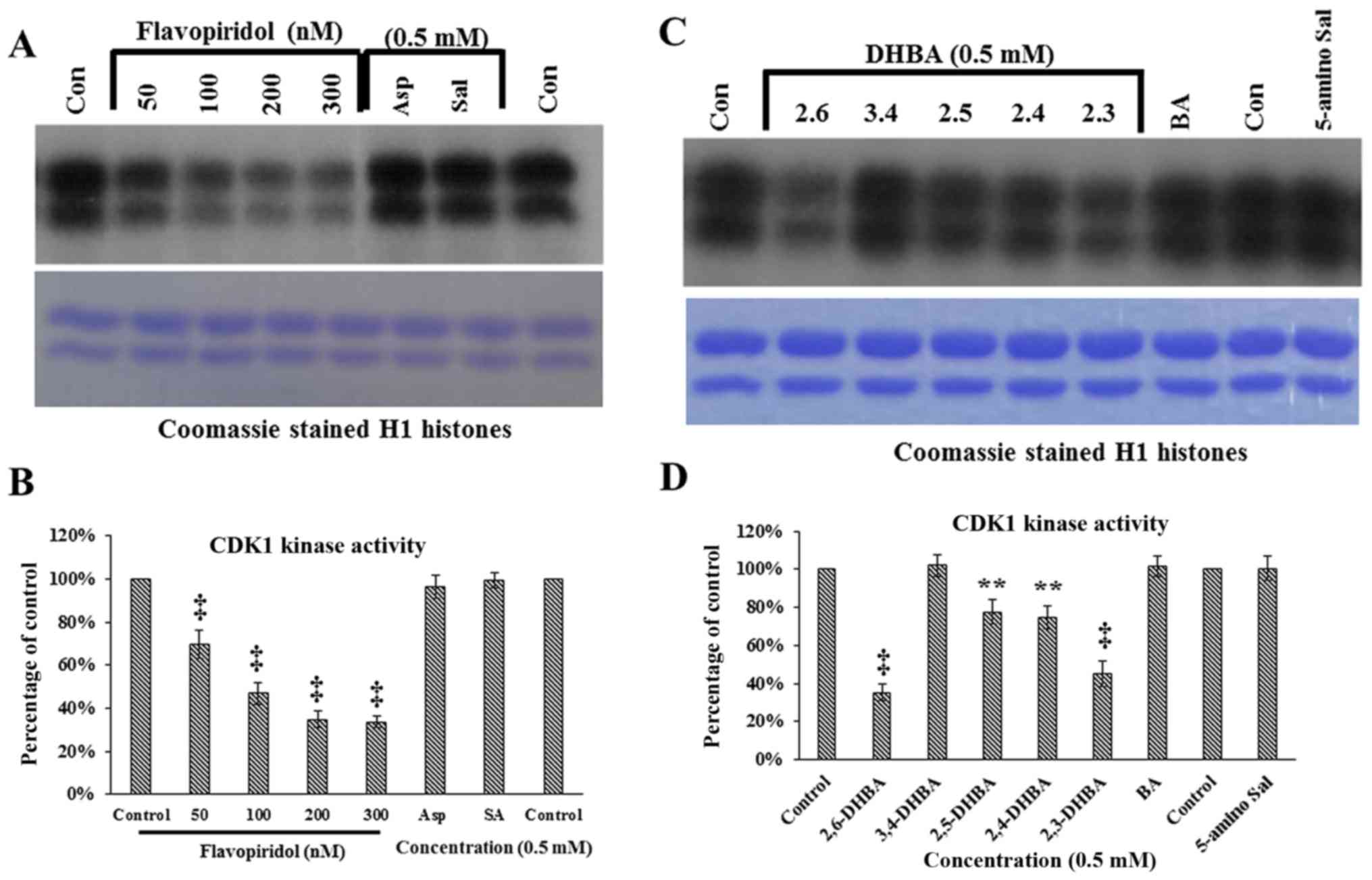
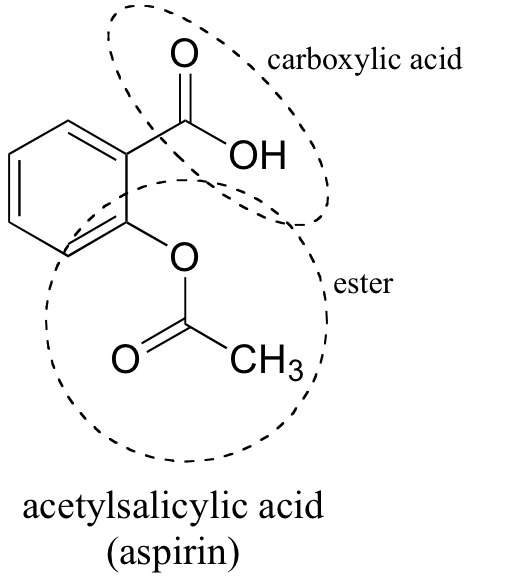
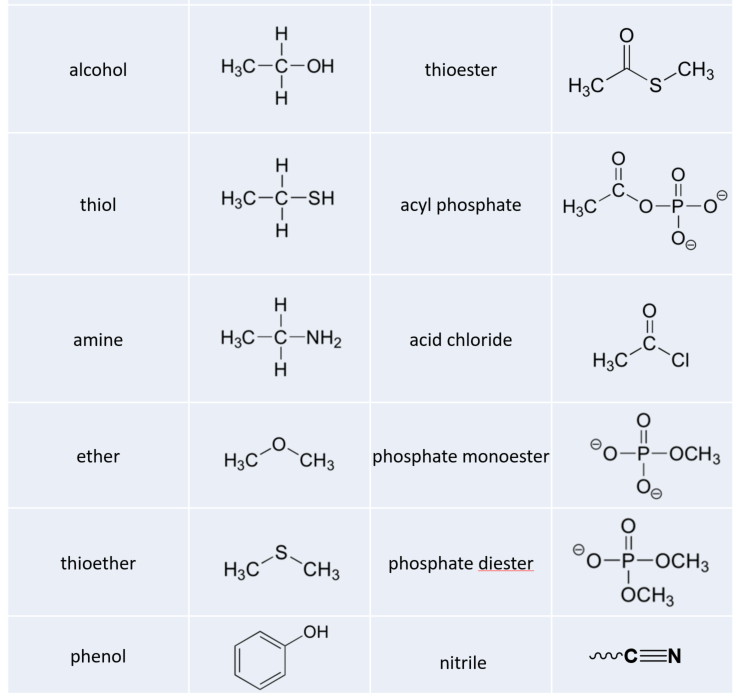
acetylsalicylic acid functional groups
What functional groups are in salicylate?
The salicylic acid starting material has two unique functional groups: a carboxylic acid and a phenol.
