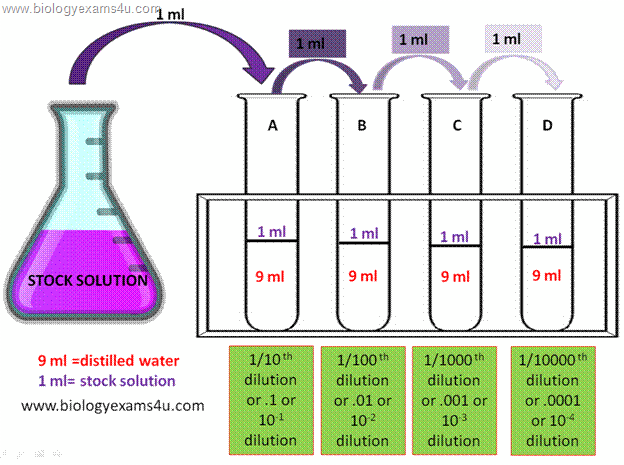
1/10 dilution factor
What is a tenfold dilution?
A tenfold dilution is required to adjust the pH by one (eg, adding 9 cm 3 of water to 1 cm 3 acid). The acid is lowering its acidity. Dilution Factor Equation - After dilution, the dilution factor (or dilution ratio) represents how much of the original stock solution remains in the entire solution.
What is dilution factor?
The dilution factor (or dilution ratio) is the notation used to express how much of the original stock solution is present in the total solution after dilution. It is often given as a ratio but can also be given as an exponent; however, this calculator will only show it as a ratio.

Calculating Dilution Factor

Dilution and Dilution Factor in Microbiology How to Calculate Dilution factor in Serial dilution?

How to Calculate Dilution Factor
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
Microbiologics Dilutions Guide
A log dilution is a tenfold dilution meaning the concentration is decreased by a multiple of ten. To complete a tenfold dilution |
|
Laboratory Math II: Solutions and Dilutions
The dilution factor is the factor by which you will dilute your sample at each step. Mathematically it is equal to the sum of the volume of your stock solution |
|
IDEXX VetTest Chemistry Analyzer Dilution Protocol: Urine Plasma
Dilution Chart. Volumes are for example only. Parts Sample + Parts DI Water = Total Parts. Parts Sample Parts DI Water. Total Parts (Dilution Factor). 1 (10 |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
material to be diluted) + 4 unit volumes of the solvent medium (hence 1 + 4 = 5 = dilution factor). Example 1: To dilute a streptavidin solution 1:300. |
|
1. dilutions and concentrations. liquid and solid samples
of this 1:10 dilution 1 ml corresponds to the sample. In this case |
|
Dilution Ratio Chart
1:10 dilution selling at $7.30 per gallon- $7.30 10= .73 or 73 cents per RTU gallon. Helpful Tips. The smaller number in the ratio is the number of parts |
|
Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor . CALCULATING THE UNDILUTED CONCENTRATION. Often we wish |
|
Counting cells - iGEM
E.g. for a 1:10 dilution add 90 µL PBS and 10 µL cell suspension to an c = mean number of cells in one big 4x4 square * 10 000 * dilution factor (= 1 ... |
|
Answers to Simple Dilution Problems 1) a. 1/10 b. 1/100 c. 1/4 d. 1
The dilution factors are: 2/5 10/1000 and 50/1000. The last one is not actually a dilution factor but is a conversion factor because we take 50 microliter but |
|
Dilutions Occasionally a solution is too concentrated to be used as it
Make a 1/10 dilution of serum with saline 5 Dilute the serum 10 times with Terms such as "1/10", "1/100", "1/500", etc are called dilution factors In each of the |
|
Dilution Factor:
Dilution Factor: A water sample was tested (with dithizone in methylene chloride to make a color solution) for lead content, but was diluted prior to obtaining the |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
material to be diluted) + 4 unit volumes of the solvent medium (hence, 1 + 4 = 5 = dilution factor) Example 1: To dilute a streptavidin solution 1:300 Mix 1 unit |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
The dilution factor is the factor by which you will dilute your sample at each step Mathematically it is equal to the sum of the volume of your stock solution you are |
|
M3013 The Dilution Solution - Students Guide Introduction: Have
A ten-fold dilution is sometimes also written as a 1:10 dilution Here 1:10 shows the ratio between the stock and the final volume of the working solution (100 mL : |
|
M3013 The Dilution Solution MiniLab - Students Guide September
11 sept 2020 · What is the dilution factor of the new solution? → Your lab partner made a 5X dilution A ten-fold dilution is sometimes also written as a 1:10 |
|
Dilutions Tutorial
1/10 where HCl is the diluate and distilled water is the diluent The dilution factor can be written as 1/10 or 1:10 A DF of 1:10 gives the fraction of H+(aq) in the |