1 to 1 dilution ratio
What is a standard dilution?
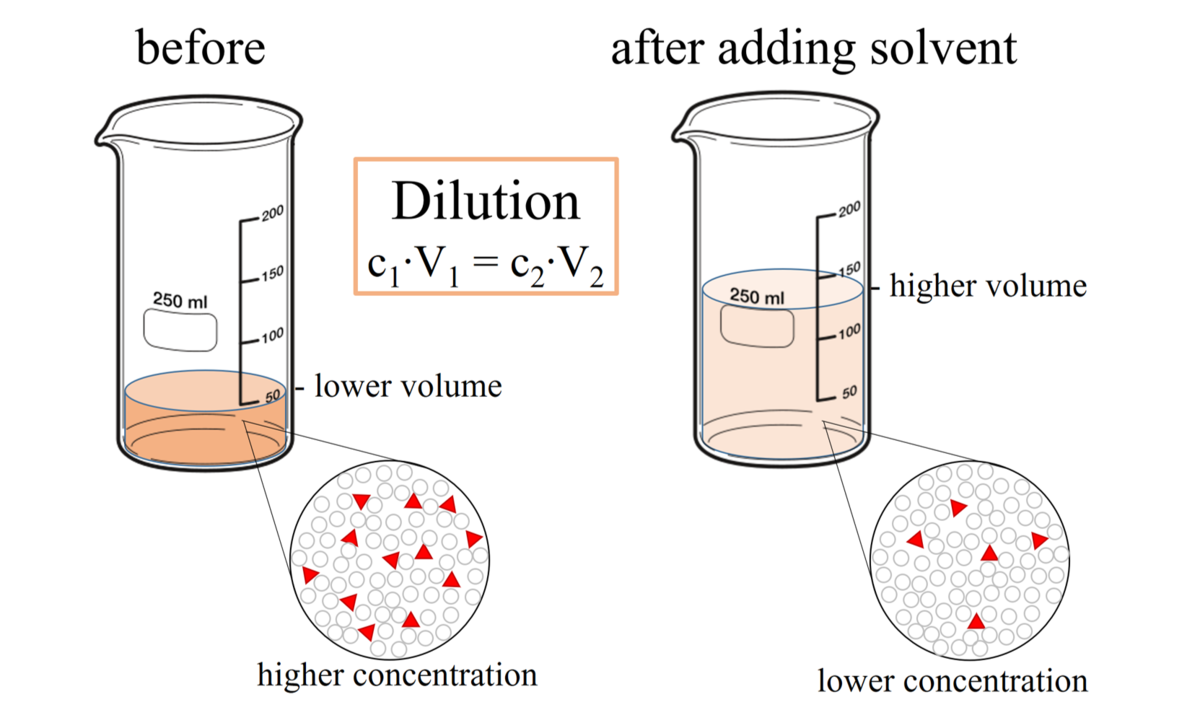
A standard dilution is a one-time event, mixing the reagent with diluent and using this solution in your analysis. We frequently dilute things times 2, which means 1 part reagent mixed with 1 part diluent. For example, I will mix 1 mL calcium reagent with 1 mL of deionized water to make a times 2 dilution.
What is a 1 5 dilution ratio?
For example, in a solution with a 1:5 dilution ratio, entails combining 1 unit volume of solute (the material to be diluted) with 5 unit volumes of the solvent to give 6 total units of total volume. In photographic development, dilutions are normally given in a '1+x' format.
What is a dilution factor?
Dilution factor is a notation often used in commercial assays. For example, in solution with a 1/5 dilution factor (which may be abbreviated as x5 dilution ), entails combining 1 unit volume of solute (the material to be diluted) with (approximately) 4 unit volumes of the solvent to give 5 units of total volume.
How do you make a liter of dilution with a solution ratio?
