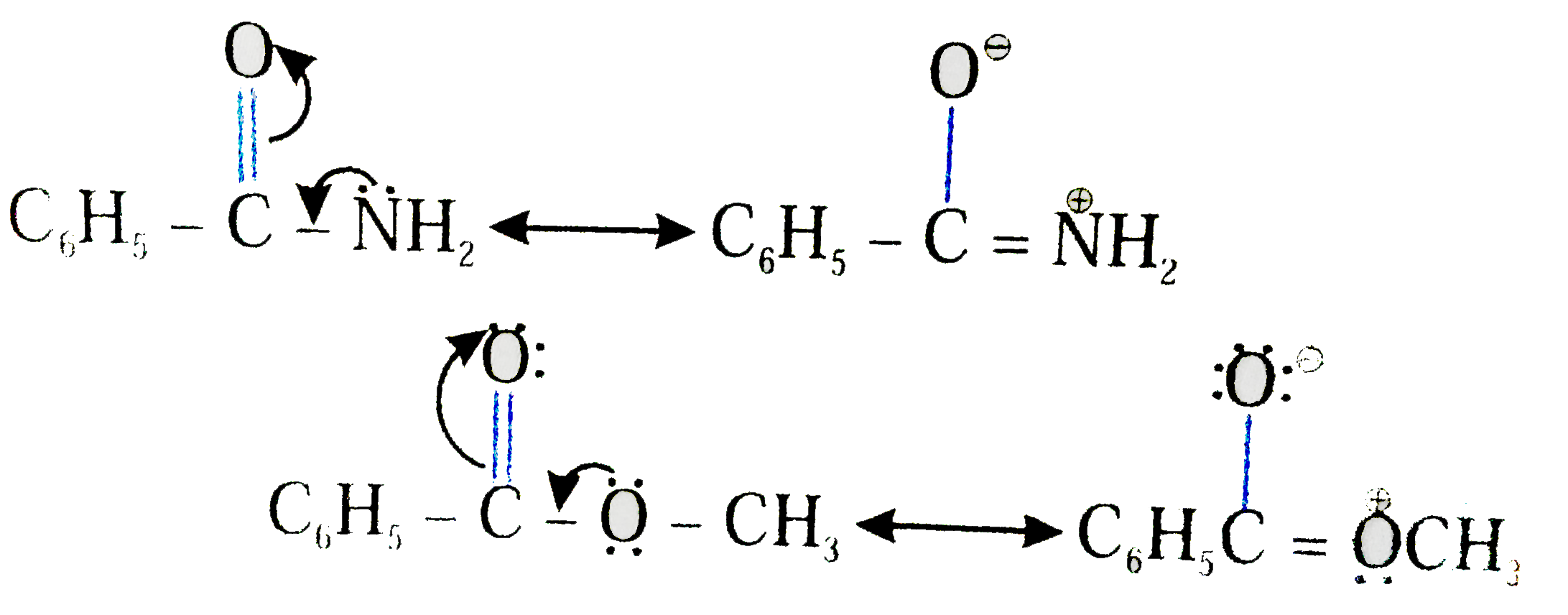basic hydrolysis of benzamide
Can ethanamide be heated with hydrochloric acid?
