acids and bases sample problems
|
Solutions to Review Problems for Acid/Base Chemistry
The resulting 800 mL of solution in Problem 3 is divided into two 400-mL samples If 5 0 mL of 6 0 M HCl are added to one sample and 5 0 mL of 6 0 M |
What are 10 examples acid and base?
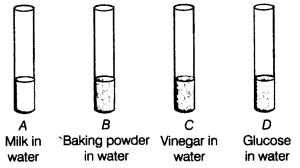
Examples of acids include vinegar, toilet bowl cleaners, lemon juice, and soda.
Examples of bases include egg whites, spinach, and chlorine bleach.
Examples of neutral substances include water, vegetable oil, table salt, and blood.What are some examples of acid base reactions?
13.
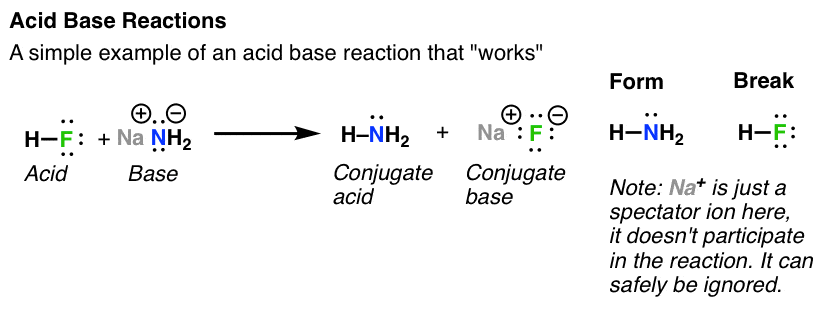
Hydrochloric acid reacts with sodium hydroxide to form sodium chloride (a salt) and water. Hydrogen bromide reacts with potassium hydroxide to form potassium bromide (a salt) and water. Hydrochloric acid reacts with ammonia to form ammonium chloride (a salt).
2) Acid-base reactions (ESBQY)Acids are defined as the substances which release H+ ions when dissolved in water.
They are sour in taste and turns blue litmus into red.
For example: Acetic acid, citric acid.
Bases are the substances which release OH- ions when dissolved in water.
What is an example of an acid base experiment?
Potatoes can also be used in an acid and base experiment.
When potatoes are boiled in acidic water, the potatoes should come out firmer; when they are boiled in water with a base, they come out softer.
To test this, first boil a potato in tap water.
Then, boil a potato in a water and vinegar (the acid) mixture.
|
Test2 ch17a Acid-Base Practice Problems
Acid-Base Chemistry. Extra Practice Problems. General Types/Groups of problems: Conceptual Questions. Acids Bases |
|
POLYPROTIC ACIDS AND BASES: Very important!
Example Problem: Calculate the pH and concentration of each species in a. 5.0 M H3PO4 solution. (a) First dissociation: H3PO4 (aq) ? H+ (aq) + H2PO4. |
|
Solutions to Review Problems for Acid/Base Chemistry
The resulting 800 mL of solution in Problem 3 is divided into two 400-mL samples. If 5.0 mL of 6.0 M HCl are added to one sample and 5.0 mL of 6.0 M. |
|
Sample Exercise 16.1 Identifying Conjugate Acids and Bases
Plan: We approach this problem as we would previous equilibrium problems. We begin by writing the chemical equation for the equilibrium and tabulating the known |
|
Section 19.1. Acid-Base Buffer Solutions
Example: Calculate [H3O+] in a solution that is 0.10 M in HF and 0.20 M in NaF. Also calculate % ionization. Problem: Use HF(aq) ? H+ (aq) + F- (aq) ? |
|
Hints for Working Acid/Base Equilibrium Problems There are only six
There are only six equations needed to solve acid base problems. Examples of Acid/Base Problems Using Different Starting Materials. |
|
Arterial Blood Gas Case Questions and Answers
interpreting acid-base status that is laid out in the Arterial Blood Gas Primer On further history |
|
Chapter 11 – Acids and Bases – Practice Problems Section 11.1
In neutralization a strong or weak acid reacts with a strong base to produce water and a salt. Example Write the balanced chemical equation for the reaction of |
|
W336-titrations-worksheet.pdf
What is the concentration of the HCl solution? 2). You are titrating an acid into a base to determine the concentration of the base. The endpoint of the |
|
Chapter 8 Acid-base equilibria
framework in which any problem in aqueous acid-base equilibria can be solved in a straightforward way. The approach we take is to distinguish four regimes:. |
|
Ch 17a Acid-Base Practice Problems
Extra Practice Problems General Types/Groups of problems: Conceptual Questions Acids, Bases, and Conjugates, Miscellaneous p1 Kb and pKb, Base |
|
Acid-Base Practice Problems
Acid-Base Practice Problems A Identify each chemical as either an “acid” or a “ base” in the following reactions, and identify “conjugate” relationships |
|
Solutions to Review Problems for Acid/Base Chemistry - CSUN
The resulting 800 mL of solution in Problem 3 is divided into two 400-mL samples If 5 0 mL of 6 0 M HCl are added to one sample, and 5 0 mL of 6 0 M |
|
Chapter 11 – Acids and Bases – Practice Problems Section 111
When H2O acts as a base, it gains one H+, which forms its conjugate acid, H3O+ Practice Problems 8 Write the formula for the conjugate base for each of the |
|
Acids and Bases - Pearson
SAMPLE PROBLEM 14 1 Names and Formulas of Acids and Bases a Identify each of the following as an acid or a base and give its name: 1 H3PO4, ingredient |
|
TOPIC 5: ACIDS AND BASES - Manitoba Education
C12-5-07 Distinguish between strong and weak acids and bases C12-5-09 Use Ka or Kb to solve problems for pH, percent dissociation, and Examples: analogies, concept frames, concept maps, manipulatives, particulate representations |
|
Acid Base Practice Problems 1 Write the formula for the conjugate
When solving problems identification of the chemicals is very important Is the chemical a strong acid (ionizes 100 ), a weak acid (partial ionization and has a |
|
Hints for Working Acid/Base Equilibrium Problems There are only six
There are only six equations needed to solve acid base problems There are only five Examples of Acid/Base Problems Using Different Starting Materials |
|
Sample Exercise 161 Identifying Conjugate Acids and Bases
Note: In this problem and all that follow, we assume, unless stated otherwise, that the temperature is 25 ºC Calculate the concentration of OH–(aq) in a solution in |
|
Practice Problems on Acid-‐Base Chemistry 1 For - Chemwiscedu
Practice Problems on Acid-‐Base Chemistry 1 For each of the following acid base reactions place one of the arrows inside the box to designate the extent of the |





/pHWorksheet-56a12dd95f9b58b7d0bcd1fc.png)