To make a liter of dilution with a solution ratio of 1:5, you'll need to mix 167 ml of stock solution in 833 ml of water. To get these amounts: Use the volume of solute formula and substitute the known quantities: solute_volume = final_volume / (solvent_ratio + solute_ratio)
Introduction
A particular laboratory calculation that confuses us when we’ve not done it for a while is dilution. Accurate dilutions are essential to accurate medical laboratory test results. If you dilute something wrong, you will not get the correct result. Incorrect results could harm patients if the physician treats the patients based on those incorrect res
Dilution vs. Ratio
A dilution is made by combining a certain volume of reagent or specimen with a certain volume of diluent. The same can be said of a ratio. No wonder it’s confusing The difference is a dilution is expressed as parts reagent or specimen to total parts of solution. A ratio is expressed as parts reagent or specimen to parts diluent. Let’s look at some
Examples of Standard Dilutions
A standard dilution is a one-time event, mixing the reagent with diluent and using this solution in your analysis. We frequently dilute things times 2, which means 1 part reagent mixed with 1 part diluent. For example, I will mix 1 mL calcium reagent with 1 mL of deionized water to make a times 2 dilution. This is written as a 1:2 dilution or a tim
Conclusion
Accurate dilutions are critical to accurate test results. This blog has hopefully explained the difference between dilution and ratio as it is used in medical laboratory science, improved the accuracy of your dilution calculations, and made it a less frustrating concept for you medlabstudyhall.com

Calculating Dilution Factor

How To Dilute Chemicals: Dilution Ratios Explained!

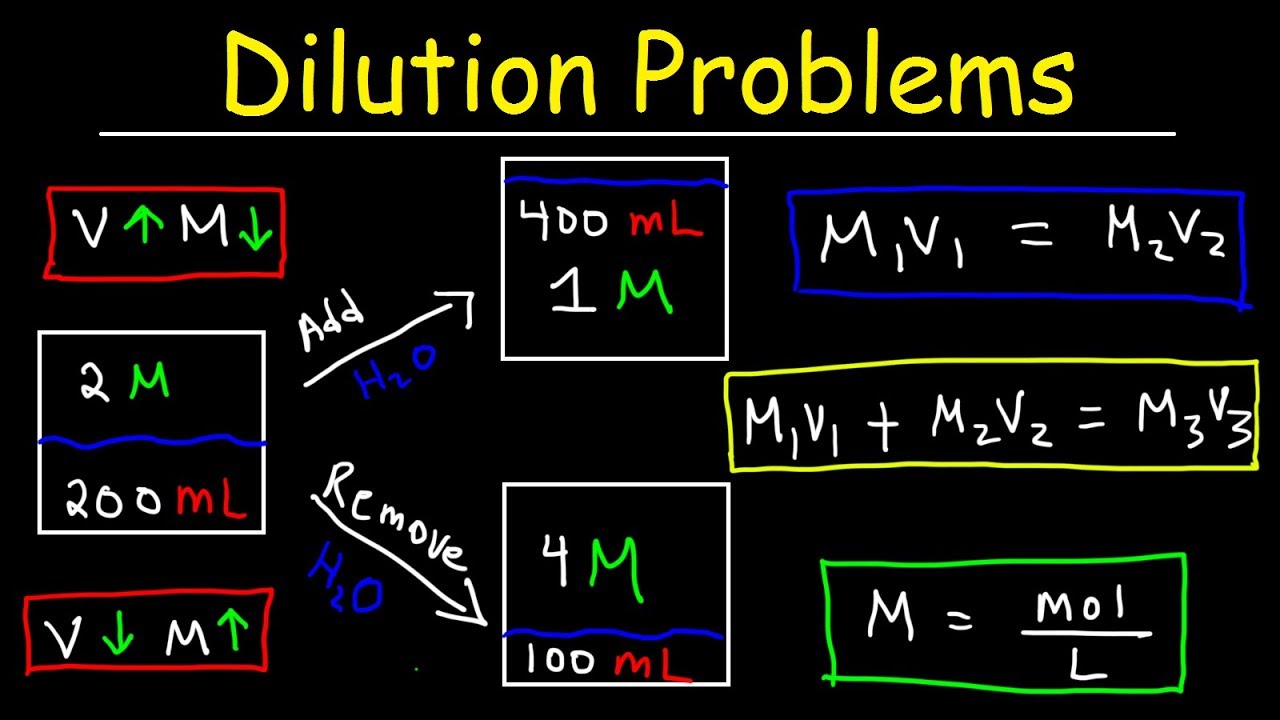
Dilution Problems Chemistry Molarity & Concentration Examples Formula & Equations
|
Dilution Chart
Dilution Chart. 1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000. 500ml |
|
Running Dilutions on the Catalyst One* Chemistry Analyzer
Load the diluted sample created in steps 1 and 2. 6. Load the slides. Note Parts Sample + Parts Diluent = Total Parts (Dilution Factor). Parts Sample. |
|
Dilution Ratio Chart
Dilution Ratio Chart. RATIO AMOUNT OF CONCENTRATE. WATER. 1:1. 128 ounces. To. 128 1:10 dilution selling at $7.30 per gallon- $7.30 10= .73 or 73 cents per ... |
