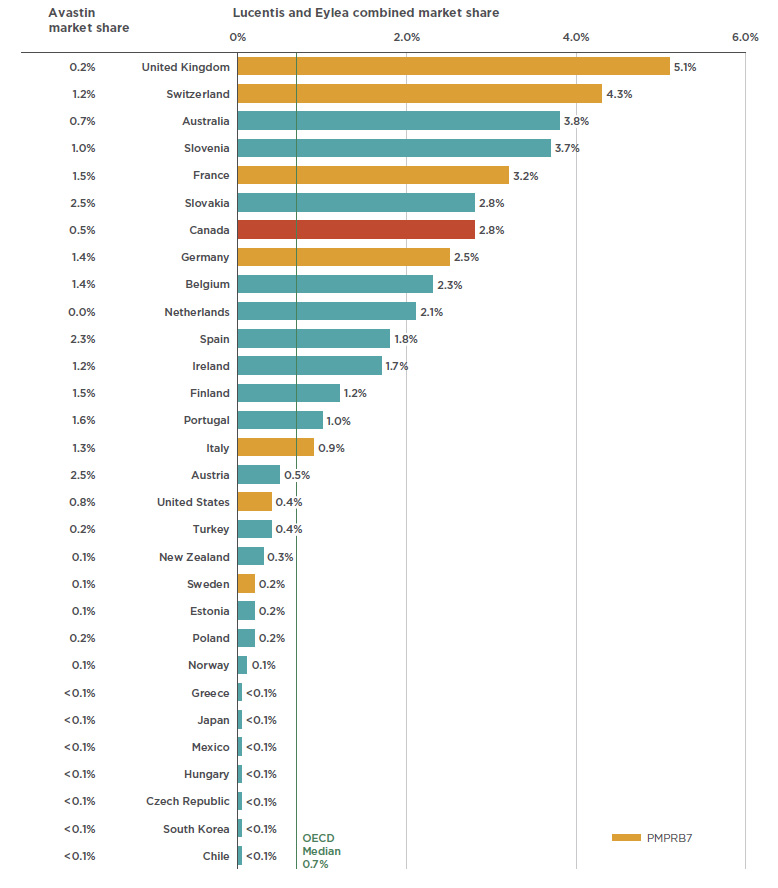
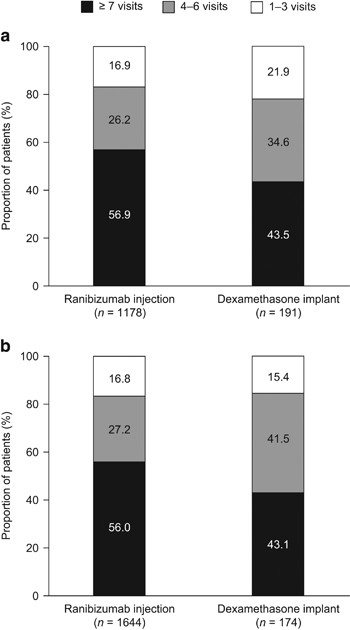
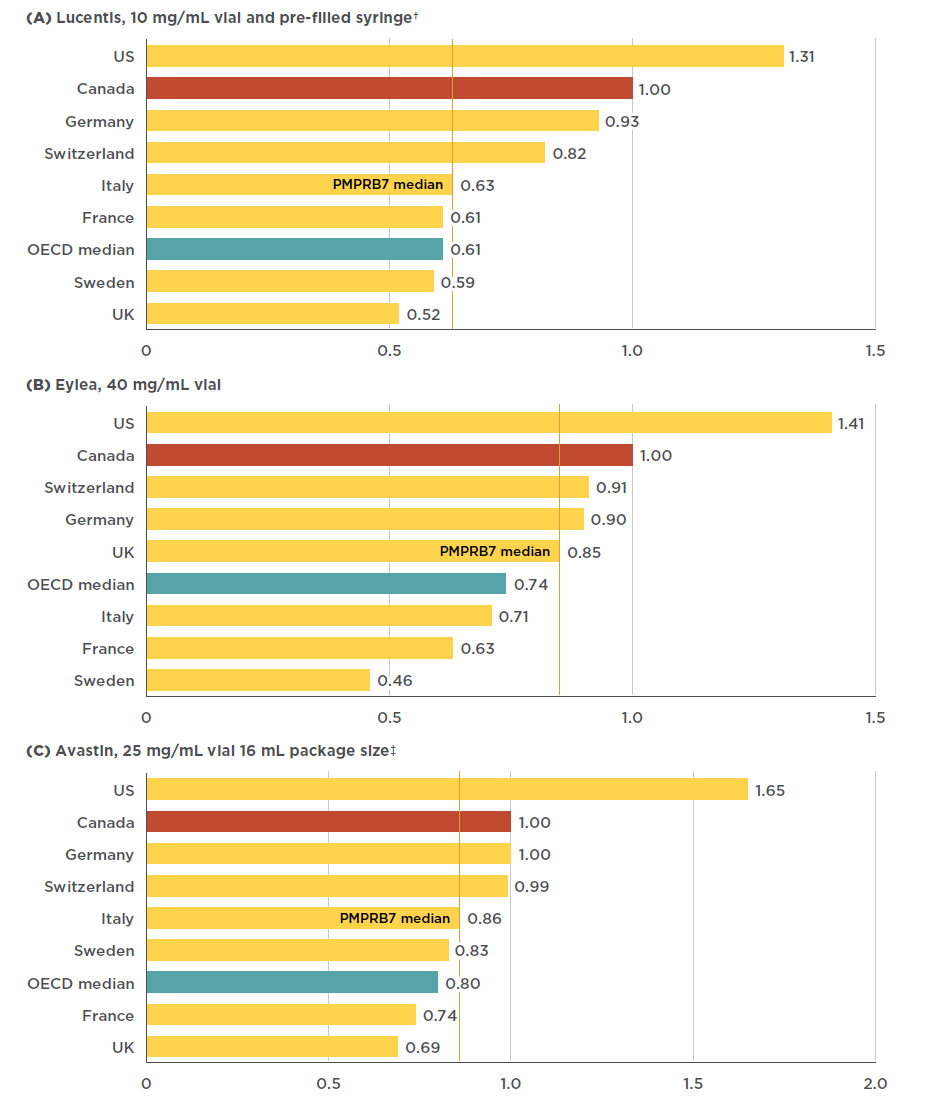
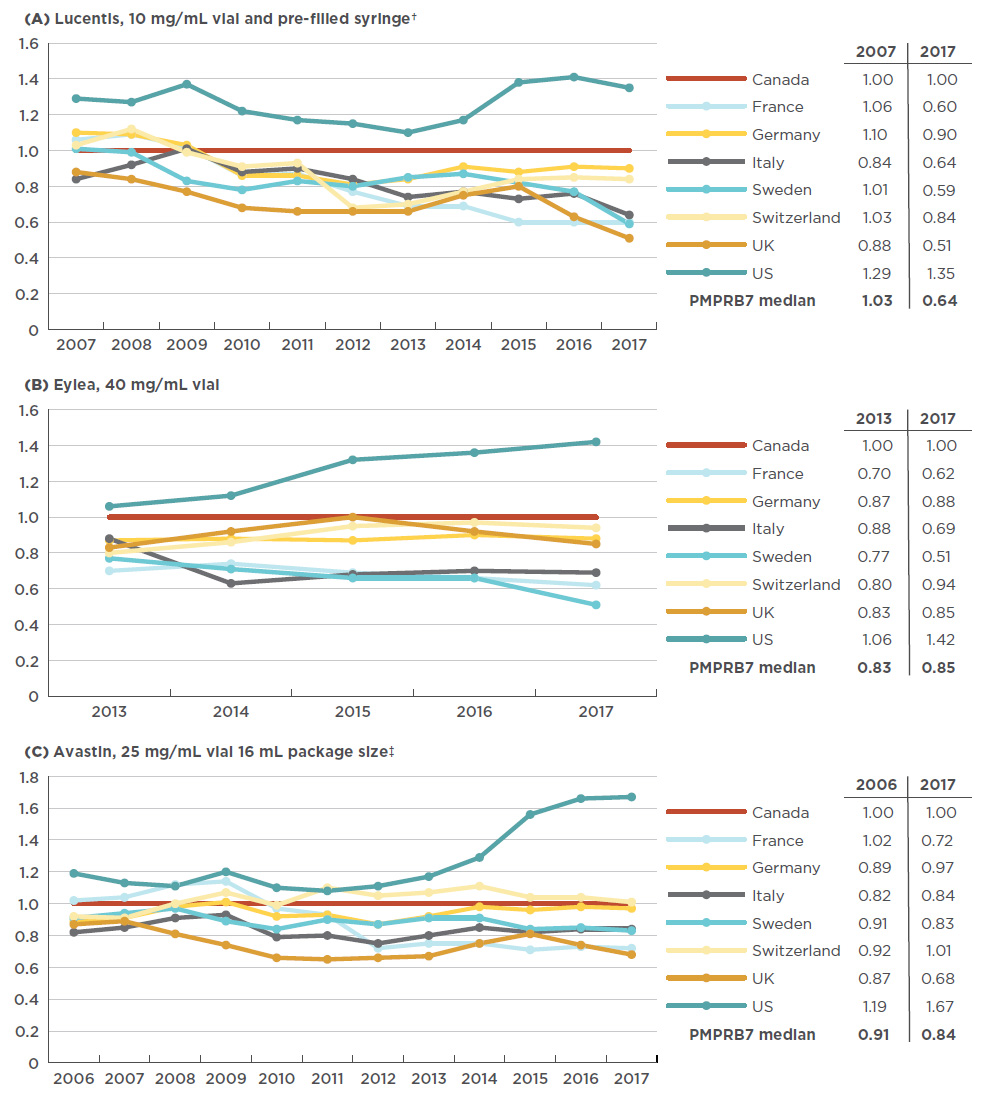
Lucentis, INN - ranibizumab - European Medicines Agency - Europa
|
Lucentis, INN-ranibizumab - European Medicines Agency - europa
EMEA/H/C/000715 Lucentis (ranibizumab) An overview of Lucentis and why it is authorised in the EU What is Lucentis and what is it used for? Lucentis is a |
|
Lucentis, INN-ranibizumab - European Medicines Agency - europa
Adults The recommended dose for Lucentis in adults is 0 5 mg given as a single intravitreal injection This corresponds to an injection volume of 0 05 ml The |
|
Lucentis, INN-ranibizumab
Read all of this leaflet carefully before you are given this medicine because it contains important information for you Detailed information on this medicine is available on the European Medicines Agency website: http://www ema europa eu |
