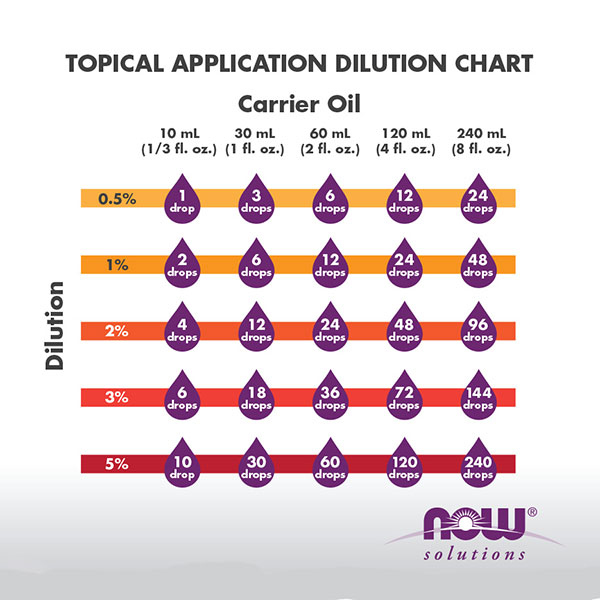
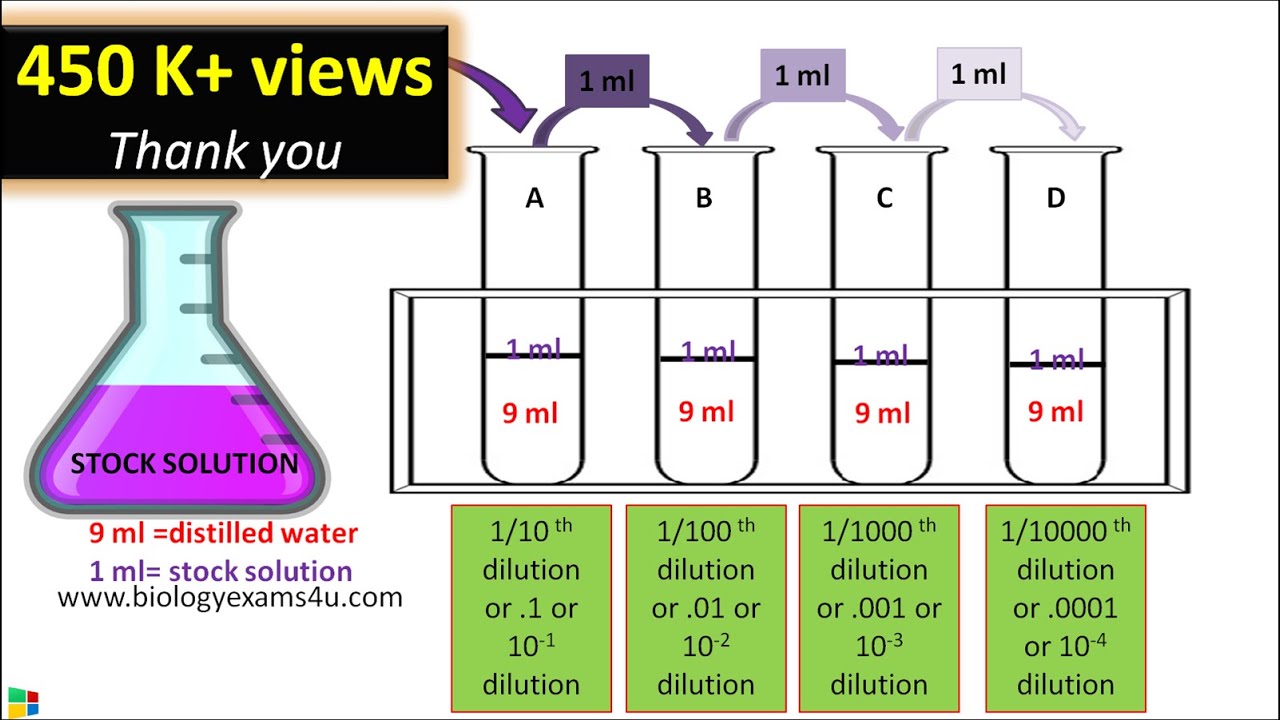
1 5 dilution calculation
What is a 1 4 dilution ratio?
For example, a 1:4 dilution ratio indicates 1 unit of solute and 4 units of solvent. Meanwhile, the dilution factor gives us the concentration of solute in the total volume of the solution (S:T). For instance, a 1:4 dilution factor indicates 1 unit of solute in 3 units of solvent; this totals 4 units of solution.
What is the difference between dilution ratio and dilution factor?
Both terms are quite different but are often used interchangeably. Where the dilution ratio is the simple dilution, the dilution factor is the ratio of concentrated solute at the start to the final dilution volume of the complete solution. What Does 1% Solution Mean?
How do you calculate diluent volume?
Multiply the final desired volume by the dilution factor to determine the needed volume of the stock solution. In our example, 30 mL x 1 ÷ 20 = 1.5 mL of stock solution. Subtract this figure from the final desired volume to calculate the volume of diluent required--for example, 30 mL - 1.5 mL = 28.5 mL.
What is a solution dilution calculator?
The solution dilution calculator tool calculates the volume of stock concentrate to add to achieve a specified volume and concentration.

