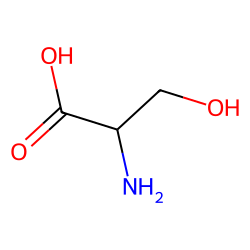
serine pka
|
Approximate pKa values of ionizable groups of amino acids and
Approximate pKa values of ionizable groups of amino acids and peptides (side Serine (hydroxyl): 13 Threonine (hydroxyl): 13 α-carboxyl of free amino acid |
|
CHAÎNE LATERALE NON POLAIRE
ACIDES AMINES : Formules à pH7 (Masse moléculaire) Sigles pKc (α-carboxyl) PKN (α-amino) PKR (chaîne latérale) pHi (pH isoélectrique) |
|
Les acides aminés : Propriétés Physico-chimiques
• Hydroxylés/soufrés: Sérine Cystéine Thréonine Méthionine • Cycliques La chaîne latérale de l'histidine a un pKa=6 0 • Chargées négativement à pH |
|
PKa and pI values of amino acids
pKa and pI values of amino acids Amino acid 3-letter code 1-letter code pKa Cα Serine Ser S 2 21 9 15 - 5 68 Threonine Thr T 2 09 9 10 - |
|
Pka-compilation-williamspdf
7 avr 2022 · pKa Data Compiled by R Williams pKa Values INDEX Inorganic 2 Phenazine 24 Serine 2 21 9 15 6 D-Lysine- 7 93 Threonine 2 63 |
|
Rapid determination of pKa values of 20 amino acids by CZE with
Abstract A rapid and universal capillary zone electro- phoresis (CZE) method was developed to determine the dissociation constants (pKa) of the 20 standard |
Quel est la formule de sérine ?
C3H7NO3La formule semi développée de la sérine est C3H7NO3, sa masse molaire est de 105,0926 g/mol.
Comment savoir si un acide aminé est acide ou basique ?
Pour savoir si un acide aminé est acide ou basique, il suffit de vérifier le groupement R de l'acide aminé : Si le groupement R contient une ou plusieurs fonctions acides carboxyliques donc c'est un acide aminé acide.
Si le groupement R contient une ou plusieurs fonctions amines, c'est un acide aminé basique.Quels sont les 4 acides aminés ?
On compte quatre acides aminés protéinogènes aromatiques : l'histidine, la phénylalanine, le tryptophane et la tyrosine.
- La L-sérine est un acide aminé qui joue un rôle important dans la production de protéines, d'ADN et de membranes cellulaires.
C'est un acide aminé non essentiel, ce qui signifie qu'il peut être fabriqué à partir d'autres acides aminés.
|
Approximate pKa values of ionizable groups of amino acids and
Serine (hydroxyl):. 13. Threonine (hydroxyl): 13 ?-carboxyl of free amino acid: 2 ?-amino of free amino acid: 9.5. C-terminal carboxyl of peptide: 3. |
|
Characterization of a Novel PKA Phosphorylation Site Serine-2030
Apr 29 2005 Abstract—Hyperphosphorylation of the cardiac Ca2 release channel (ryanodine receptor |
|
Phosphorylation of Serine Induces Lysine pKa Increases in Histone
Nov 15 2018 ABSTRACT: The mild acetylating agent |
|
Molecular Basis for Ser/Thr Specificity in PKA Signaling
Jun 25 2020 Although PKA strongly prefers serine over threonine substrates |
|
Control of Adipose Triglyceride Lipase Action by Serine 517 of
Thus serine 517 is a novel “master regulator” of PKA-stimu- lated adipocyte lipolysis. Triglyceride (TAG)3 and other neutral lipids are stored in. |
|
Structural Basis of Perturbed pKa Values of Catalytic Groups in
protease; glycosidase; serine protease; thioredoxin. In addition the pKa value of 9.7 for the phenol group of tyrosine is slightly lower than the pKa. |
|
EXERCICE 1 : Relation entre les structures et les propriétés
(5) Sérine Puis quand le pH atteint et dépasse le pKa du groupement amine ... EXERCICE 6 : Pour la tyrosine en solution aqueuse |
|
Ezrin-anchored PKA phosphorylates serine 369 and 373 on
PKA phosphorylates Cx43 on serine 369 and 373. Keywords: Cx43 as the major PKA phosphorylation sites that increases gap junction assembly at the. |
|
PKa and pI values of amino acids
Custom peptide synthesis |
|
Approximate pK values - University of California Los Angeles
Approximate pKa values of ionizable groups of amino acids and peptides Hydropathy values These numbers are taken from one of many scales that describe the hydrophobicity of the amino acids The more positive the value the more hydrophobic the aa (Likewise the more negative the value the more hydrophilic the aa ) |
Why does phenol have a low pKa?
