base promoted hydrolysis of methyl acetate
What is hydrolysis of ethyl propanoate in a sodium hydroxide solution?
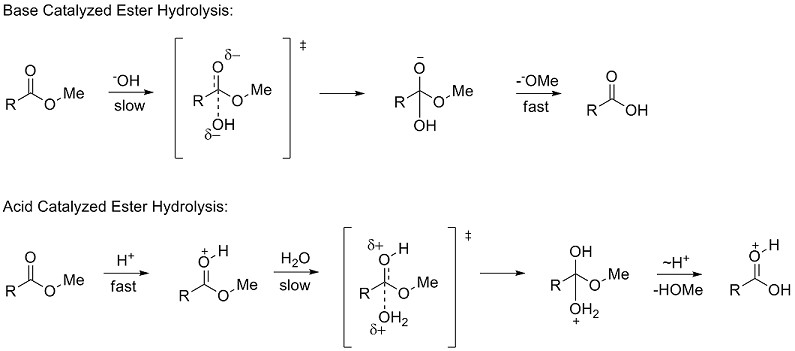
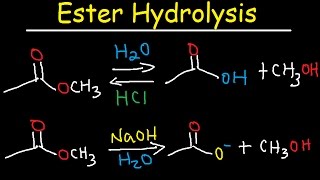
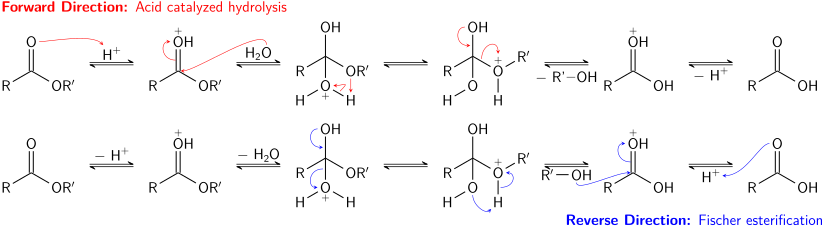
Write the equation for the hydrolysis of ethyl propanoate in a sodium hydroxide solution. Hydrolysis is a most important reaction of esters. Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol. Basic hydrolysis of an ester gives a carboxylate salt and an alcohol.

