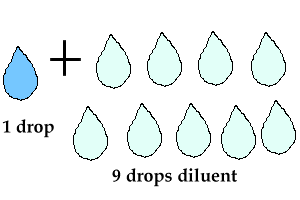1 to 10 dilution example
What is a 1:10 dilution?
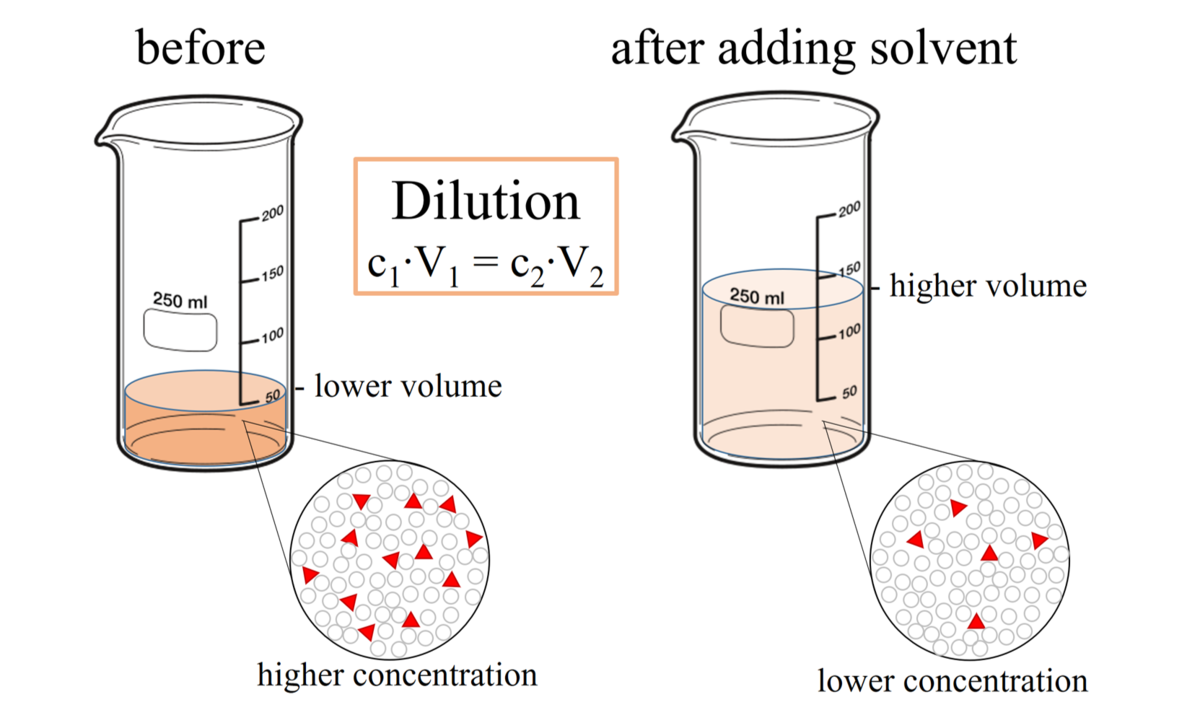
This type of dilutions describes the ratio of the solute to the final volume of the diluted solution. For example, to make a 1:10 dilution of a 1M NaCl solution, you would mix one "part" of the 1M solution with nine "parts" of solvent (probably water), for a total of ten "parts."
