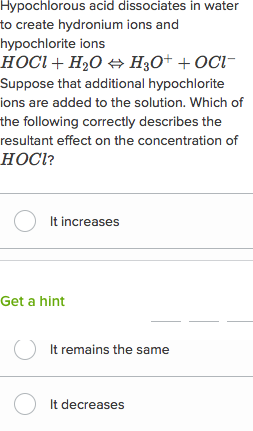
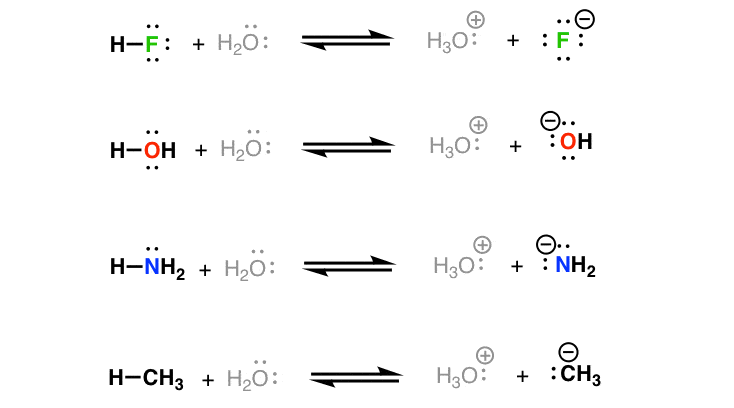
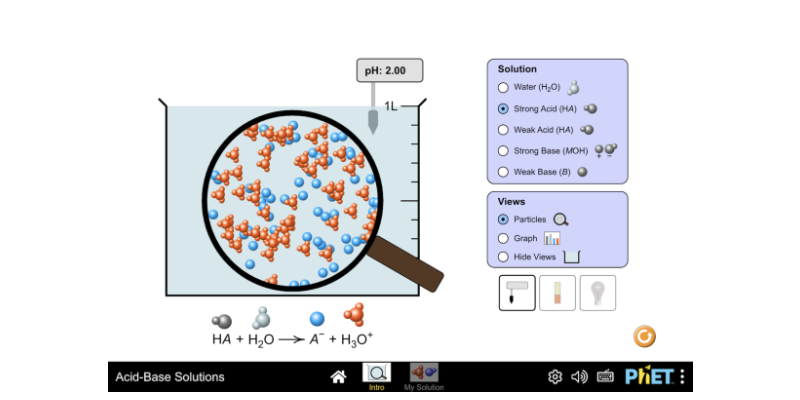
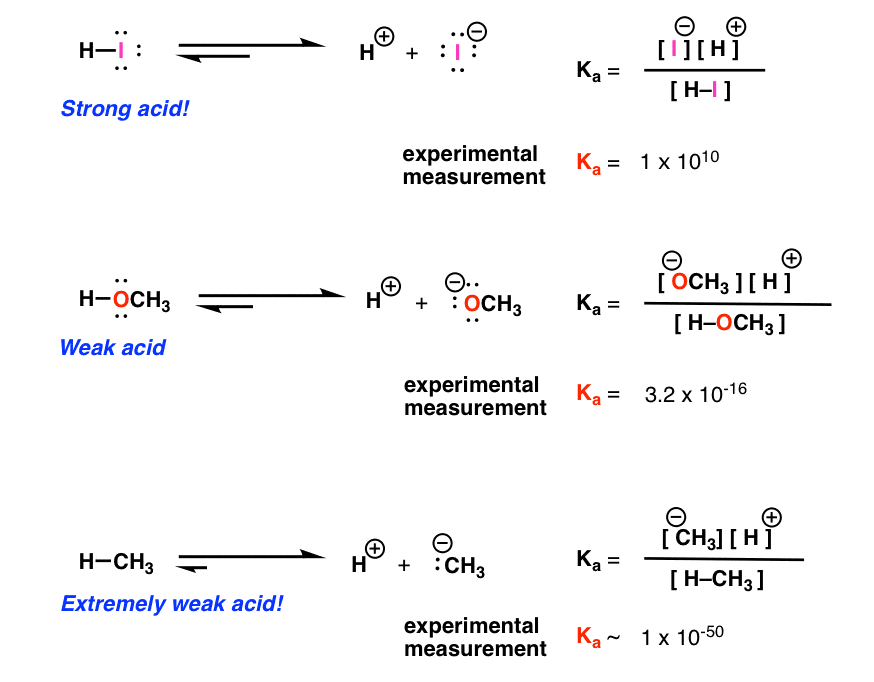acid base practice problems with answers
What is acid question answer?
An acid is a substance that donates protons or accepts electrons.