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations

Dilution Problems

Calculating Dilution Factor
|
Dilution Chart
1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000. 12.5 litre 5 litre 2.5 litre 1250ml 1 litre |
|
Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution In other words another 1/5 dilution. D i l u t i o n 1 ... |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
For example a. 1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 unit volume of diluent (the material to be diluted) + 4 unit volumes of the |
|
Dr. Gs DILUTION PRIMER
Fortunately it is very simple to do if you follow the steps given below. 1. SIMPLE DILUTIONS: Let's look at an example of a 1:5 dilution. A 1: |
|
1:5 1:10 1:20 1:20 1:5 1:10
Diluent Volume Calculations: 1:5. 1:10. 1:20. Example: You have 7.5 mL of sample and want to prepare 1:10 dilution. x. –. = Weight of Sample. Weight of Sample. |
|
2 Amount and concentration: making and diluting solutions
Example 1: Converting g/L to M. You have weighed out 20 g KCl (formula weight of KCl is 1:5 dilution = 1 unit volume of diluent + 4 unit volumes of solvent;. |
|
1. dilutions and concentrations. liquid and solid samples
1.2. How would you prepare 10 ml of a 10-5 dilution in 3 steps? What if the sample is not liquid for example |
|
CHEMICAL HAZARD ASSESSMENT AND RISK MANAGEMENT
1.2 10-5 (1:81000). 2.2 10-5 (1:45 |
|
Pharmaceutical calculation
16 июл. 2021 г. If 50 mL of a 1:20 w/v solution are diluted to 1000 mL what is the ratio strength (w/v)?. 1:20 = 5%. 1000 (mL) = 5 %. 50 (mL) x %. X= 0.25 % = ... |
|
How to Make Simple Solutions and Dilutions 1. Simple Dilution
For example a 1:5 dilution (verbalize as "1 to 5" dilution) entails combining. 1 unit volume of solute (the material to be diluted) + 4 unit volumes of the |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
material to be diluted) + 4 unit volumes of the solvent medium (hence 1 + 4 = 5 = dilution factor). Example 1: To dilute a streptavidin solution 1:300. |
|
Dilutions Help
1 mL or 1 volume. 4 mL or. 4 volumes mix->. We have just made a dilution. The total volume is 5 mL ( or 5 total volumes). To calculate the. |
|
DILUTION CHART.pdf
MIXTURE. Dilution Chart. 1:1. 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000. 500ml 200ml 100ml 50ml. |
|
Dr. Gs DILUTION PRIMER
Fortunately it is very simple to do if you follow the steps given below. 1. SIMPLE DILUTIONS: Let's look at an example of a 1:5 dilution. A 1: |
|
1. dilutions and concentrations. liquid and solid samples
of this 1:10 dilution 1 ml corresponds to the sample. Expressing by an equation: How would you prepare 10 ml of a 10-5 dilution in 3 steps? |
| Untitled |
|
2 Amount and concentration: making and diluting solutions
calculate how much insulin would need to be dissolved in a certain volume of saline 1:5 dilution = 1 unit volume of diluent + 4 unit volumes of solvent;. |
|
Laboratory Math II: Solutions and Dilutions
weight out. ? Example: How much glucose would you need to make 50ml of a 1 uM solution (MW = 180g/ |
|
1:5 1:10 1:20 1:20 1:5 1:10
3M™ Petrifilm™ Plates. Diluent Volume Calculations: 1:5. 1:10. 1:20. Example: You have 7.5 mL of sample and want to prepare 1:10 dilution. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
5. Dilute the serum 10 times with saline. 6. Make a dilution of 1 part serum and 9 parts saline. Example: A 5M solution of HCl is diluted 1/5. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
5 Dilute the serum 10 times with saline 6 Make a dilution of 1 part serum and 9 parts saline 7 Make a dilution of Example: A 5M solution of HCl is diluted 1/5 |
|
Dilution and Concentration
has no drug substance in it; examples are sterile water and petrolatum Pharmaceutical Calculations for the Pharmacy Technician 208 4 5 6 X 1 2 3 – 0 |
|
261_Labs_Making Solutions and Dilutions - Penguin Prof Pages
For example, a 1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 unit volume of solute (the material to be diluted) + 4 unit volumes of the solvent |
|
Dr Gs DILUTION PRIMER
SIMPLE DILUTIONS: Let's look at an example of a 1:5 dilution A 1:5 dilution really means - one part, in a total of 5 parts If 1ml is added to 4mls, you are creating |
|
How to Make Simple Solutions and Dilutions
('ollcgr How to Make Simple Solutions and Dilutions i,r i,_:j, _:i'-: 1 , , :1 ,, L (r'erbalizc as "l lo 5' dilution) entails combining I unil volume of diluent (the in this example uses a I ml tolal volumc lhe initial step combines I unit volume of |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
Slide 1 Laboratory Math II: Solutions and Dilutions Philip Ryan, PhD For example, to make a five liter solution of 50 millimolar NaCl and ten millimolar tris- HCl |
|
DILUTION CHART NEW - Caprichem
DILUTION CHART 1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1: 1000 CHEMICAL 500ml 200ml 100ml 50ml 40ml 20ml 10ml 7ml 5ml 4ml 2ml |
|
Concentrations and Dilutions INTRODUCTION
So, the final weight/volume concentration is 5 EXAMPLE 6 11 Melanex® solution contains 0 9 g of hydroquinone in every 1 oz bottle What is the final percent |