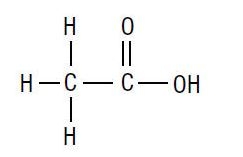
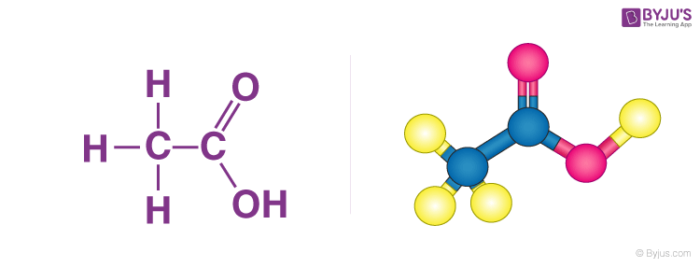
acetic acid formula
Biologically, acetic acid is an important metabolic intermediate, and it occurs naturally in body fluids and in plant juices.
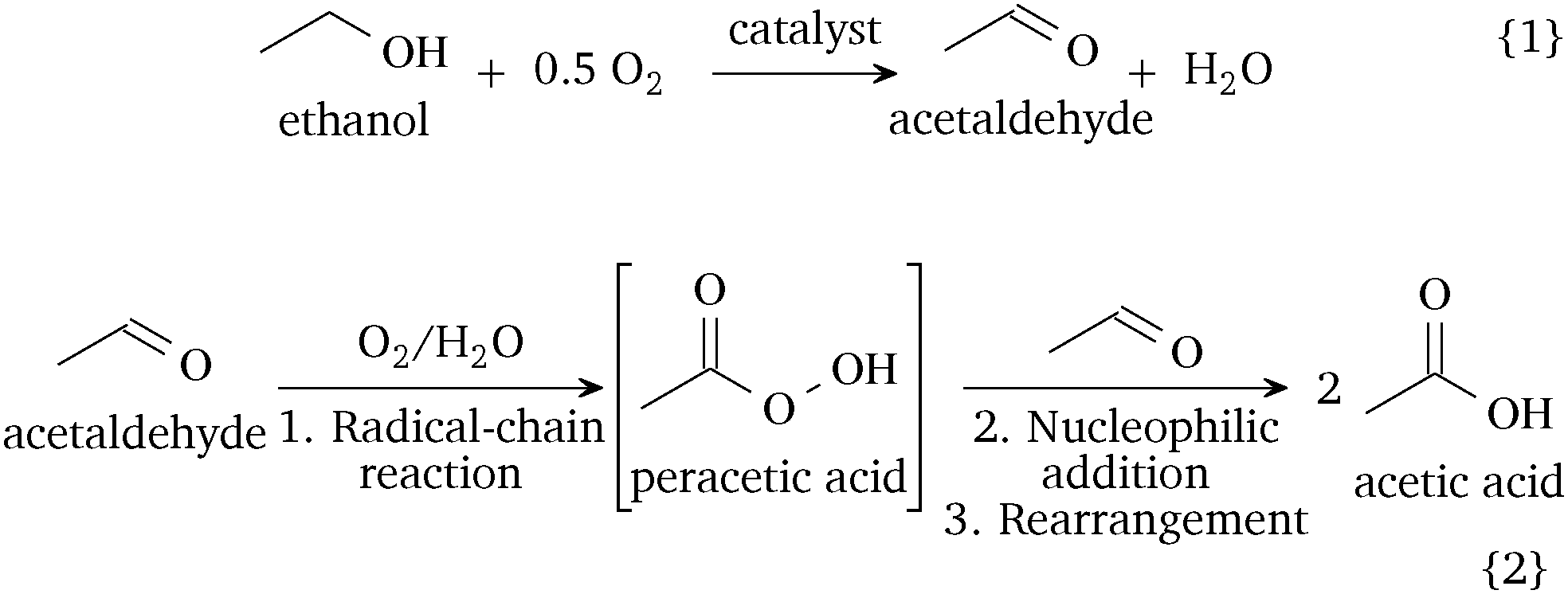
Acetic acid has been prepared on an industrial scale by air oxidation of acetaldehyde, by oxidation of ethanol (ethyl alcohol), and by oxidation of butane and butene.