|
Dilution Protocol
Select Manual and use the up/down arrows to specify the desired dilution factor (total parts). It is recommended to start with a 1:1 dilution unless directed |
|
Below are some titer dilutions depicting a fourfold increase and
infection. 1:1. NR. Titers can fluctuate after treatment by increasing twofold (one dilution) while still decreasing overall |
|
Dr. Gs DILUTION PRIMER
this is a 1 to 5 dilution or 1:5 dilution. This process can be used to determine how to perform any dilution. All you need to know is what the dilution factor |
|
Celebrates
Powder chemical amounts are indicated in brackets. For example a cleaner has 10L of water and the chemical has a 1 to 25 dilution ratio. By checking the |
|
Microbiologics Dilutions Guide
A log dilution is a tenfold dilution meaning the concentration is decreased by a multiple of ten. To complete a tenfold dilution |
|
2 Amount and concentration: making and diluting solutions
The concentration of a solution of 1 mole dissolved in 1 L is 1 M (“one molar”) Step 1: work out what the dilution factor is .. 1/0.32 = 3.125. Step 2: turn ... |
|
Dilution of solutions for nurses
ratio it could also be expressed as a percentage. For practical purposes weak or very dilute solutions ratios of 1:1000 1:2000 and 1:5000 maybe written as. 1. |
|
DILUTION CHART.pdf
500ml 200ml 100ml 50ml. |
|
BOTOX® RECONSTITUTION AND DILUTION PROCEDURES
Table 1: Dilution Instructions for BOTOX Vials (100 Units and 200 Units)** baseline value as covariate and stratification factor treatment group and ... |
|
Running Dilutions on the Catalyst One* Chemistry Analyzer
The Catalyst One* Chemistry Analyzer supports automated dilutions (the Parts Sample + Parts Diluent = Total Parts (Dilution Factor). Parts Sample. |
|
Celebrates
dilution prior to use and ready-to-use diluted product contains 1 litre of ... DILUTION. RATIO. Add. Add. Chemical. Water. Chemical. Water. 1 to 1. |
|
Dr. Gs DILUTION PRIMER
Diluting a sample by half is a 1:2 dilution. Can we be sure of this? Let's test it out. 200mls divided by 2 (the dilution factor) is 100mls |
|
DILUTION CHART
PIC's dilution ratios are written as parts. Therefore when PIC's label suggest a dilution ratio of 1-to-4 (1:4) that means 1 part product and 4 parts water |
|
Dilution of solutions for nurses