Saponification

Ester Hydrolysis Reaction Mechanism

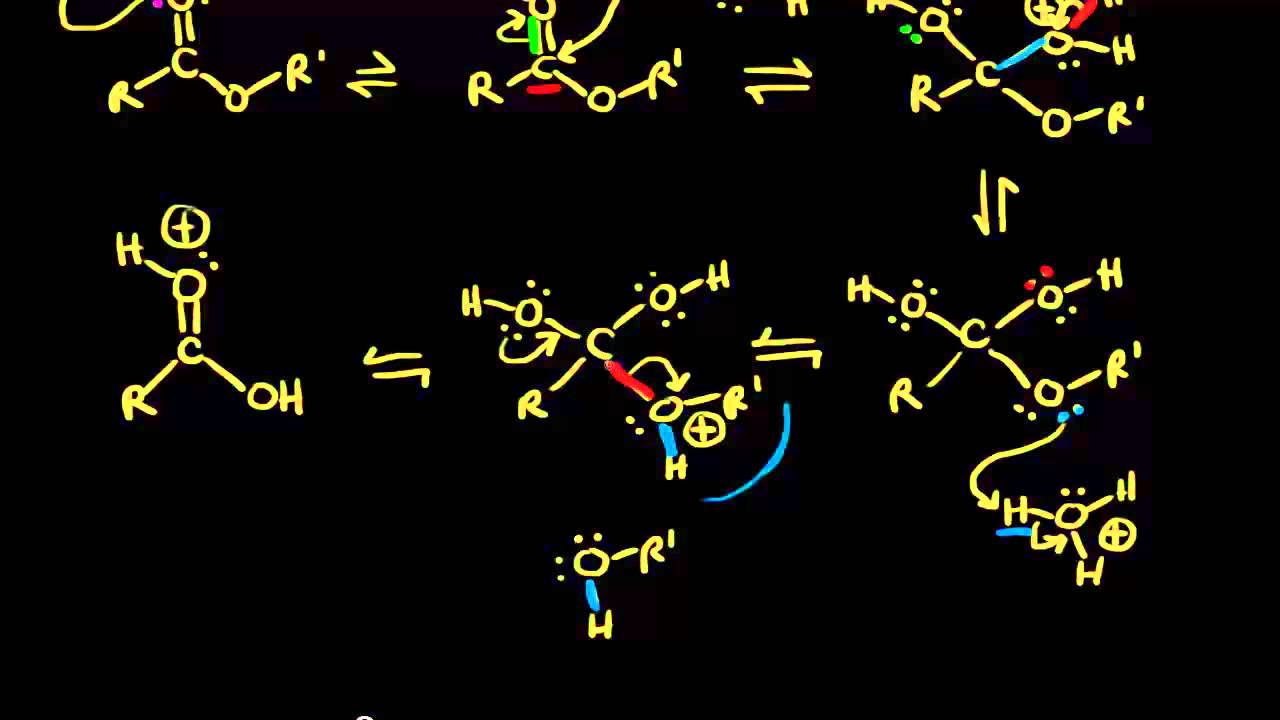
Acid and base-catalyzed hydrolysis of amides Organic chemistry Khan Academy
|
The Acid Hydrolysis of Methyl Acetate in Dioxane-Water Mixtures
For this purpose the acid-catalyzed hydrolysis of a simple ester (methyl acetate) has been selected. Dioxane is apparently an inert solvent |
|
Reaction Pathways and Energy Barriers for Alkaline Hydrolysis of
energy barriers for the base-catalyzed hydrolysis of alkyl esters and explore the role of solvent water in the hydrolysis process taking methyl acetate and |
|
Hydrogen secondary isotope effect in acyl transfer reactions. Origins
Margolin for the hydroxide-promoted hydrolysis of ethyl acetate has been observed also ported here to note that (a) only the basic hydrolysis of methyl. |
|
Experiment 5
To determine the rate constant for the acid-catalyzed hydrolysis of methyl acetate. THEORY. Methyl acetate undergoes hydrolysis in the presence of an acid |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VII
Sampling system for high-pressure apparatus. TABLE I. Acid-catalyzed hydrolysis of methyl acetate in aqueous solution at 35.00" C with 0.1-M |
|
High pressure studies. XVI. Hydrolysis of p-nitrophenyl esters
but in a different order than found for base-catalyzed hydrolysis: (ester AV* cat) acetate |
|
Nonlinear Structure-Reactivity Correlations. The Imidazole
for base catalysis of imidazole-catalyzed hydrolysis varies in a regular manner as the leaving group is varied in the series of acetate esters. |
|
A DFT investigation of methanolysis and hydrolysis of triacetin
At least two reports exist on the mechanism of base-catalyzed methanolysis of methyl acetate based on the previous hydrolysis work of Zhan and coworkers |
|
Hydration and hydrolysis of methyl pyruvate. A bifunctional probe of
acetate arsenate |
|
Cleavage of the Alkyl-Oxygen Bond in the Hydrolysis of Esters. t
The base-catalyzed hydrolysis ester hydrolysis is made by cleavage of the alkyl-oxygen bond. ... hydrolysis falls off that of methyl acetate being. |
|
Hydrolysis of Methyl Acetate
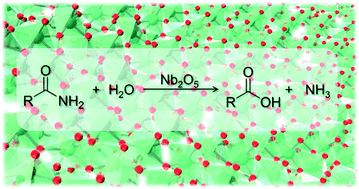
Methyl acetate hydrolyzes in water to give methanol and acetic acid, according to the The reaction is catalyzed by hydrogen ions and, in fact, it does not proceed with neutral salts, inorganic bases, or organic materials may be studied |
|
Experiment 5
To determine the rate constant for the acid-catalyzed hydrolysis of methyl acetate THEORY Methyl acetate undergoes hydrolysis, in the presence of an acid |
|
Determination of adsorption and kinetic parameters for methyl
for methyl acetate esterification and hydrolysis reaction catalyzed by Amberlyst 15 hydrolysis of methyl acetate and recovery of acetic acid Like reactive |
|
KINETIC STUDY OF CATALYTIC HYDROLYSIS - ResearchGate
Hydrolysis of methyl acetate (MeOAc) to acetic acid and methanol is one of the The methyl acetate hydrolysis reaction catalyzed by Amberlyst 15 has been |
|
A COMPREHENSIVE STUDY OF ESTERIFICATION AND - CORE
catalyzed by Amberlyst 15 ion exchange resin was considered for the synthesis and hydrolysis of methyl acetate using the experimentally verified fulfill the basic criteria for regeneration of the solid adsorbent and the eluent for recycling |
|
Experiment C: Hydrolysis of a Carboxylic Acid Ester:
Hydrolysis of a Carboxylic Acid Ester: Neutral and Base Enhanced Reaction of neutral and base enhanced Acid catalyzed rate = kA [H+] [ester] methyl dichloroacetate 2,4-dinitrophenyl acetate phenyl acetate ethyl acetate Figure 1: |