It dissociates in an aqueous solution to give H+ ions.
A base is a substance that abstracts protons or donates electrons.
It dissociates in an aqueous solution to give OH– ions.What is an example of an acid answer?
Examples of acids include the inorganic substances known as the mineral acids—sulfuric, nitric, hydrochloric, and phosphoric acids—and the organic compounds belonging to the carboxylic acid, sulfonic acid, and phenol groups.
(e) [CO(CO)4P The conjugate acid is HCO(CO)4, shown below.
A drawing of the HCO(CO)4 molecule, the conjugate acid of the tetrahedral CO(CO)4- anion.
The C atoms of the CO ligands are bound to the Co atom.
|
Test2 ch17a Acid-Base Practice Problems
the more concentrated the conjugate base. c. the more concentrated the acid. 11. Ammonia (NH3) acts as a weak base in aqueous solution. What is the |
|
Arterial Blood Gas Case Questions and Answers
Acid-base status: • The patient has a low pH (acidemia). • The PCO2 is high (respiratory acidosis) and the bicarbonate is high (metabolic. |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships. |
|
OChem1 Practice Problems
“standard” valence when forming neutral molecules for example halogens and H are monovalent |
|
Where To Download Redox Practice Problems With Answers Copy
As this Redox Practice Problems With Answers it ends taking place innate one of Chemistry Quiz Questions and Answers (Book 4) - Acids |
|
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY. 1. For each of the species below identify the most acidic proton and provide the. |
|
Read PDF Chemistry Practice Problems Answers ? - covid19.gov.gd
Topical problem solving will focus on limiting reactant stoichiometry identifying types of chemical reactions |
|
Multiple Choice Questions- Acid Base Balance
Multiple Choice Questions- Acid Base Balance What is the underlying acid-base ... An arterial blood sample yields the following values: pH – 6.90. HCO3. |
|
Chapter 11 – Acids and Bases – Practice Problems Section 11.1
Practice Problems. 1. Indicate whether each of the following statements is characteristic of an Arrhenius acid Arrhenius base |
|
Acid-Base Chemistry. Extra Practice Problems
ANSWERS. Acid-Base Chemistry. Extra Practice Problems. 1. A. 2. B. 3. A. 4. D. 5. B. 6. B. 7. A. 8. C. 9. B. 10. B. 11. B. 12. C. 13. A. 14. E. 15. C. 16. D. |
|
Ch 17a Acid-Base Practice Problems
Extra Practice Problems General p4 Answers p12 Ka: Sense + In the Brønsted–Lowry definition of acids and bases, an acid ______ a is a proton donor |
|
Acid-Base Practice Problems
Acid-Base Practice Problems A Identify each chemical as either an “acid” or a “ base” in the following reactions, and identify “conjugate” relationships |
|
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For each
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For each of the following acid base reactions place one of the arrows inside the box to designate the |
|
Chapter 11 – Acids and Bases – Practice Problems Section 111
When H2O acts as a base, it gains one H+, which forms its conjugate acid, H3O+ Practice Problems 8 Write the formula for the conjugate base for each of the |
|
Solutions to Review Problems for Acid/Base Chemistry - CSUN
The resulting 800 mL of solution in Problem 3 is divided into two 400-mL samples If 5 0 mL of 6 0 M HCl are added to one sample, and 5 0 mL of 6 0 M |
|
Acid Base Practice Test - Houston ISD
Acid Base Practice Test ____ 1 Acids ____ 11 Which of the following chemical reactions represents an acid-base reaction? a Answer Section MULTIPLE |
|
Acid Base Practice Problems 1 Write the formula for the conjugate
When solving problems identification of the chemicals is very important Is the chemical a strong acid (ionizes 100 ), a weak acid (partial ionization and has a |
|
Arterial Blood Gas Case Questions and Answers
Acid-base status: • The patient has a low pH (acidemia) • The PCO2 is high ( respiratory acidosis) and the bicarbonate is at the upper end of normal The low pH |
|
Acid Base Problems
exercise Answer initially to the best of your ability Mixed acid-base disorder — The simultaneous presence of more than one As an example, a patient with |
|
Multiple Choice Questions- Acid Base Balance
Multiple Choice Questions- Acid Base Balance What is the underlying acid- base An arterial blood sample yields the following values: pH – 6 90, HCO3 Answers- 1-a, 2-b, 3-c, 4-d, 5-a, 6-c, 7-b, 8-a, 9-d, 10-b, 11-a, 12-a, 13-c, 14-d, 15- c, |












/pHWorksheet-56a12dd95f9b58b7d0bcd1fc.png)