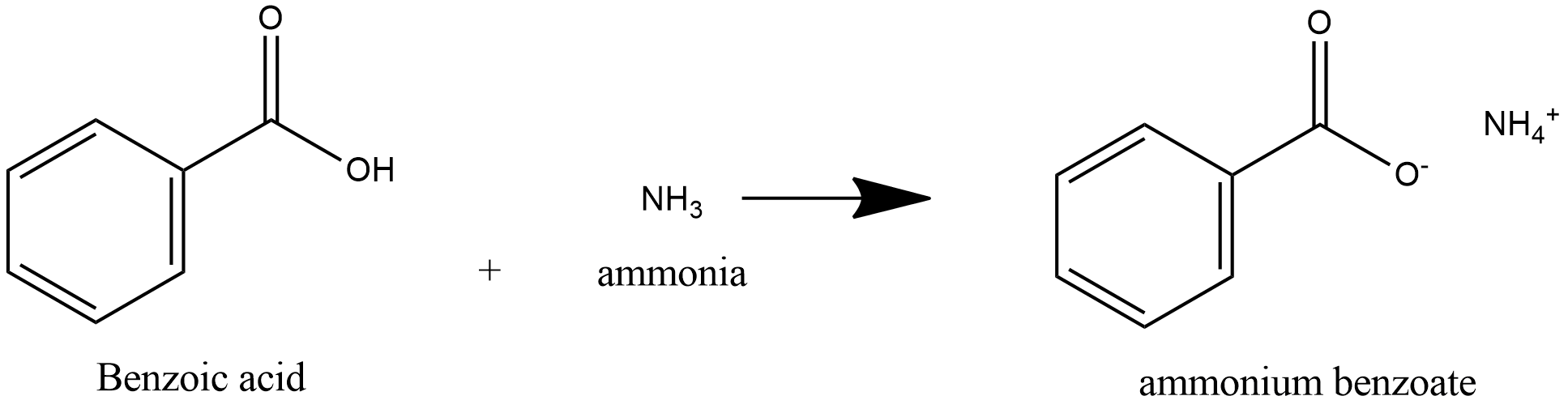
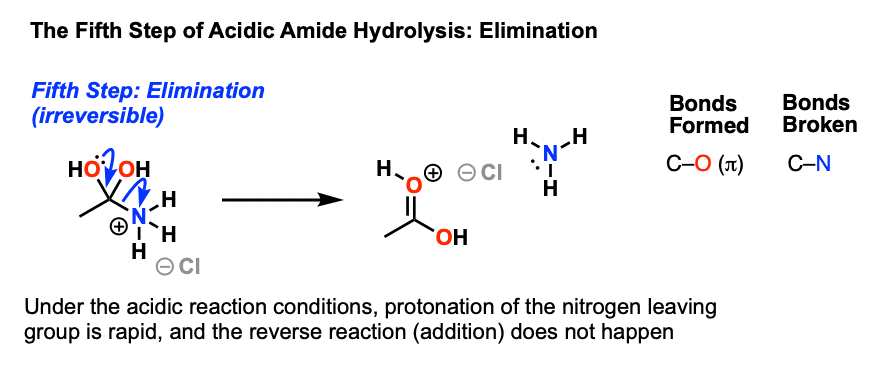
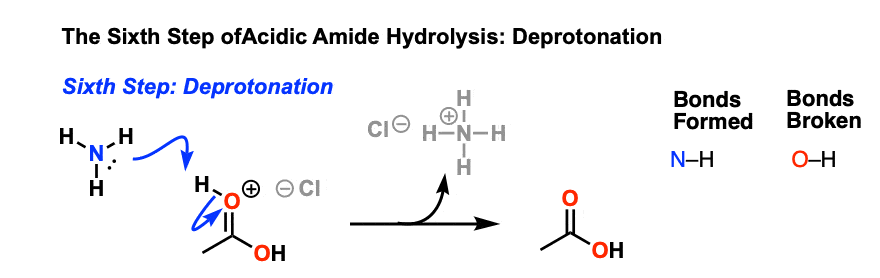
Taking ethanamide as a typical amide. If ethanamide is heated with a dilute acid (such as dilute hydrochloric acid), ethanoic acid is formed together with ammonium ions. So, if you were using hydrochloric acid, the final solution would contain ammonium chloride and ethanoic acid.