The O-H phenol (3230–3000 cm-1), O-H carboxylic acid (3000–2500 cm-1, 881 cm-1) and the C=O carboxylic acid (1652 cm-1) absorbing bands are all labeled in the top spectrum.What functional groups are used in the synthesis of aspirin?
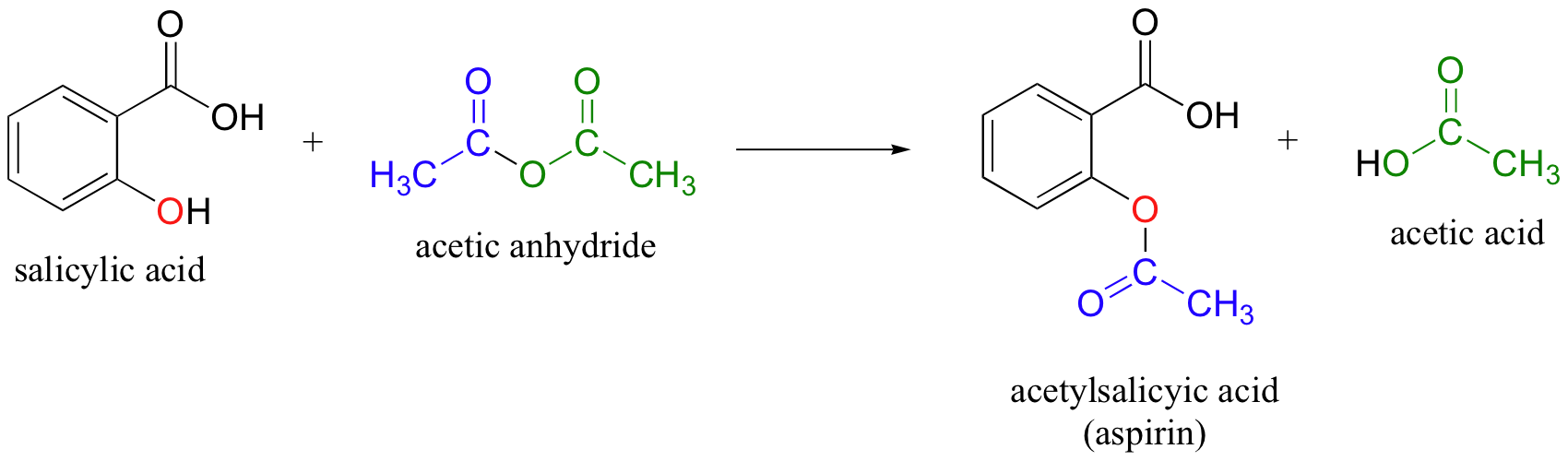
Aspirin is prepared by chemical synthesis from salicylic acid, through acetylation with acetic anhydride.
