1 in 10 dilution definition
What is a dilution factor in biology?
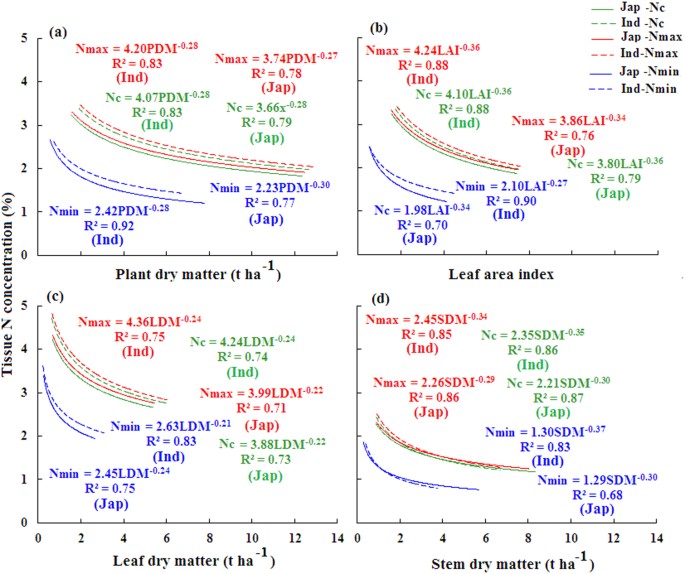
This way of expressing a dilution as a ratio of the parts of solute to the total number of parts is common in biology. The dilution factor (DF) can be used alone or as the denominator of the fraction, for example, a DF of 10 means a 1:10 dilution, or 1 part solute + 9 parts diluent, for a total of 10 parts.
|
Microbiologics Dilutions Guide
A log dilution is a tenfold dilution meaning the concentration is decreased by a multiple of ten. To complete a tenfold dilution |
|
Solvants solutés et règles de dilution
Quelle est la concentration de ma solution ? Quel volume dois-je prélever pour préparer ma seringue ? C1 = 05 g x 1 mL / 10 mL. |
|
Laboratory Math II: Solutions and Dilutions
Slide 1. Laboratory Math II: Solutions and Dilutions. Philip Ryan PhD This means that you add 10 milliliters of the 5M NaCl stock solution to a ... |
|
Serial Dilution Vortex mixer Spectrophotometer
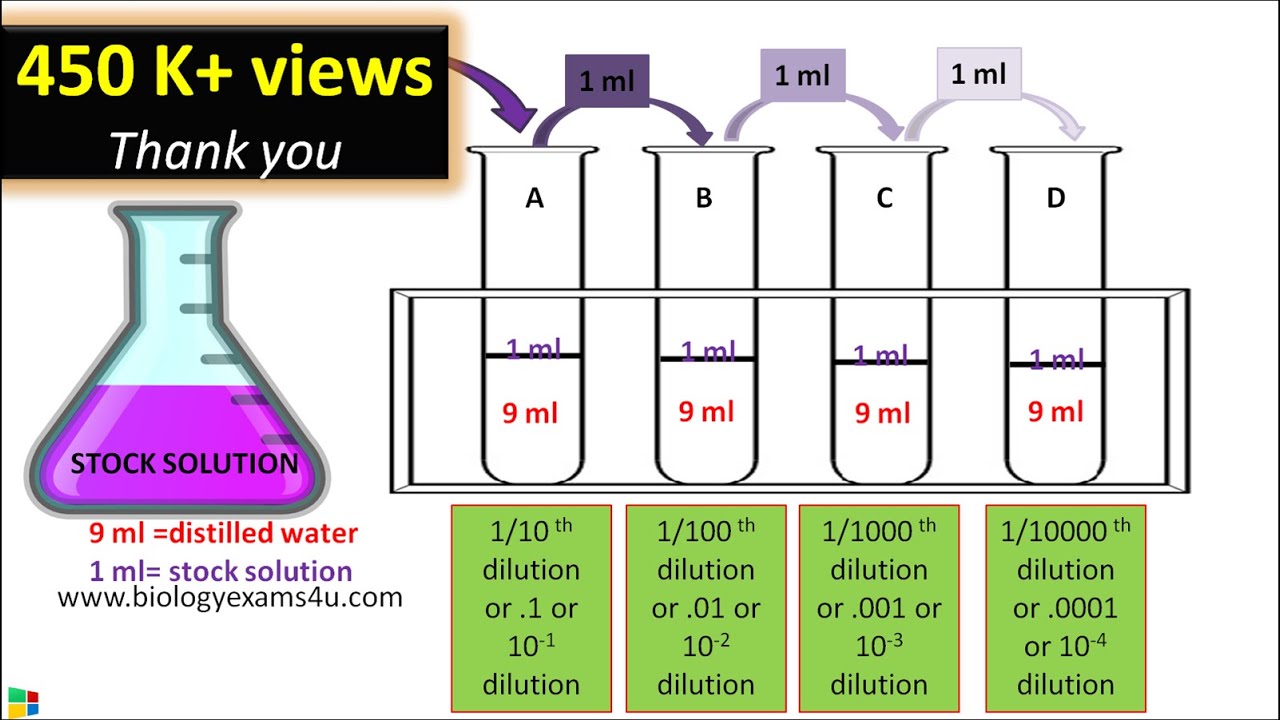
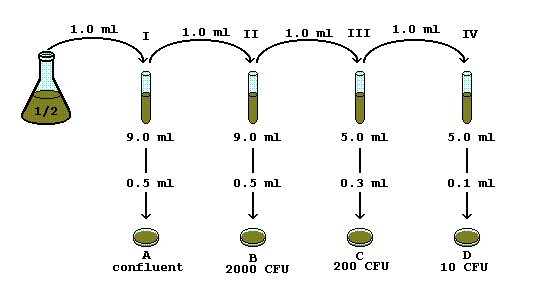
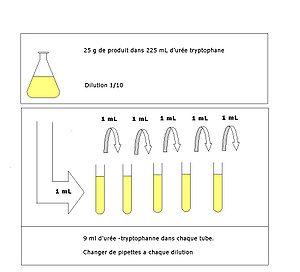
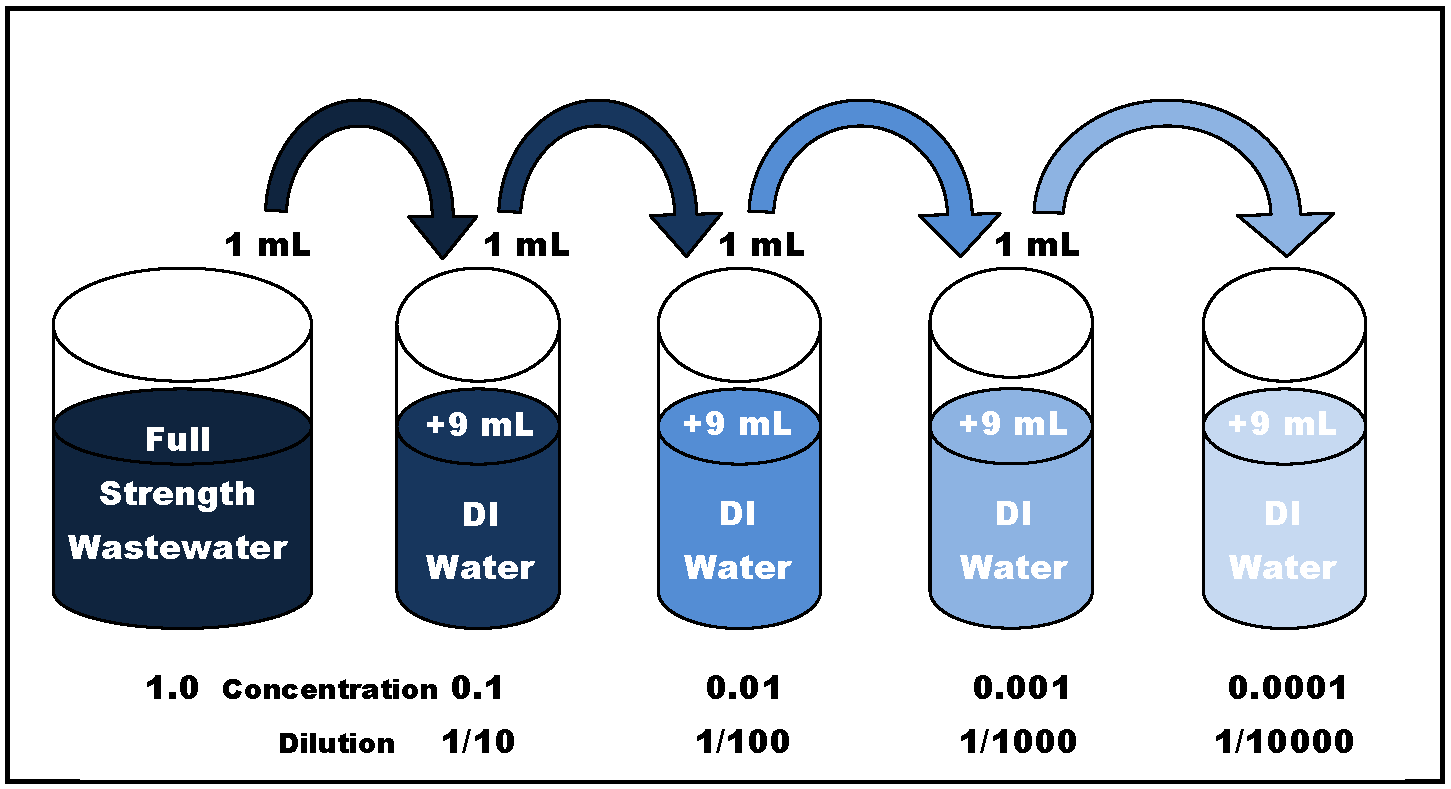
A serial dilution definition is: the repeated dilution of a solution from tube 1:10 and add it to the 9 mL of dilution liquid in the tube 1:100. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