|
Microbiologics Dilutions Guide
sample. For example a sample size of 1 ml is added to 9 ml of diluent to equal a total of 10 ml. Example: 1:10 dilution - if the concentration is 1 |
|
1. dilutions and concentrations. liquid and solid samples
100 ml of a water sample were filtered and then the filter was suspended in 10 ml of saline solution and shaked vigorously. What is the concentration factor? |
|
Laboratory Math II: Solutions and Dilutions
weight out. ? Example: How much glucose would you need to make 50ml of a 1 uM solution (MW = 180g/ |
|
Dilutions Occasionally a solution is too concentrated to be used as it
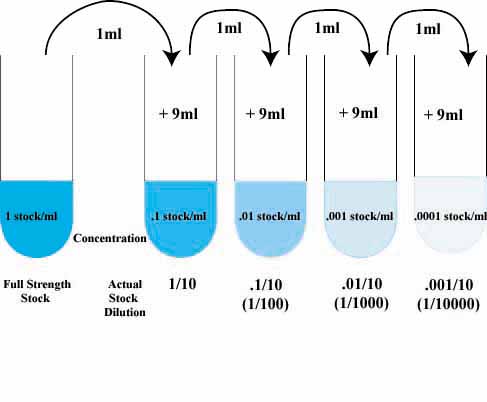
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
1:5 1:10 1:20 1:20 1:5 1:10
3M™ Petrifilm™ Plates. Diluent Volume Calculations: 1:5. 1:10. 1:20. Example: You have 7.5 mL of sample and want to prepare 1:10 dilution. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
examples are used with his permission. Unit Definitions milligram = mg = 1/1000 of a g or 10-3 g Example 1: To dilute a streptavidin solution 1:300. |
|
IDEXX VetTest Chemistry Analyzer Dilution Protocol: Urine Plasma
Dilution Chart. Volumes are for example only. Parts Sample + Parts DI Water = Total Parts. Parts Sample Parts DI Water. Total Parts (Dilution Factor). 1 (10 |
|
2 Amount and concentration: making and diluting solutions
calculate how much insulin would need to be dissolved in a certain volume of If your original sodium chloride solution is 1M and you dilute it 1:10 |
|
Problems Part 1
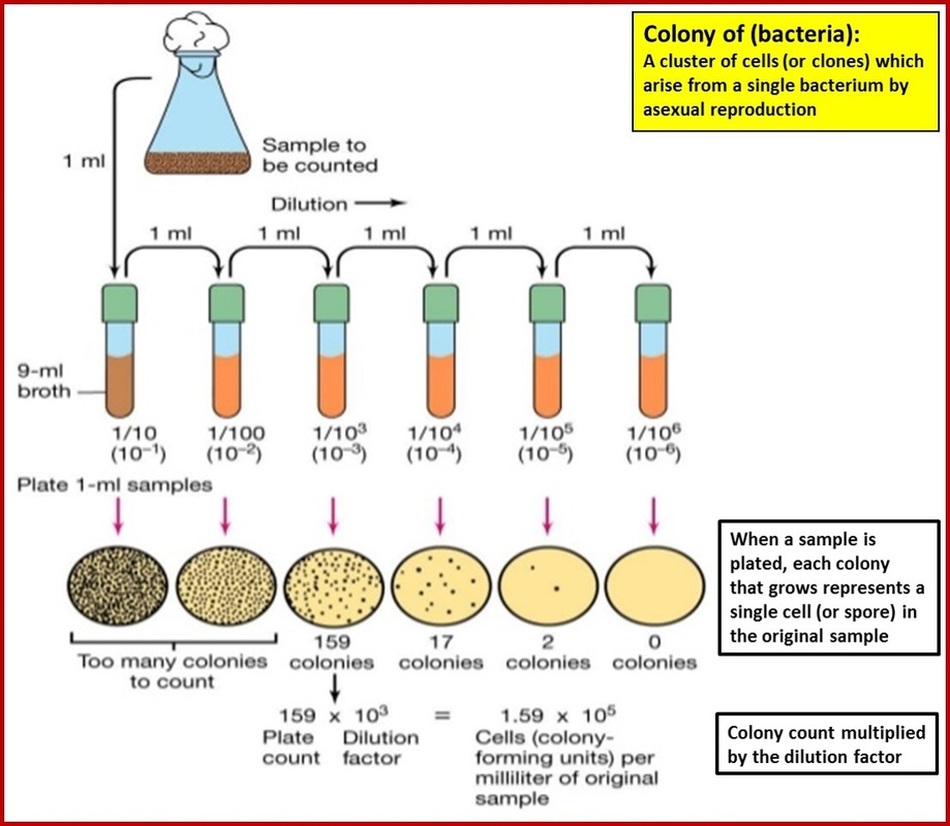
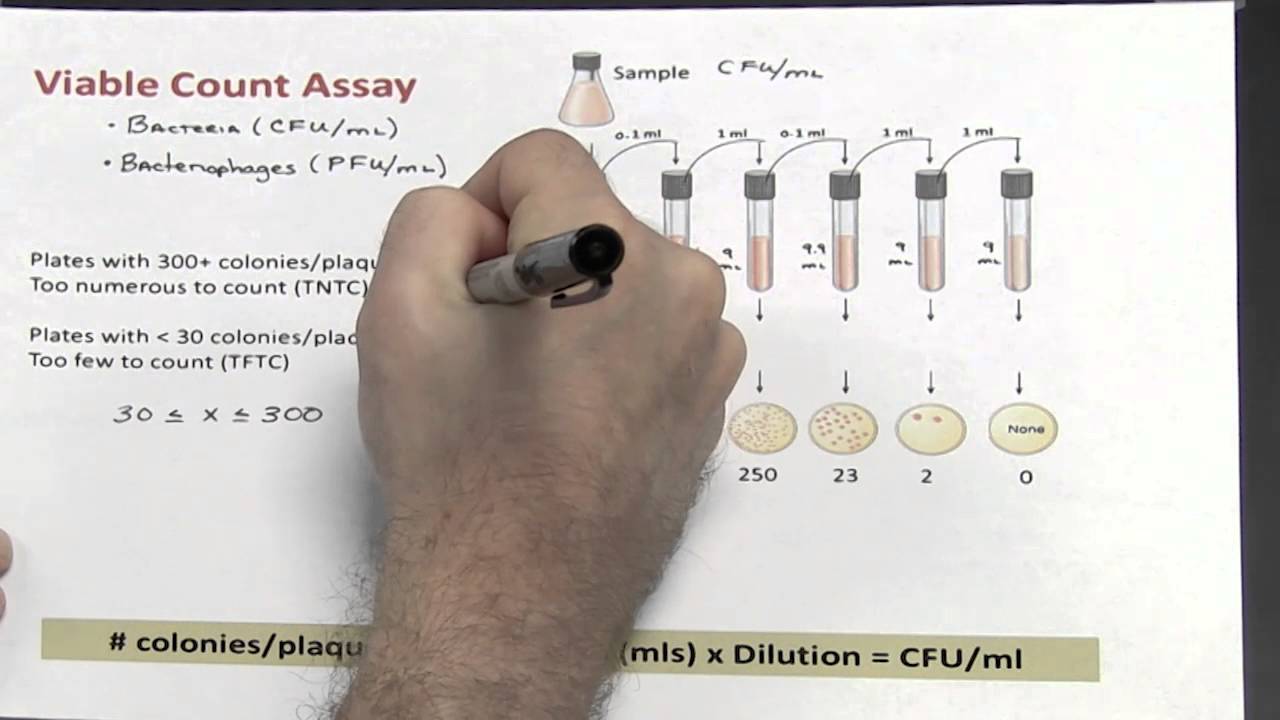
In order to determine the microbial density in a yogurt sample the 10-2 dilution is required. Indicate the steps to follow and detail how to prepare the |
|
Daily Culturing Routine and Culture Dilution
Example 2: For a 1:10 dilution of a 10% parasitemia 10mL stock to a new large 25mL culture… 2.5mL stock culture. 2.5mL washed blood. 20mL warm complete medium. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
