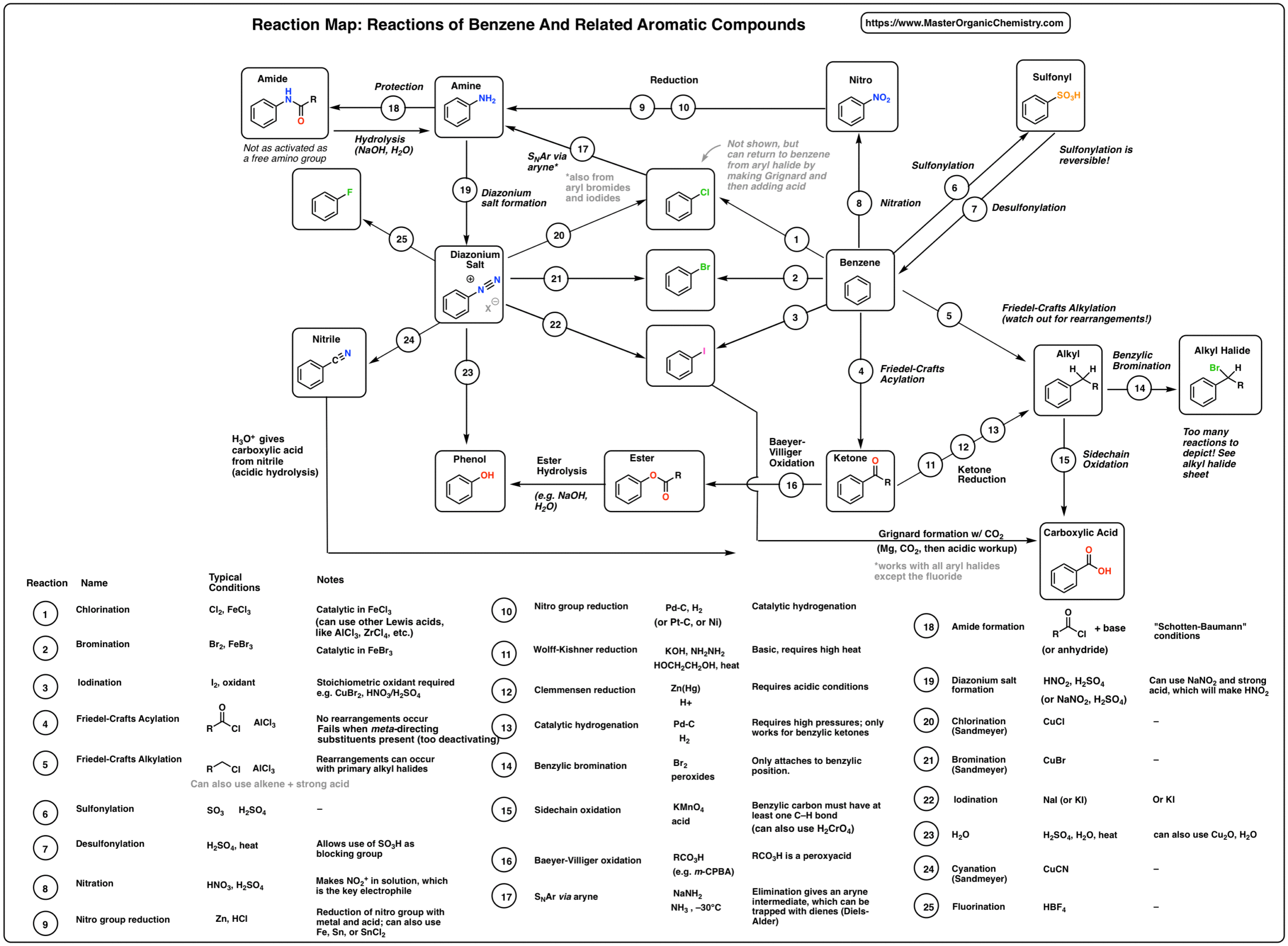
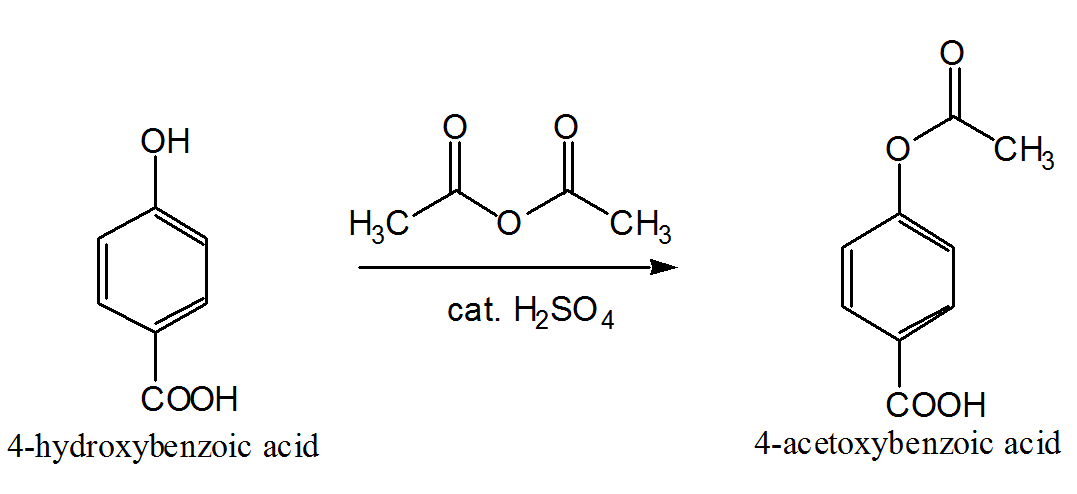
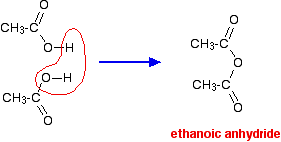
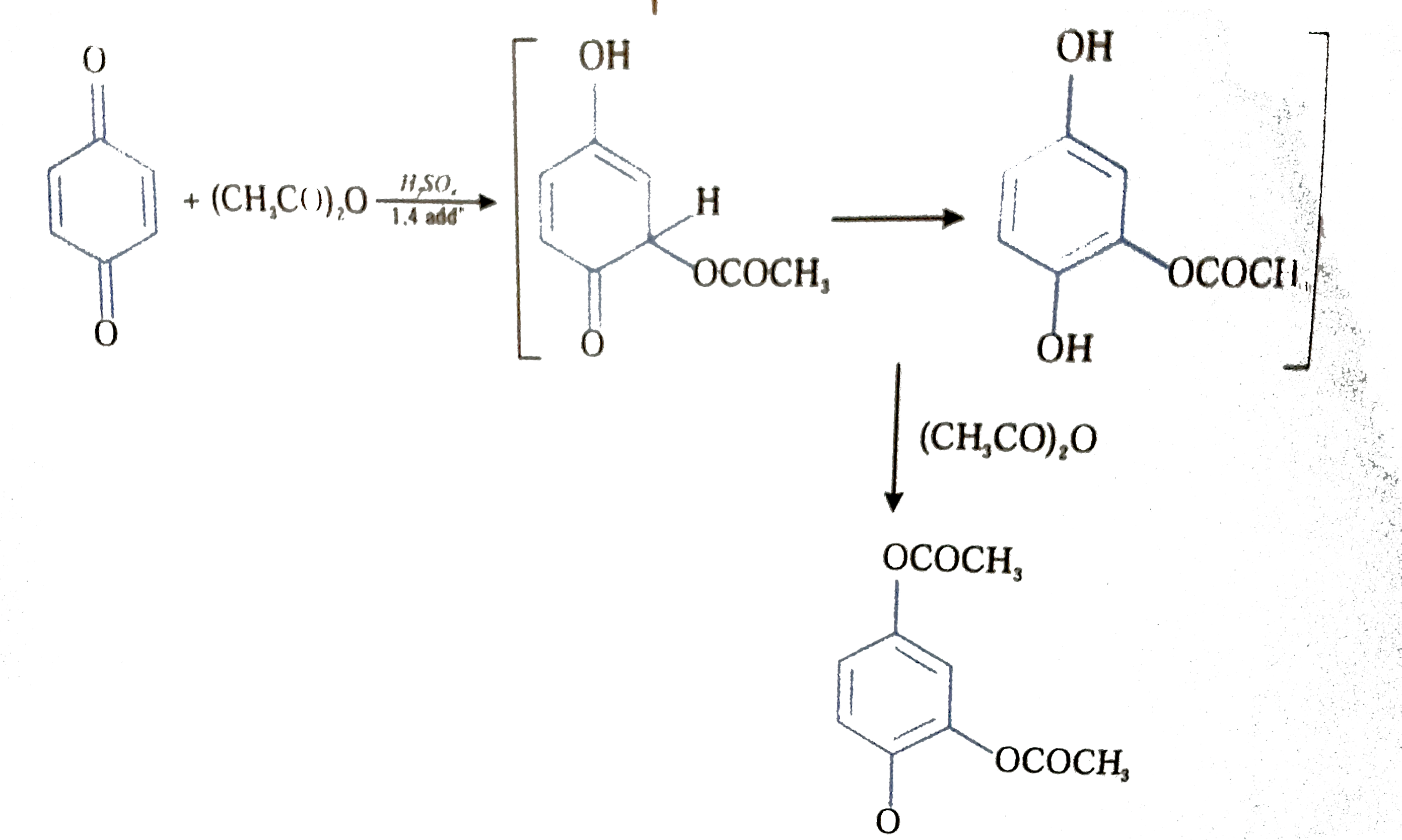
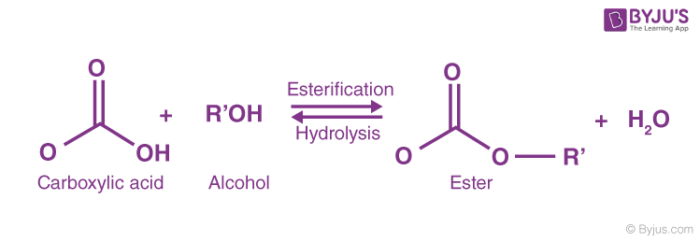
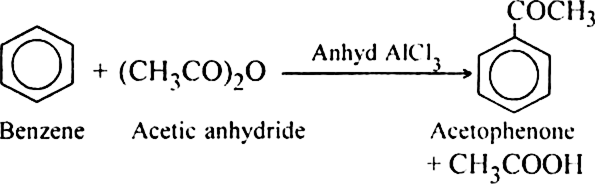acid anhydride acylation mechanism
Anhydrides are highly reactive to nucleophilic attack and undergo many of the same reactions as acid chlorides that were explored in section 21.4.