means the volume of original substance in the total volume of final solution. In dilution statements Make a 1 to 10 dilution of serum with saline. |
|
Labo de Microbiologie
1 août 2018 10%. Examen final théorique. 40%. Les solutions. 5. Définitions. • Solution ... salin la dilution serait écrite 1/10 ou dite. |
|
1. dilutions and concentrations. liquid and solid samples
The simplest case is the preparation of 10 ml of the. 1:10 dilution of the sample. For this 1 ml of sample is added to 9 ml of diluent; consequently |
|
Preparation of samples and dilutions plating and sub-culture - GOV
(CFU per g) (ie single 0.5mL plate at 10-1 dilution) rather than <10 CFU/g tasks (or processes) are defined as “…one that if carried out incorrectly or. |
|
IEC 60079-10-1 (2020).pdf
Part 10-1: Classification of areas – Explosive gas atmospheres Assessment of ventilation and dilution and its influence on hazardous area .......... 53. |
|
61 MICROBIOLOGICAL EXAMINATION OF NONSTERILE
WaterSoluble Products— Dissolve or dilute (usually a 1 in 10 dilution is prepared) the Use either the total contents or a defined number of metered. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
means the volume of original substance in the total volume of final solution 1 Make a 1 to 10 dilution of serum in saline 2 Make a 1 in 10 dilution of serum in |
|
Dilutions Tutorial
It is very important to remember that “dilute 1:10” means adding one part of diluate to nine parts of diluent The term part can refer to µL, mL, L, etc as long |
