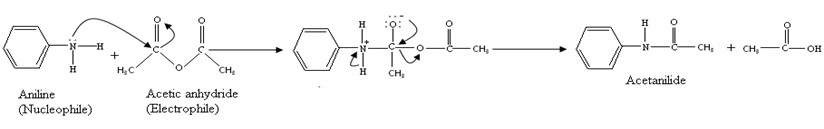
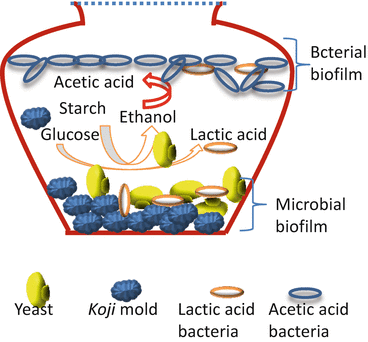
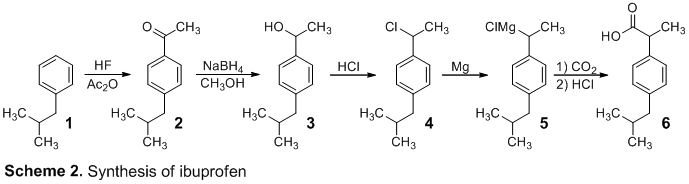
acetic anhydride synthesis mechanism
|
Investigation of the synthesis of acetic anhydride by homogeneous
These observations indicate that the mechanism of the acetic anhydride synthesis may be analogous in some respects to that of methanol carbonylation in the |
What is the mechanism of anhydride synthesis?
Synthesis of anhydrides
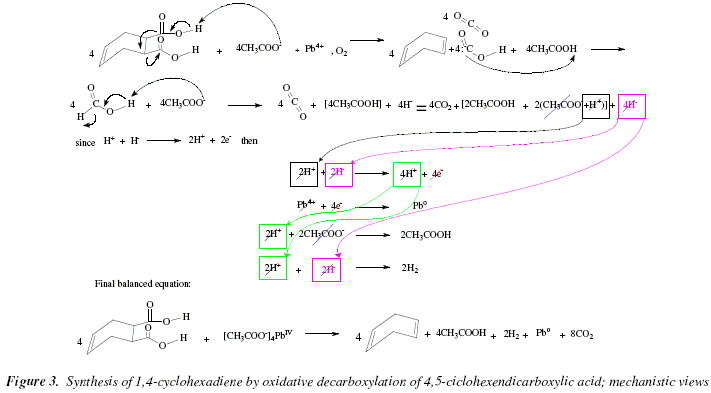
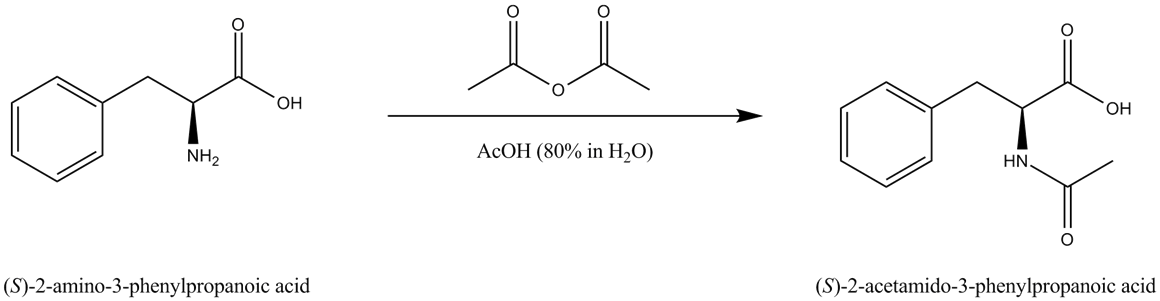
The mechanism involves a double addition of the acid to the dicarbonate, affording a carboxylic anhydride and CO2.The mechanism involves four steps-nucleophilic attack by alcohol, deprotonation by pyridine, leaving group removal and protonation of carboxylate.
When acetic anhydride reacts with alcohol, it does not produce two molecules of ester, but an ester and ethanoic acid.
How is acetic anhydride synthesized?
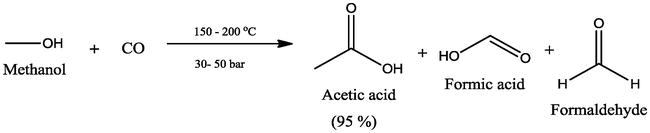
Acetic anhydride is produced by carbonylation of methyl acetate: CH 3CO 2CH 3 + CO → (CH 3CO) 2O.