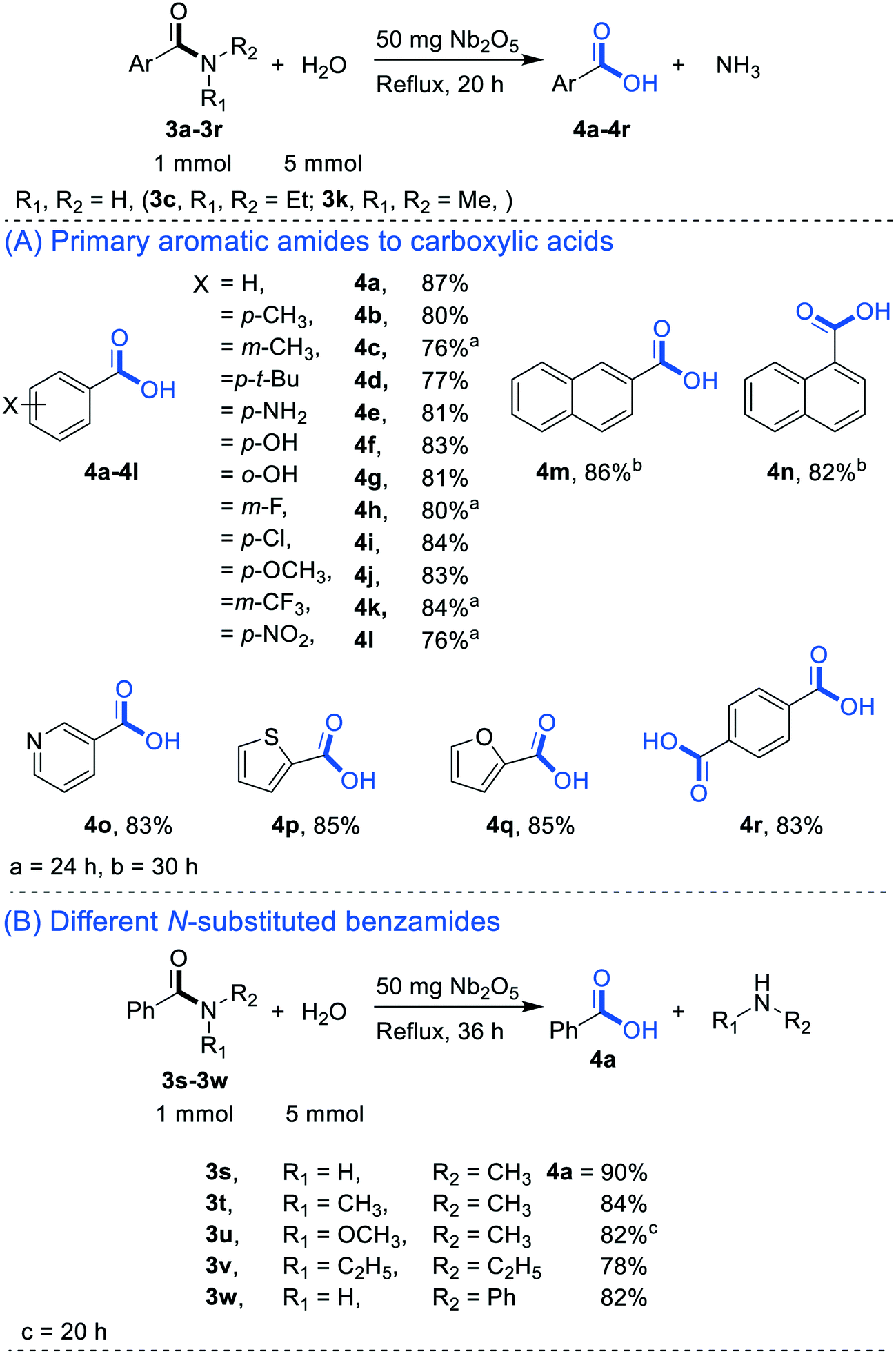
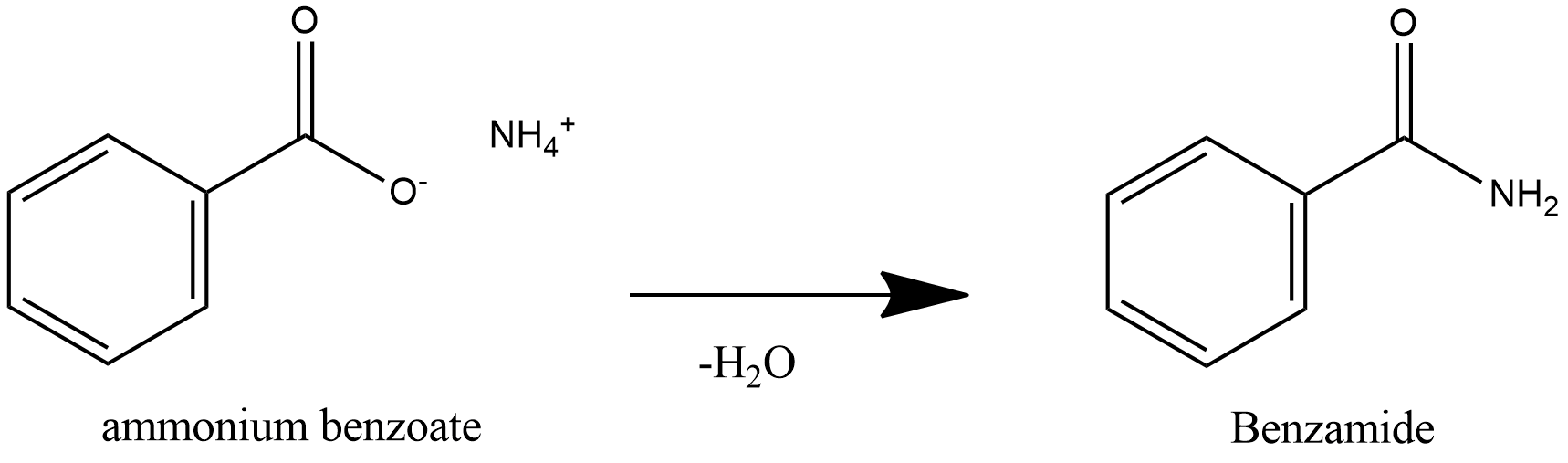
How do you convert benzamide to benzoic acid?