|
Math and Dilutions
Microbiology is full of Math and Dilution Terminology Luckily, once you A 10-2 (“ten to the minus two”) dilution means a “one to one hundredth dilution”, so to |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
Unit Definitions milligram = mg = 1/1000 of a g or 10-3 g molar = M = term used to discuss the molarity of solutions (e g , 2 5 M means 2 5 moles of 1:5 dilution ( verbalize as "1 to 5" dilution) entails combining 1 unit volume of diluent (the |
|
Dilution and Concentration
Define the alligation methods of problem solving • Use the alligation taining 10 g of active ingredient with a strength of 20 or 1 : 5 w/v is diluted to 100 mL, |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
Slide 1 Laboratory Math II: Solutions and Dilutions Philip Ryan, PhD This means that you add 10 milliliters of the 5M NaCl stock solution to a container and |
|
Objectif Facteur de dilution Vocabulaire spécifique à la - archimede
C'est un nombre obligatoirement supérieur à 1 Exemple : pour une dilution au dixième ( f = 10 ) : fiole de 100 mL et pipette de 10 mL ; fiole de 200 |
|
Dilutions - Weber State University
Making dilutions is one of the most common practices in chemistry contains 2mL, then adding “one volume” of alcohol means that 4mL of alcohol should be Consider the case where 10mL of soda pop that is being prepared for analysis |