acetic acid into methane
How will you convert CH3COOH to CH3 CH3?
Acetic acid is reduced to ethane on reaction with HI in the presence of Phosphorus.
The reaction is CH3COOH + 6HI P,500K → CH3-CH3 + 2H2O +3I2Here the function of P is same as in the reaction alkyl halide with HI and red P.How will you prepare methane and ethane from acetic acid?
Answer: Acetic acid reacts with NaOH to form sodium acetate.
This sodium acetate on distillation with soda lime gives methane.
This sodium acetate on electrolysis gives ethane.What is acetic acid? Acetic acid is also known as ethanoic acid, ethylic acid, vinegar acid, and methane carboxylic acid.
Acetic acid is a byproduct of fermentation, and gives vinegar its characteristic odor.
Vinegar is about 4-6% acetic acid in water.
How do you convert acetate to methane?
Decarboxylation reaction: It is the chemical reaction in which a carboxyl group leaves and releases carbon dioxide.
Sodium acetate is converted to methane by the decarboxylation with soda lime.
Soda-lime is a mixture of sodium hydroxide and calcium hydroxide and the molecular is $CaHNa{O_2}$.
|
THE DECOMPOSITION OF ACETIC ACID IN THE PRESENCE OF
temperature acetone and methane being two ofthe products. Results. In passing acetic acid over silica gel the maximum yield of acetone is. |
|
Methane Upgrading of Acetic Acid as a Model Compound for a
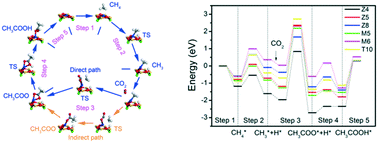
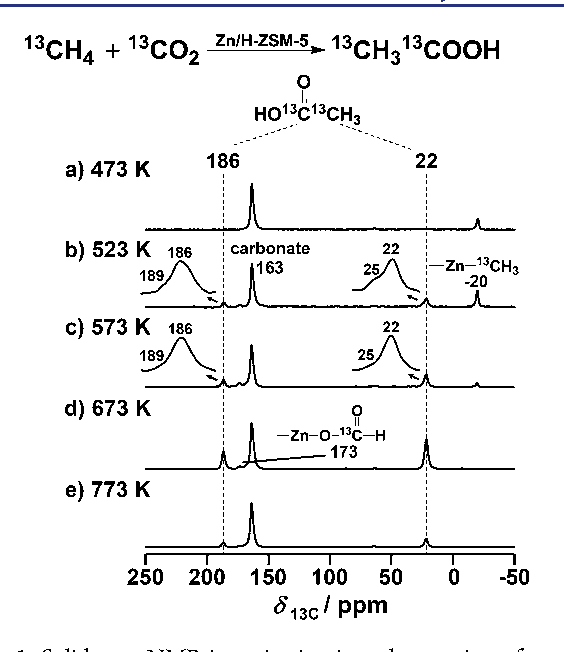
Direct evidence of methane incorporation into aromatics over 5%Zn- Keywords: Methane; Acetic acid; Catalyst; Aromatization; ZSM-5. |
|
Anaerobic Degradation of Individual Components from 5
Apr 13 2021 For the retarded degradation |
|
KETENE FROM ACETIC ACID
ammonia system so the methyl group is the analog in the methane system. On this basis carbonic acid |
|
Singleâ•Pot Conversion of Methane into Acetic Acid in the Absence
Single-Pot Conversion of Methane into Acetic. Acid in the Absence of CO and with Vanadium. Catalysts Such as Amavadine**. Patrícia M. Reis JosØ A. L. Silva |
|
Methane Reacts with Heteropolyacids Chemisorbed on Silica to
Dec 26 2012 With these systems |
|
Diffusion coefficient of acetic acid into trichloro-methane and water
C2 H4 O2. (1) acetic acid. 64-19-7. C H Cl3. (2) trichloro-methane. 67-66-3. H2 O. (3) water. 7732-18-5. Mutual Diffusion Coefficient Matrix: Dij (xi |
|
Direct and Remarkably Efficient Conversion of Methane into Acetic
methane into acetic acid in trifluoroacetic acid (TFA) under moderate conditions |
|
Methane to Acetic Acid over Cu-Exchanged Zeolites: Mechanistic
Jan 6 2015 In this work |
|
Pt-catalyzed oxidative carbonylation of methane to acetic acid in
in the presence of CO this product undergoes carbonylation to acetic acid. acid from methane in a single step |
|
Conversion of acetic acid to methane by thermophiles
A thermo- philic syntrophic coculture converts acetate to methane by a two-step process in which acetate is first oxidized to H 2 and CO2 by an acetate oxidiz- ing rod (AOR) followed by methanogenesis from H e and CO z by a thermophific Methanobaeterium |
|
Formation of methane from propionate - CORE
First, ethanol was oxidized to acetic acid by the removal of hydrogen Second, carbon dioxide acted as a hydrogen acceptor and was reduced to methane gas |
|
Catalytic, Oxidative Condensation of CH4 to CH3COOH in - Science
25 jui 2003 · conversion of methane (or coal) to syn-gas, conversion of syn-gas to methanol, and finally carbonylation of the methanol to acetic acid |
|
2C2H5OH + C02 -2CH3COOH + CH4 - PNAS
Several years ago' it was shown that the formation of methane in the fermentation of acetic acid arises by an oxidation of ethyl alcohol, carbon dioxide being |
|
STUDIES ON THE METHANE FERMENTATION: V BIOCHEMICAL
Acetic acid formed by the oxidation of ethanol was identified by the sodium uranyl acetate micro- chemical reaction and was shown to be pure acetic acid by Du- |


![PDF] Mechanistic insight into the formation of acetic acid from PDF] Mechanistic insight into the formation of acetic acid from](https://pubs.rsc.org/en/Content/Image/GA/C9CY01803D)