Although slower reacting than acid chlorides, anhydrides react with water to form carboxylic acids, with alcohols to form esters, and with amines to form amides.25 sept. 2022
What is the mechanism of anhydride preparation?
Anhydrides of aromatic carboxylic acids are usually prepared by rereaction of powerful acylating agents (e.g., acyl chloride) with sodium salts of the same or another carboxylic acid or by treating acyl chloride with pyridine and decomposing the reaction mixture with water.
What is the mechanism of formation of acid anhydride?
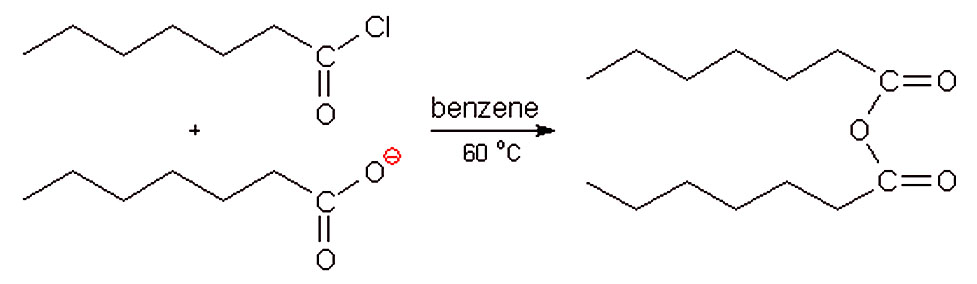
Acid anhydrides are typically prepared by nucleophilic acyl substitution reaction of an acid chloride with a carboxylate anion, as we saw in the previous section.
Both symmetrical and unsymmetrical acid anhydrides can be prepared in this way.20 sept. 2023
What is the mechanism of the reaction of acetic anhydride with alcohol?