|
Lucentis - WHO World Health Organization
28 nov 2014 · 4 International Nonproprietary Name (INN, generic name) of the medicine 4 injection to the WHO's list of essential eye medicines 2 Information as provided in the approved ranibizumab (Lucentis®) EU SmPC, dated Sep http://www ema europa eu/docs/en_GB/document_library/EPAR_- |
|
Biosimilar medicines - Medicines For Europe
Medicines for Europe and represents the leading companies developing Although the European Medicines Agency The INN is also approved by the EMA during Ranibizumab Lucentis® Antivascular endothelial growth factor ( VEGF) |
|
The Panel - ISPOR
medicine Product INN Number of biosimilar versions licensed in Europe European Medicines Agency, Biosimilar Medicines (ranibizumab) Lucentis |
|
Extensions of indication in the European Union – a - DGRA
EMA European Medicines Agency EPAR European public assessment report EPAR research be considered for any particular EU member state (13) Compared to the product name of the existing MA, the INN of the MP “will be the same for 27/05/2015 tolvaptan See MA date X Lucentis 22/01/2007 ranibizumab |
|
INFORMATION - Parallels Plesk Panel
Ranibizumab and stroke 14 The harmonization of regulatory requirements between Europe, Japan and the USA ment guideline is expected to be pub- ( Lucentis®) has advised healthcare registered by the National Drug Authority |






![Full text] Cost-effectiveness of ranibizumab versus aflibercept in Full text] Cost-effectiveness of ranibizumab versus aflibercept in](https://imgv2-1-f.scribdassets.com/img/document/361560226/original/b2e03edb22/1610081596?v\u003d1)








![[Full text] Cost-effectiveness of ranibizumab versus aflibercept in the [Full text] Cost-effectiveness of ranibizumab versus aflibercept in the](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/0a49b140-72f0-49a1-8103-f04e2f57910b/gr1.jpg)