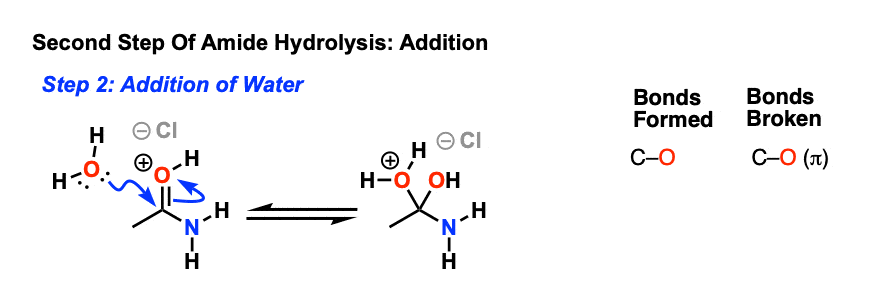
Let's say our goal was to go from benzamide to benzoic acid. We just learned two ways to do that. One way to do that would be to add water and an acid. We get H3O plus and if we heat things up we know that we can hydrolyze our amide that way and give us our benzoic acid, so that's one possibility. That's acid-catalyzed amide hydrolysis.
|
Intermediates in the Reactions of Carboxylic Acid Derivatives. IV
between benzamide-O18 and water during the basic hydrolysis of benzamide but no exchange was ob- served during its acidic hydrolysis. |
|
Alkaline hydrolysis of benzamide and N-methyl- and NN
hydrolysis of benzamide kBlo/kD |
|
Activation Energies of the Hydrolysis of Esters and Amides Involving
alkaline hydrolysis and carbonyl oxygen exchange of benzamide and ethyl benzoate has been determined. In eachcase an increase in temperature leads to a |
|
Stereo-electronic Effects in the Alkaline Hydrolysis of Benzamides
(Pseudo)-first-order rate constants for the alkaline hydrolysis of ¿-substituted benzonitriles and benzamides are reported. |
|
OXYGEN EXCHANGE DURING THE ACIDIC AND BASIC
ENZYMATIC HYDROLYSIS OF ESTERS1. Sir: Oxygen exchange has been found to occur be- tween benzamide-O28and water during the basic hydrolysis of benzamide |
|
Substituent Effects in the Acid and Base Hydrolyses of Aromatic
In the present paper are reported kinetic data for the acid and base hydrolysis in a 60-40 (by vol- ume) alcohol-water mixture |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VIII
and benzamide in both dilute and concentrated perchloric acid has been measured The acid-catalyzed hydrolysis of amides is in inally ways similar to the ... |
|
Polar Effects on Rates and Equilibria. V. Relationships between
the rates of acid and of basic hydrolysis of ethyl benzoates and between therates of acid andbasic hydrolysis of benzamides. |
|
The Concurrent Alkaline Hydrolysis and Isotopic Oxygen Exchange
by any carboxylic acid derivative.4·5 In fact in the hydrolysis of benzamide in 662/3% dioxane- water the rate constant for exchange is 17 times. |
|
The mechanism of acid hydrolysis of benzamide, N
D J Cram, Fundamentals of Carbanion Chemistry, Academic Press (1965) THE MECHANISM OF ACID HYDROLYSIS OF BENZAMIDE, N-METHYLBENZAMIDE , |
|
Comparative Stability of Benzamide, Salicylamide, and Some N
Meloche and Laidler (2) made a systematic study on the effect of ring substituents in the acid and base hydrolysis of aromatic amides These authors noted that in |