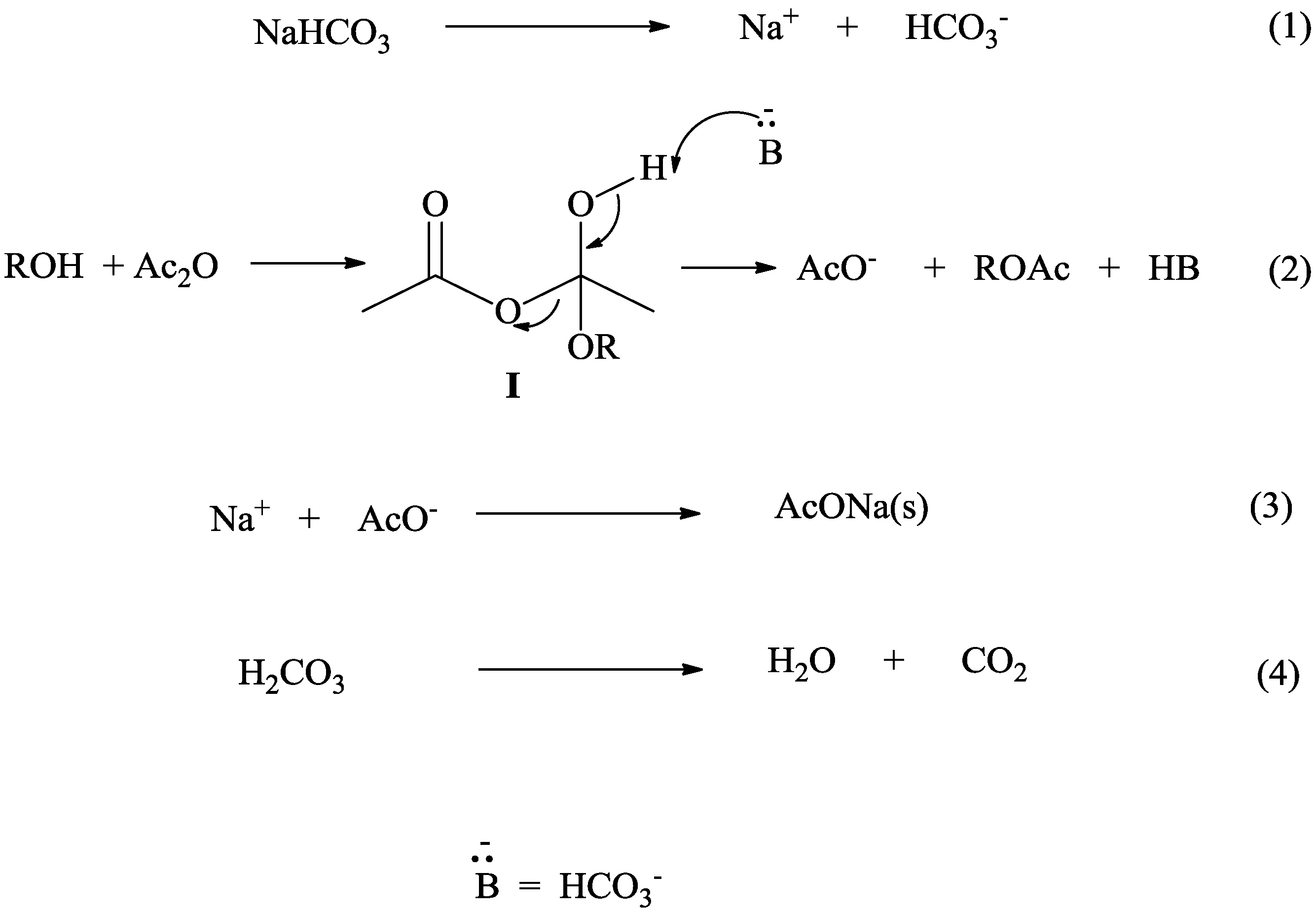
The mechanism involves four steps-nucleophilic attack by alcohol, deprotonation by pyridine, leaving group removal and protonation of carboxylate.