This leaflet explains how dilution calculations are performed. For practical purposes weak or very dilute solutions ratios of 1:1000 1:2000 and 1:5000 ... |
|
Dilution Ratio Chart
Dilution Ratio Chart. RATIO AMOUNT OF CONCENTRATE. WATER. 1:1. 128 ounces. To. 128 ounces. 2:1. 64 ounces. To. 128 ounces. 4:1. 32 ounces. |
|
USING THE JEROME 10:1 DILUTION MODULE (AZI P/N: 990-0223
The sample stream is diluted in a. 10:1 ratio within ±10% |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
Dilution Ratio Chart - Fulton Distributing
Dilution Ratio Chart RATIO AMOUNT OF CONCENTRATE WATER 1:1 128 ounces To 128 ounces 2:1 64 ounces To 128 ounces 4:1 32 ounces To |
|
DILUTION CHART NEW - Caprichem
1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000 CHEMICAL CHEMICAL 1 litre 400ml 200ml 100ml 80ml 40ml 20ml 13ml 10ml 8ml 4ml 2ml |
|
DILUTION CHART - Jangro
ISSUED WITHOUT PREJUDICE Dilution Ratio Chemical Measure Water Container 1:5 1:10 1:25 1 : 50 1 : 1 : 200 100 1 :400 1:1 1 part Product to 1 parts |
|
Dilution Control Conversion Chart - Triple S
Dilution Ratio Add this amount of concentrate 1:4 6 5 oz 1:4 25 5 oz 1:4 128 oz (1 Gal) 1:4 4 75 oz 1:10 3 oz 1:10 11 5 oz 1:10 59 oz 1:10 2 25 oz 1:12 |
|
Dilution Conversions - Dosatron
Ratio Ounce = 29 57 Milliliters 1 Gallon = 3 79 liters Dilution Conversions Percentage* Ratio* Ounces per US gallon* ml/l* 10 1:10 12 800 oz/gal 100 |
|
DILUTION CHART
PIC's dilution ratios are written as parts Therefore, when PIC's label suggest a dilution ratio of 1-to-4 (1:4) that means 1 part product and 4 parts water Some |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor, and so on until the final concentration is known Example: A 5M solution of HCl is diluted 1/5 The resulting solution is diluted 1/10 Determine |
|
Chemical Dilution Rates - Prochem
ratio of product to water of 1:4, for every 1 litre of concentrated product you would need to add 4 litres of water to make up a total of 5 litres of diluted liquid |
|
Diluting cleaning products using ratios - BBC
Diluting means measuring an amount of cleaning product and adding it to water Neat means not diluted Using ratios A ratio of 1:10 means add 1 part of product |

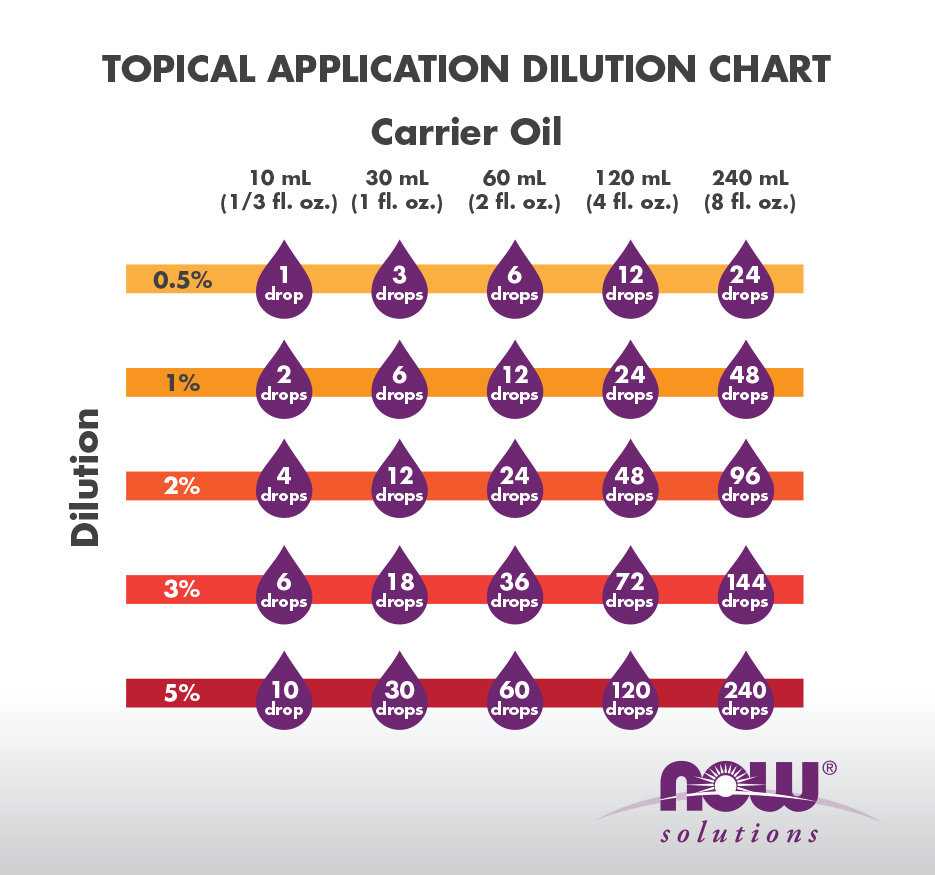


















![PDF] Development of an Alcohol Dilution-Lyophilization Method for PDF] Development of an Alcohol Dilution-Lyophilization Method for](https://www.nowfoods.com/sites/default/files/drop-to-tsp-ml-chart_0.png)


.jpg)