Mayfield City Schools
Label the type of solution hypertonic, hypotonic, or isotonic on the line below the picture 3 Explain what happens to animal cells if they are put in fresh water |
|
PLANT CELLS & OSMOSIS - Esalq
Thus, a hypotonic solution has more water than the cell and water has a tendency to move (diffuse) into the cell Placing Elodea cells into 100 water, which is |
|
LAB TWO
A Hypertonic solution has more solute (so LESS water) than the cell A cell placed in ]In animal cells, a hypertonic environment forces water to leave the cell so |
|
Osmosis & Diffusion, What you need to know:
See also all your cell membrane, diffusion, and osmosis-related handouts 1 is why plant cells do not burst like animal cells do when in a hypotonic solution) |
|
Potato Osmosis - California State University, Bakersfield
plant cell; this is due to the cell wall that plant cells have When an animal cell is in a hypotonic solution, it usually swells up and since there is no cell wall to |
|
Cell Membrane & Tonicity Worksheet
__A __ solution in which the solute concentration is the same B Hypertonic __ A__ condition plant cells require __ A__ condition that animal cells require |



















![Solved] Questions 21 Compare and contrast the reaction the animal Solved] Questions 21 Compare and contrast the reaction the animal](https://img.yumpu.com/31018901/1/500x640/osmosis-and-tonicitypdf.jpg)