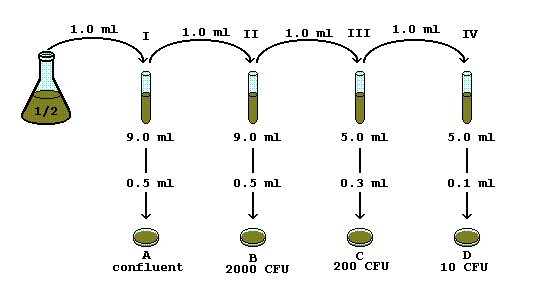
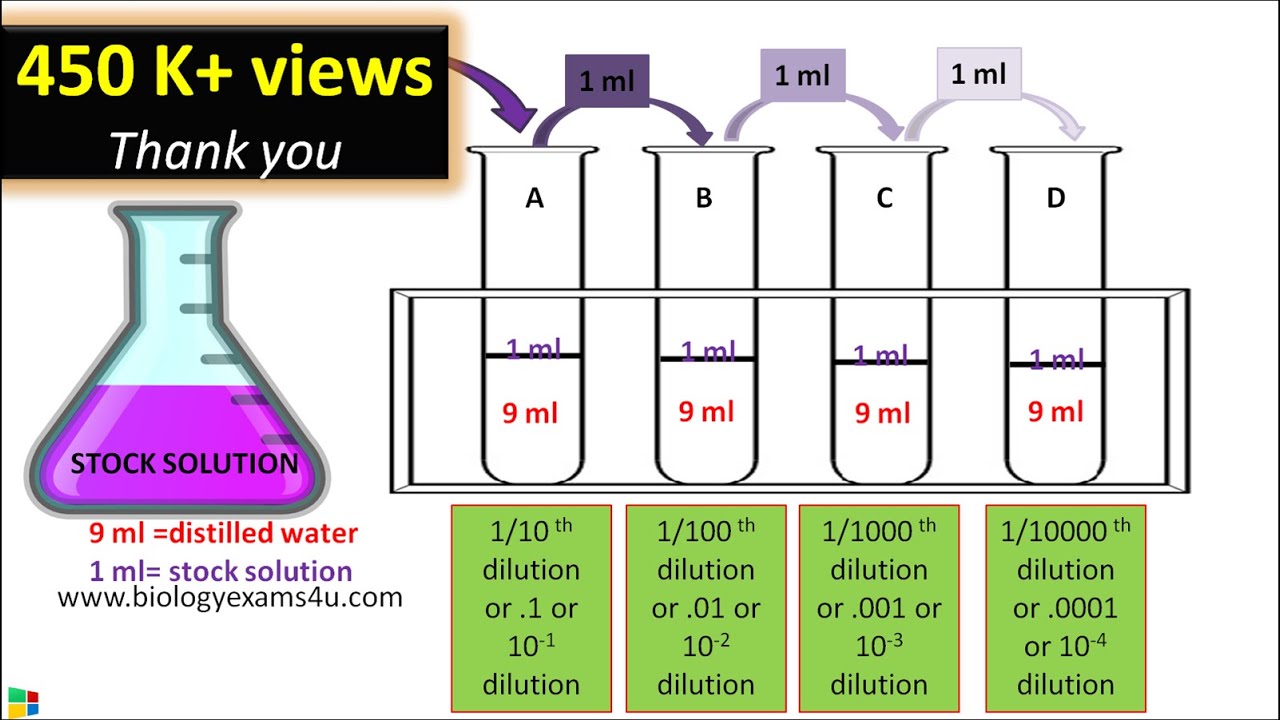
1 in 10 dilution concentration
What does 1 to 10 dilution mean?
A 1 to 10 dilution means that one part of the original solution is diluted with nine parts of the solvent (usually water or another diluent). This is often written as "1:10" or "1/10." To calculate this dilution, you mix one part of the solute with nine parts of the diluent. What is dilution ratio 1 to 10?
How do you make a 1:10 dilution from a 1M NaCl solution?
To make a 1:10 dilution from a 1M NaCl solution, mix one volume of the 1M NaCl solution with nine volumes of a suitable diluent, such as water. What does "1 of 10" mean? "1 of 10" refers to a ratio where one part of a substance is combined with ten parts of another substance, often a diluent.
What is a 1:10 dilution?
A 1:10 dilution is less diluted than a 1:100 dilution. In a 1:10 dilution, one part of the original solution is mixed with nine parts of the diluent, while in a 1:100 dilution, one part of the original solution is mixed with ninety-nine parts of the diluent. How do you make a 1:10 dilution from a 1M NaCl solution?
How do you identify a dilution solution?
You can identify a dilution solution by the amount of solute in the total volume, expressed as a proportion. For example, a chemical may be prepared in a 1:10 dilution of alcohol, indicating that a 10 mL bottle contains one milliliter of chemical and nine milliliters of alcohol.

Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations

What Are Dilutions Chemical Calculations Chemistry FuseSchool

Serial dilution
|
Microbiologics Dilutions Guide
A log dilution is a tenfold dilution meaning the concentration is decreased by a multiple of ten. To complete a tenfold dilution |
|
1. dilutions and concentrations. liquid and solid samples
100 ml of a water sample were filtered and then the filter was suspended in 10 ml of saline solution and shaked vigorously. What is the concentration factor? |
|
Laboratory Math II: Solutions and Dilutions
This will again be a one to ten dilution giving a concentration that is one one hundredth of the stock solution. This step is repeated four more times |
|
Solvants solutés et règles de dilution
Quelle est la concentration de ma solution ? Quel volume dois-je prélever pour préparer ma seringue ? C1 = 05 g x 1 mL / 10 mL. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
FICHE DE BON USAGE
Seringue de 10 ml + aig bleue. Morphine. Dilution : 1 ml ( soit 10 mg) + 9 ml de sérum Physiologique. Concentration : 1 mg /ml. Kétamine 50 mg/5 ml. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
milligram = mg = 1/1000 of a g or 10-3 g Making fixed volumes of specific concentrations from liquid reagents: V1C1=V2C2. |
|
Disinfection with Bleach
1 juin 2011 Understanding the concentration of your bleach solution is ... water for a 1:10 dilution (~6000 ppm) or 1/4 cup of bleach in. |
|
Labo de Microbiologie
1 août 2018 EX. Une dilution de 1/10 représente un facteur de dilution de 10X. • Le facteur de dilution permet de déterminer la concentration originale. |
|
GUIDE DADMINISTRATION DES MÉDICAMENTS INJECTABLES
concentration indiquée est valable pour l'administration par VVP. Etape 6. Particularités Dilution avec NaCl 0.9% 1:10 possible. A utiliser de suite. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
Another way is to dilute the stock 1/10 twice and then perform a further 1/100 dilution: 1/10 x 1/10 x 1/100 = 1/10,000 dilution This would yield 100 ml of a 1/10,000 dilution of stock in water |
|
Dilution and Concentration
So if 50 mL of a solution con- taining 10 g of active ingredient with a strength of 20 or 1 : 5 w/v is diluted to 100 mL, Page 3 the original volume is doubled, but |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
milligram = mg = 1/1000 of a g or 10-3 g gram = g 1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 unit volume of diluent (the material to be Making fixed volumes of specific concentrations from liquid reagents: V1C1= V2C2 |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
Slide 1 Laboratory Math II: Solutions and Dilutions Philip Ryan, PhD The concentration of a solution is how much of the solute is present per unit of This means that you add 10 milliliters of the 5M NaCl stock solution to a container and |
|
Dilution (and Concentration) Theory
The second “rule” of dilutions/concentrations is an obvious one: there is a limit to 10µl divided by (990 + 10) µl total volume = 10/1000 = 1/100 = 10-2 |
|
Microbiologics Dilutions Guide
For example, a sample size of 1 ml is added to 9 ml of diluent to equal a total of 10 ml Example: 1:10 dilution - if the concentration is 1,000 CFU, a one log |
|
2 Amount and concentration: making and diluting - Mathcentre
Simple Dilutions Make a 1:10 dilution (say this as “a one in ten dilution”) Take 1 part of your stock solution and add 9 parts of solvent (usually water but sometimes |
|
Concentrations and Dilutions Part One
To answer this question lets examine data table 2 below for the dilution of a 10 (m/v) solution table two Stock Solution Concentration (10 ) Volume of Stock |
|
Concentrations and Dilutions INTRODUCTION
tration as a percentage So, the final weight/volume percent strength is 10 EXAMPLE 6 9 9 g of sodium chloride is diluted in 1 L of SWFI (sterile water for |