What is the basic formula of acetic acid?
Acetic Acid (CH3COOH)
Why is CH3COOH called acetic acid?
The systematic name "ethanoic acid", a valid IUPAC name, is constructed according to the substitutive nomenclature.
The name "acetic acid" derives from the Latin word for vinegar, "acetum", which is related to the word "acid" itself. "Glacial acetic acid" is a name for water-free (anhydrous) acetic acid.
What is the chemical name of acetic acid?
Acetic acid is also known as ethanoic acid, ethylic acid, vinegar acid, and methane carboxylic acid.
Acetic acid is a byproduct of fermentation, and gives vinegar its characteristic odor.
|
Recommendation from the Scientific Committee on Occupational
Occupational Exposure Limits for acetic acid Structural formula: ... The largest use of acetic acid is as a chemical intermediate in the manufacture of ... |
|
Degradation of acetic acid with sulfate radical generated by
15 mai 2020 ions yield a greater mineralization of acetic acid than the ... reactions with the carbon mineralized inhibit the reaction of the solute ... |
|
The Reaction of 4 7-Dichloroquinoline with Acetic Acid
the formation of IV. The latter is a powerful acylating agent and reacts with a molecule of acetic acid (or acetate ion if available) to form V. |
|
Research paper determination of adsorption and kinetic parameters
reaction was found to be 45.4kJ mol? 1. KEYWORDS: butyl acetate acetic acid |
|
Esterification of glycerol with acetic acid over Lewatit catalyst
Esterification reaction with a phosphotungstic acid catalyst reactant ratio of 3.8% |
|
Kinetics of esterification of acetic acid and methanol using Amberlyst
Amberlyst 36 was found to be an effective solid catalyst for this reaction compared to the other solid catalysts. The reaction was performed in the temperature |
|
Recent advances in the methanol carbonylation reaction into acetic
21 déc. 2021 Thus 70 % of acetic acid and 22 % methyl acetate are produced with small amounts of methane |
|
Acetic acid - assay procedure
This corrected. A2 value will provide the acetic acid concentration. Total acetate concentration (including esters) can be determined by allowing the reaction |
|
Acetic Acid Solution 6M
23 janv. 2009 MSDS # 3.00. Acetic Acid Solution 6M. Scholar Chemistry. Section 9: Physical and Chemical Properties. Molecular formula. CH3COOH. |
|
RESEARCH PAPER Esterification of acetic acid with methanol over
KEYWORDS: ion exchange resin methyl acetate |
|
ACETIC ACID, GLACIAL
as the reaction product of methanol and carbon dioxide Chemical name Acetic acid, ethanoic acid C A S number 64-19-7 Chemical formula C2H4O2 |
|
Acetic acid - Sigma-Aldrich
Appearance: Clear colorless liquid Molecular formula: CH3COOH Molecular weight: 60 05 Density: 1 049 g/mL Melting point: 16 7EC Boiling point: 118EC |
|
Acetic Acid, Glacial
It is one of the most impor- tant large-volume, synthetic organ- ic acids The major use for Acetic Acid is as a raw material for vinyl acetate, either by reaction with |
|
Vinegar - Kingsborough Community College
In the reaction between acetic acid and sodium hydroxide, the acetic acid donates a proton to the hydroxide ion and acts as an acid The hydroxide ion accepts a |
|
PH in Acetic Acid - Yokogawa
Acetic acid (systematically named ethanoic acid is an organic compound with the chemical formula CH3COOH (also written as CH3CO2H or C2H4O2) It is a |
|
Acetic Acid in Water
Q: What ions will this salt produce in an aqueous solution? Predict whether the addition of sodium acetate will have an impact on the reaction of acetic acid with |
|
Acetic Acid Solution, 6M
23 jan 2009 · Molecular formula CH3COOH Appearance Clear, colorless liquid Molecular weight 60 05 Odor vinegar Specific Gravity |

















/45855026674_f85153017e_b-5c49cb0ac9e77c0001da1c9f.jpg)