Why does phenol have a low pKa? Nitro groups are very powerful electron-withdrawing groups. The phenol derivative picric acid has a pKa of 0.25, lower than that of trifluoroacetic acid. Use a resonance argument to explain why picric acid has such a low pKa. Notice that the methoxy group increases the pKa of the phenol group - it makes it less ...
Is pKa the same as Pi?
pI (or isoelectric point) is the pH at which a molecule has no net charge. Mathematically, it is defined as the mean of the pKa values for a molecule. Simple! So, the pI of a protein is determined by the pKa of every amino constituent amino acid. The situation is complicated somewhat by the fact that proteins fold, however.
What pKa value to use for citric acid?
pKa values are 5.21, 4.28 and 2.92 at 25 °C (extrapolated to zero ionic strength). Buffering properties : Citric acid is a good buffering agent for solutions between about pH 2 and pH 8.
What is the definition of PKA in chemistry?
pKa is the negative log value of the dissociation constant (Ka) of the acid. The lower value of pKa reveals the fact that the particular acid is a strong acid and it completely dissociates in water. And the higher value indicates that the particular acid is a weak acid and it partially dissociates in water.
|
Les acides aminés - UNF3S
Constantes caractéristiques des différents acides aminés Nom Code pKa du COOH pKa du Hydroxylés/soufrés: Sérine, Cystéine, Thréonine, Méthionine |
|
Approximate pKa values of ionizable groups of amino acids and
Tyrosine (hydroxyl): 10 5 Lysine (amino): 10 5 Arginine (guanidino): 12 5 Serine (hydroxyl): 13 Threonine (hydroxyl): 13 α-carboxyl of free amino acid: 2 |
|
Chapter 2: Protein Structure and Function
HO pKa=1 80 pKa=9 33 NH3 O Glutamic acid Glutamine Glycine Proline Serine Tyrosine Phenylalanine Threonine Tryptophan Valine arginine Arg HO |
|
Bi102 2015-TDAA-corrigé
(5) Sérine b (1) Glycine c (16) Lysine et (15) Arginine d (11) Méthionine e EXERCICE 6: Pour la tyrosine en solution aqueuse, les pka sont les suivants : |
|
Rapid determination of pKa values of 20 amino acids by - CORE
14 sept 2007 · Like- wise, other amino acids were better detected by C4D than UV: alanine, aspartic acid, isoleucine, leucine, proline, serine, threonine, valine |
|
A L-Phosphoserine is a modified amino acid that is generated in
generated in proteins by phosphorylation of serine residues The amino acid side chain has two acidic protons, which exhibit different pKa values, as shown at |
|
Exam 1 with Answer Key
21 sept 2009 · Serine 9 2 Threonine 26 10 4 Cysteine 1 8 Asparagine 8 8 Glutamine When pH = pka of the R group, you well have 50 of each form |






![Serine biosynthesis from hydroxypyruvate in plants - [PDF Document] Serine biosynthesis from hydroxypyruvate in plants - [PDF Document]](https://www.coursehero.com/doc-asset/bg/80ecffcb4b761d75f34db071a56acfe4fa8b66b3/splits/v9.2.qiv2.clean/split-0-page-1-html-bg-unsplit.png)









![PDF] ✓ Free Download ✓ Red : by Kate SeRine Ù PDF] ✓ Free Download ✓ Red : by Kate SeRine Ù](https://upload.wikimedia.org/wikipedia/commons/thumb/1/1a/L-Cystein_-_L-Cysteine.svg/1200px-L-Cystein_-_L-Cysteine.svg.png)