base hydrolysis of aspirin mechanism
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
base catalysis includes a term for each conjugate base in the reaction Aspirin I acetylsalicylic acid is an ester The equation for its hydrolysis to salicylic and acetic acids may be written very simply The exact mechanism of hydrolysis is a bit more difficult to describe since the hydrolysis of aspi- |
What is the difference between benzoic acid and aspirin hydrolysis?
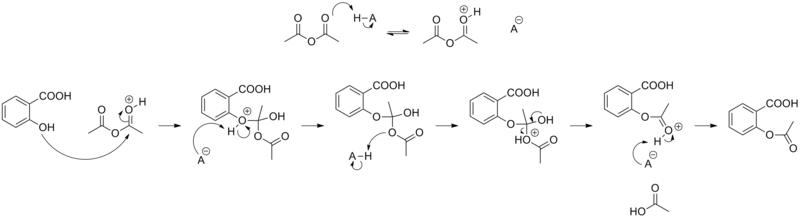
Aspirin hydrolysis reaction can occur with acidic or basic condition. Catalysis also occurs where an un-dissociated acid exists in the transition state of the reaction. This is termed general acid catalysis. Below is a typical mechanism involving an un-dissociated acid (benzoic acid).
Is ASPI-Rin acetylsalicylic acid a salicylic or acetic acid?
Aspirin I, acetylsalicylic acid, is an ester. The equation for its hydrolysis to salicylic and acetic acids may be written very simply. The exact mechanism of hydrolysis is a bit more difficult to describe, since the hydrolysis of aspi-rin may occur by one or more of the mechanisms described above.
What is the mechanism of hydrolysis of aspirin?
The exact mechanism of hydrolysis is a bit more difficult to describe, since the hydrolysis of aspi-rin may occur by one or more of the mechanisms described above. It is the purpose of this experi-ment to determine what effect a change in pH (ie a change in [H3O+] or [OH-]) will have on the rate of aspirin hydrolysis.
What is the catalysis of acetate with aspirin?
Catalysis of the hydrolysis by oxyanions is observed, including weak catalysis by acetate ion. This reaction of acetate with aspirin is compared with the corresponding reaction with phenyl acetate, which is known to involve general base catalysis, and with aspirin hydrolysis.
|
Hydrolysis of aspirin. Intramolecular general base catalysis of ester
mechanism of hydrolysis of aspirin is intramolecular general base catalysis by the carboxylate group. Specifically the reaction is thought to involve |
|
Structure and mechanism in intramolecular catalysis. Hydrolysis of
mechanism may allow definite conclusions about the mechanism of intramolecular catalysis in the hydrolysis of aspirin. Experimental Section. Materials. |
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
the reaction mixture. A similar situation exists for basic hydrolysis (the mechanism is on the top of the next page). When the base is hydroxide (OH-) |
|
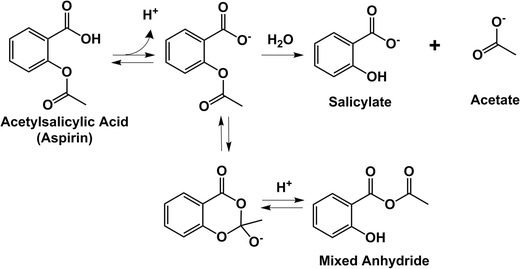
Stability Studies of Lysine Acetylsalicylate (Aspirin Derivative
The pH-rate profile (log kobs = f(pH)) shows that the hydrolysis reaction of the LAS |
| THE HYDROLYSIS OF ASPIRIN |
|
191 STUDY OF HYDROLYSIS OF ACETYLSALICYLIC ACID Aspirin
The acetylsalicylic acid hydrolysis reaction is a Mechanism of hydrolysis acetilsalicylic acid ... Hydrolysis of Aspirin. In-tramolecular General Base. |
|
Series nucleophilic catalysis in the hydrolysis of 3-acetoxyphthalate
The hydrolysis of aspirin involves intramolecular general base catalysis by the carboxylate group and no contribution from the nucleophilic mechanism can. |
|
Intramolecular nucleophilic catalysis of ester hydrolysis by the
boring ester function as a general base rather than as a nucleophile. Oxy anions |
|
Mechanism of Acetyl Salicylic Acid (Aspirin) Degradation under
14 avr. 2016 The mechanism of degradation of acetylsalicylic acid and its reaction pathway ... the predominant species at neutral or basic pH levels [4]. |
|
HYDROLYSIS 2016.pdf
Hydrolysis reactions are generally enhanced by both acids and bases and three independent reaction mechanisms account for neutral acid and base hydrolysis. |
|
Catalysis: The pH-Rate Dependence of the Hydrolysis of Aspirin
Note that there is no term in the equation for any undissociated acid present in the reaction mixture A similar situation exists for basic hydrolysis (the mechanism |
|
191 STUDY OF HYDROLYSIS OF ACETYLSALICYLIC ACID Aspirin
The acetylsalicylic acid hydrolysis reaction is a first order reaction, proven by the value of the reaction velocity constant relatively constant In this experience it is observed that the acetylsalicylic acid hydrolysis reaction is faster at acid pH and at 60 ° C |
|
Rate of hydrolysis of aspirin
Aspirin (2-ethanoyloxybenzoic acid or acetylsalicylic acid) hydrolyses to produce 2-hydroxybenzoic acid and ethanoic acid Here is the equation for the reaction: |
|
Hydrolysis as a Function of pH - OECD
12 mai 1981 · Aspirin – Diazinon These substances need not be employed in all cases The hydrolysis reaction may be influenced by acidic or basic species H30+ (H+) and OH-, lives are independent of concentration (equation 3) |
|
Hydrolysis of Acetylsalicylic Acid (Aspirin) - Diman Regional
Lab: Hydrolysis of Acetylsalicylic Acid (Aspirin) to Salicylic Acid You will receive a portion of crystalline acetylsalicylic acid (C9H8O4) Measure the mass in |

![PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin](https://imgv2-2-f.scribdassets.com/img/document/461824299/298x396/5dfdc28885/1589719714?v\u003d1)


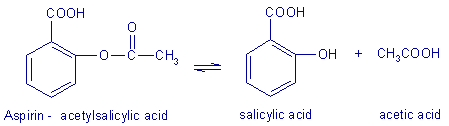





![Full text] New insights into the mechanisms of action of aspirin Full text] New insights into the mechanisms of action of aspirin](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fbjc.2014.271/MediaObjects/41416_2014_Article_BFbjc2014271_Fig1_HTML.jpg)
![POSTLAB] Experiment 3 - Kinetics of Aspirin Hydrolysis - Google POSTLAB] Experiment 3 - Kinetics of Aspirin Hydrolysis - Google](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/asset/186624ed-445f-4481-a14c-06350ea5d983/gr1.jpg)


![PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin PDF] Stability Studies of Lysine Acetylsalicylate (Aspirin](https://www.intechopen.com/media/chapter/72930/media/F5.png)



![Full text] New insights into the mechanisms of action of aspirin Full text] New insights into the mechanisms of action of aspirin](https://i.ytimg.com/vi/QSVCHVjWEyw/maxresdefault.jpg)