The molecular weight of aspirin is 180.16g/mol.
It is odourless, colourless to white crystals or crystalline powder.What are the functional groups of paracetamol?
Paracetamol contains three functional groups: the hydroxyl group (OH), the amide group (HN-CO-R), and the aromatic group (benzene ring), as shown in Fig.
Salicylic acid (2-hydroxybenzoic acid) is formed of a benzene ring to which 2 adjacent groups, carboxylic group and hydroxy group, are attached.
We don't normally consider benzene to be a functional group, so that's why the hydroxyl and carboxylic are the ones that count.
|
Yin and Yang in Chemistry Education: The Complementary Nature
The purpose of the experiment was to synthesize aspirin (acetylsalicylic acid) and wintergreen identify functional groups within a molecule and can be. |
|
Yin and Yang in Chemistry Education: The Complementary Nature
The purpose of the experiment was to synthesize aspirin (acetylsalicylic acid) and wintergreen identify functional groups within a molecule and can be. |
|
ORGANIC CHEMISTRY: Aspirin and Esters
The modified compound contains a functional group called an ester. The ester of salicylic acid called acetylsalicylic acid is now known as aspirin and is |
|
Section 3 General Self Assessment Answers 2.3 Solving pH/pK
Match the pKa values provided with the appropriate functional groups. For each functional group identify the name of the group and whether it is acidic or |
|
SYNTHESIS OF ASPIRIN
synthesize acetylsalicylic acid (aspirin) by carrying out a simple organic As a result the two acidic functional groups serve to lower the pH of. |
|
Didactic Strategy for Learning and Teaching of Functional Groups in
each functional group provides specific chemical properties and (such as ethyl alcohol citric acid |
|
Chapter 5 Carboxylic Acids and Esters
Learn to recognize the carboxylic acid ester |
|
1 University of Wisconsin Chemistry 116 Preparation and
and the alcohol functional groups and is capable of two different esterification reactions To make acetylsalicylic acid |
|
Experiment 8 – Synthesis of Aspirin
In 1899 the Bayer Company in. Germany patented a drug they called aspirin |
|
Didactic Strategy for Learning and Teaching of Functional Groups in
The molecules that were selected contain products of common use in daily life. (such as ethyl alcohol citric acid |
|
What are the functional groups in the aspirin - usersmiamiohedu
functional groups in the aspirin molecule? 1 Benzene ring 2 Carboxylic acid 3 Ester 10 3 Ester Formation Ester functional groups are often found in drugs, |
|
The Complementary Nature of FT-IR and NMR Spectroscopies
to synthesize aspirin (acetylsalicylic acid) and wintergreen oil (methyl salicylate) identify functional groups within a molecule and can be used to quantitatively |
|
SYNTHESIS OF ASPIRIN - Chem21Labs
synthesize acetylsalicylic acid (aspirin) by carrying out a simple organic reaction, • separate Salicylic acid contains two functional groups, a carboxylic acid |
|
Aspirin An ab Initio Quantum-Mechanical Study of Conformational
The potential energy surface of acetylsalicylic acid, aspirin, has been explored at the between the functional groups in all aspirin isomers including the |
|
Experiment 12 - Organic Chemistry
1) Waft the aspirin crystals carefully, can you detect an odor? What is it? 2a) Which functional group in the salicylic acid compound was affected? 2b) What is the |
|
1 University of Wisconsin Chemistry 116 Preparation and
and the alcohol functional groups and is capable of two different esterification reactions, depending To make acetylsalicylic acid, the -OH group of the salicylic |
|
ASPIRIN SYNTHESIS - University of Helsinki
Which functional groups can be found in salicylic acid and aspirin? Salicylic acid: Benzene, hydroxyl and carboxylic acid Aspirin: Benzene, carboxylic acid and |
|
Experiment 5 - Synthesis of Aspirin
To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride A small amount of a strong acid is used as a catalyst which speeds up the reaction |
|
Esterification of Salicylic Acid - Westfield State University
Esterification of Salicylic Acid: The synthesis of cool smelling acetylsalicylic acid (aspirin) see equation 1 OH OH O O OH O O O functional group Thus |

























![Chapter 5 Carboxylic Acids and Esters - [PDF Document] Chapter 5 Carboxylic Acids and Esters - [PDF Document]](https://i1.rgstatic.net/publication/7330490_Synthesis_and_transdermal_properties_of_acetylsalicylic_acid_and_selected_esters/links/5a33e3e1aca27247eddc0998/largepreview.png)