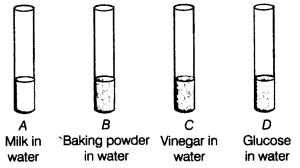
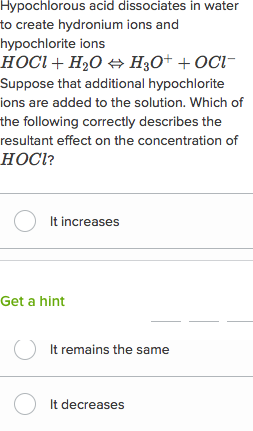
acids and bases questions and answers pdf
Fun Facts
Fun Facts
Acids and bases can help neutralize each other.Acids turn litmus paper red, bases turn it blue.Strong bases can be slippery and slimy feeling.Acids taste sour, bases taste bitter.Proteins are made up of amino acids.Vitamin C is also an acid called ascorbic acid.Ammonia is a base chemical.
|
Jasperse Acid-Base Chemistry. Extra Practice Problems
solution the acid reactant is ______ and its conjugate base product is ... trick question |
|
ACIDS BASES AND SALTS
Add about 2 mL of dilute HCl to both the test tubes. Pass the gas produced in each case through lime water (calcium hydroxide solution) as shown in below figure |
|
Acid Base Practice Test - Houston ISD
neither acids salts |
|
PHYSICAL SCIENCES PAPER 2 (CHEMISTRY) GRADE 12 TERMS
ACIDS AND BASES Terms definitions |
|
CHAPTER - Acids Bases and Salts
What happens when a solution of an acid is mixed with a solution of a base in a test tube? (i) The temperature of the solution increases. |
|
Acids Bases and Salts
Now shake the test tube gently. Do you observe any change in colour of the acid? Add to the acidic solution a drop of sodium hydroxide solution by a |
|
CHAPTER - Acids Bases and Salts
1 What Happens to an Acid or a Base in a Water Solution? Do acids produce ions only in aqueous solution? Let us test this. Activity 2.9. ▫ Take |
|
Stanley Acids and Bases Study Guide
Distinguish between strong acids/bases and weak acids/bases with examples. This may assist especially when answering multiple choice questions. Base 1. Acid 1. |
|
CHAPTER 18 ACID-BASE EQUILIBRIA
Weak Acids: dissociate only partially in solution; it is an equilibrium. HA Question: What are [H3O+] and [OH-] in 0.0012 M NaOH soln at 25 ºC? Answer: NaOH ... |
|
Acids and Bases Acids and Bases Conjugate Pair Question
A buffer solution withstands changes in pH when a limited amount of acid or base is added. ▫. It is composed of a weak acid and a salt of a weak acid with a |
|
Acid Base Practice Test - Houston ISD
neither acids salts |
|
Test2 ch17a Acid-Base Practice Problems
p4 Answers p12. Ka: Sense + Calculations. Using Ka or pKa to Calculate. [H+] and/or pH; using pH to calculate Ka or pKa p5. Conceptual Questions. Acids |
|
PHYSICAL SCIENCES PAPER 2 (CHEMISTRY) GRADE 12 TERMS
answers. If you do not know how to answer a question the answers are there Contain a large amount (number of moles) of acid/base in proportion to the. |
|
Y8 – Acids and Alkalis Science homework booklet Core questions
8 What is a base? Any substance which neutralises an acid. 9 Name 3 common bases in the home. Soap Toothpaste and Baking Powder. |
|
Multiple Choice Questions- Acid Base Balance
Multiple Choice Questions- Acid Base Balance. Q.1- A person was admitted in a coma. Analysis of the arterial blood gave the following values: PCO2 16 mm Hg |
|
Chemistry Chapter 5 Acids Bases and Salts Long Answer Type
Acids Bases and Salts. Long Answer Type Questions. Q1. Write chemical equation when zinc granules react with a) Sulphuric acid b) Hydrochloric acid. |
|
ACIDS BASES AND SALTS
Answer. Soluble bases are called alkalies e.g. sodium hydroxide (NaOH). How will you test for the gas which is liberated when hydrochloric acid reacts ... |
|
Acids And Bases Chapter Review Chemistry Answers - m
23 2022 by guest. Acids And Bases Chapter Review Chemistry. Answers The authors' question-and-answer ... "Practice Test 1 " Answer Key Detailed. |
|
Questions with Answers- Nucleotides & Nucleic Acids A. The
_____ Which structural feature is found in the single-stranded DNA molecule? a) It can have a negatively-charged backbone composed of nitrogenous bases. b) Each |
|
Ncjescoa02.pdf
sulphuric acid nitric acid and hydrochloric acid. Bases are chemical compounds which have bitter ... is mixed with a solution of a base in a test. |
|
Acid Base Practice Test - Houston ISD
a acidic b basic c neutral _____ 6 What is the pH when the hydrogen ion concentration is 1 x 10 –3 M a 2 b Which of the following chemical reactions represents an acid-base reaction? a HBr + KOH Practice Test Answer Section |
|
Ch 17a Acid-Base Practice Problems
p4 Answers p12 Ka: Sense + Calculations Using Ka or pKa to Calculate [H+] and/or pH; using pH to calculate Ka or pKa p5 Conceptual Questions Acids |
|
Unit 12-Solutions & Acids and Bases
27 avr 2020 · Questions-textbook # 1-36 odds (answers in the back of the book) Study Guide: chapter 14 and chapter 18-connects with your textbook |
|
Acids and Bases
Book Pages 126 – 131 Questions Answer individually on YOUR OWN piece of notebook paper 1 Define “Acid” 2 Define “Base” 3 What happens when acids |
|
Quiz - Equilibrium, Acids & Bases
Quiz - Equilibrium, Acids Bases Multiple Choice Identify the letter of the choice that best completes the statement or answers the question CONTENT |
|
ACID BASE HOMEWORK QUESTIONS Part A
a Identify HCl and HF as acids or bases Explain your answer b The pH of the 0 1 M HCl solution is expected to be lower |
|
Multiple Choice Section - Ms Bunneys Classes
Е 03 Acid Base Study Guide - Multiple Choice - Page 1 of 47 THE URGE TO LOOK AT THE ANSWER KEY until you have given all the questions in the section |
|
TEXTBOOKS AND TEACHERS AWARENESS OF DIFFERENT
Abstract: Acid-base reactions can be described in several ways: by formula equations as reactions Second, semi-structured interviews were conducted December 11, http://www nobel se/chemistry/laureates/1903/arrhenius-lecture pdf |
|
QUIZ: CHEMICAL PROPERTIES OF ACIDS AND BASES - NROER
(d) Sodium hydroxide OXIDES Answer questions 3 and 4 on the basis of the following four reactions of four different substances with an acid/base W + Acid |
|
Acids and Bases - Pearson
technicians must also interpret and analyze the test results, which are iar with the sour tastes of vinegar and lemons and other common acids in foods In 1887 |