Example: A serum sample is diluted twice with buffer A series of five dilutions is made of this first dilution by diluting it 1/10, re-diluting 1/10, and then three times more, each resulting solution then being a 1/10 dilution of the previous one in the series |
|
DILUTION CHART NEW - Caprichem
1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000 CHEMICAL CHEMICAL 1 litre 400ml 200ml 100ml 80ml 40ml 20ml 13ml 10ml 8ml 4ml 2ml |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
milligram = mg = 1/1000 of a g or 10-3 g Example 1: To dilute a streptavidin solution 1:300 Mix 1 unit The initial step combines 1 unit volume culture (10 l) |
|
Dilution of solutions for nurses - Mathcentre
mc-nursing1-1-dilution Introduction This leaflet explains how dilution calculations are performed Solution 10 solution strength is equivalent to 1/ 10 |
|
Dilution and Concentration
taining 10 g of active ingredient with a strength of 20 or 1 : 5 w/v is diluted to Calculating the percentage or ratio strength of a solution made by diluting or con |
|
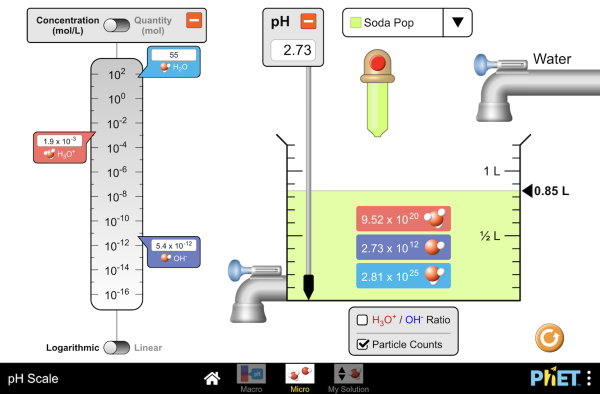
Dilutions - Weber State University
For example, if 1 volume of sample is to be diluted by adding 2 volumes Consider the case where 10mL of soda pop that is being prepared for analysis |
|
DILUTION CHART - Jangro
Dilution Ratio Chemical Measure Water Container 1:5 1:10 1:25 1 : 50 1 : 1 : 200 100 1 :400 1:1 1 part Product to 1 parts Water 1 part Product to 5 parts |
|
Diluting cleaning products using ratios - BBC
Many cleaning products have to be diluted before use Diluting means measuring an amount of Example: to mix a product with a ratio of 1:10 measure : |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
Slide 1 Laboratory Math II: Solutions and Dilutions Philip Ryan, PhD media, cell lysates, liquid acids and bases are all examples of solutions This means that you add 10 milliliters of the 5M NaCl stock solution to a container and |