The Tennessee Eastman acetic anhydride process involves the conversion of methyl acetate to methyl iodide and an acetate salt.
|
Experiment 8 – Synthesis of Aspirin
higher yield (since acetic anhydride is much more reactive than acetic acid). The reaction is shown on the following page. C. OH. O. Salicylic Acid. |
|
Study on Gas-Phase Mechanism of Chloroacetic Acid Synthesis by
15-Jan-2014 The process of acetic acid catalysis and chlorination for synthesizing chloroacetic acid can exist in not only gas phase but also liquid phase. |
|
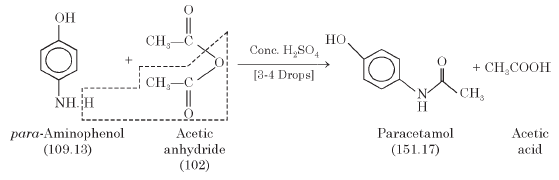
Synthesis of paracetamol by acetylation
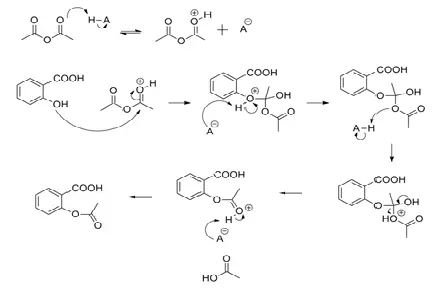
The reaction mechanism can be devised in the following scheme for the general acetylation of an alcohol by acetic anhydride originating an ester and acetic acid |
|
MECHANISM OF CHLORINATION PROCESS: FROM ACETIC ACID
Fig. 1 – The effect of catalyst amount of acetic anhydride on monochloroacetic acid generation. As shown in Figure 1 the production amount of. |
|
MECHANISM OF CHLORINATION PROCESS: FROM ACETIC ACID
Fig. 1 – The effect of catalyst amount of acetic anhydride on monochloroacetic acid generation. As shown in Figure 1 the production amount of. |
|
Experiment 5 - Synthesis of Aspirin
The synthesis reaction of aspirin is shown below: Since acetic acid is very soluble in water it is easily separated from the aspirin product. The aspirin. |
|
ON THE MECHANISM OF FATTY ACID SYNTHESIS IN VIVO
Acetic acid or a closely related 2-carbon unit is the most likely imme- diate precursor for fatty acids in the animal organism (1). This Cz unit. |
|
Friedel Crafts Acetylation of Ferrocene
Do the reaction below and follow in the footsteps of chemistry giants! Acetic anhydride and phosphoric acid react to form the acylium ion electrophile. |
|
Acetic Anhydride Polymerization as a Pathway to Functional Porous
20-Apr-2021 Additionally direct incorporation of catalytic moieties within catalyst synthesis is generally better than postfunctionalization or ... |
|
Mechanism Studies of LCP Synthesis
04-May-2011 functionality accompanied by the elimination of acetic acid. The model system was the reaction of. p-t-butylphenyl acetate with benzoic ... |
|
Synthesis of paracetamol by acetylation - The Royal Society of
The reaction mechanism can be devised in the following scheme for the general acetylation of an alcohol by acetic anhydride originating an ester and acetic acid |
|
8-Synthesis of Aspirin
The phenol group on the salicylic acid forms an ester with the carboxyl group on the acetic acid If acetic anhydride is used instead of acetic acid, the reaction is much faster and has a higher yield (since acetic anhydride is much more reactive than acetic acid) The reaction is shown on the following page |
|
Experiment 5 - Synthesis of Aspirin
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride A small amount of a strong acid is used as a catalyst which speeds up the reaction |
|
Synthesis of Aspirin
Synthesis of Aspirin Synthesis Purification Characterization Reaction mechanism OH Mix salicylic acid (solid) with acetic anhydride (liquid) and a catalytic |
|
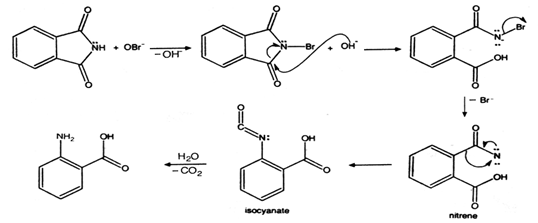
CHEM–333: Lab Experiment 1: Synthesis of N-Acetylanthranilic Acid
At this point the mechanism likely is a bit advanced but you will learn some acetic anhydride to produce an intermediate, benzisoxazinone (not isolated) which |
|
Organic Experiments - School of Chemistry University of Leeds
The synthesis of paracetamol can be broken down into 3 parts: Acetic anhydride, hydrochloric acid and sodium hydroxide are corrosive – wear The ' curly' arrow mechanism below shows the bond forming/breaking process that occurs + |
|
MECHANISM OF CHLORINATION PROCESS: FROM ACETIC ACID
monochloroacetic acid in a laboratory scale glass tube reactor at 105°C This synthesis reaction was an autocatalytic process, acetic anhydride was the catalyst |


















.gif)