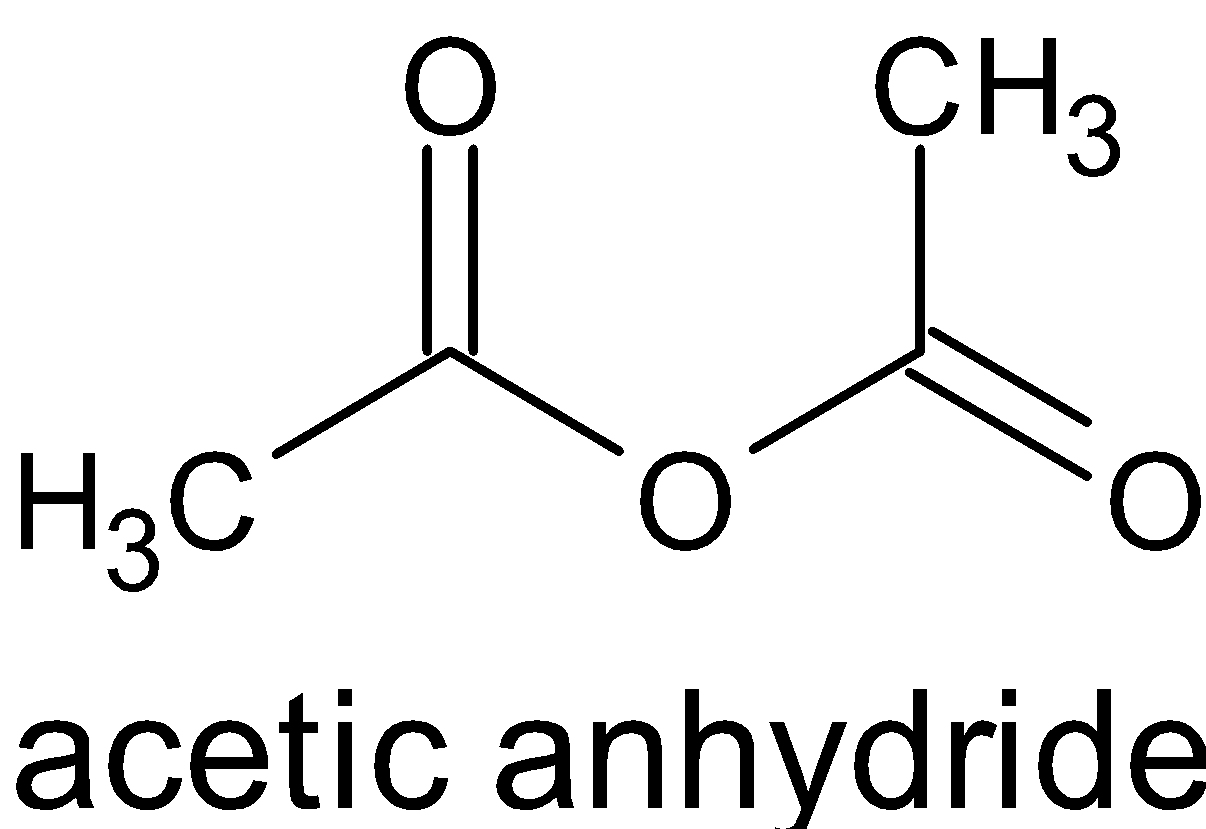
When acetic anhydride reacts with alcohol, it does not produce two molecules of ester, but an ester and ethanoic acid.
|
Facile and Efficient Acetylation of Primary Alcohols and Phenols with
03-Dec-2013 acetylation of alcohols using acetic anhydride but is toxic |
|
Kinetics of 2-Methylfuran acylation with Fatty Acid Anhydride for
Abstract. Friedel Crafts acylation using long chain fatty acid derivatives and biomass-derived furans is the key reaction to produce 'alkyl furan ketones' |
|
Mechanism of Acetylation of Ketone Enol Acetates with Acetic
tion of methyl ethyl ketone with acetic anhydride by boron trifluoride produces first the enol acetate of 3-methylpentanedione-24 (I) which is then. |
|
The Decarboxylative Acylation of Arylacetic Acids
Inthis instance acid anhydride molecules function as both the ad- dendum and the acceptor and there is presented for the reaction a mechanism |
|
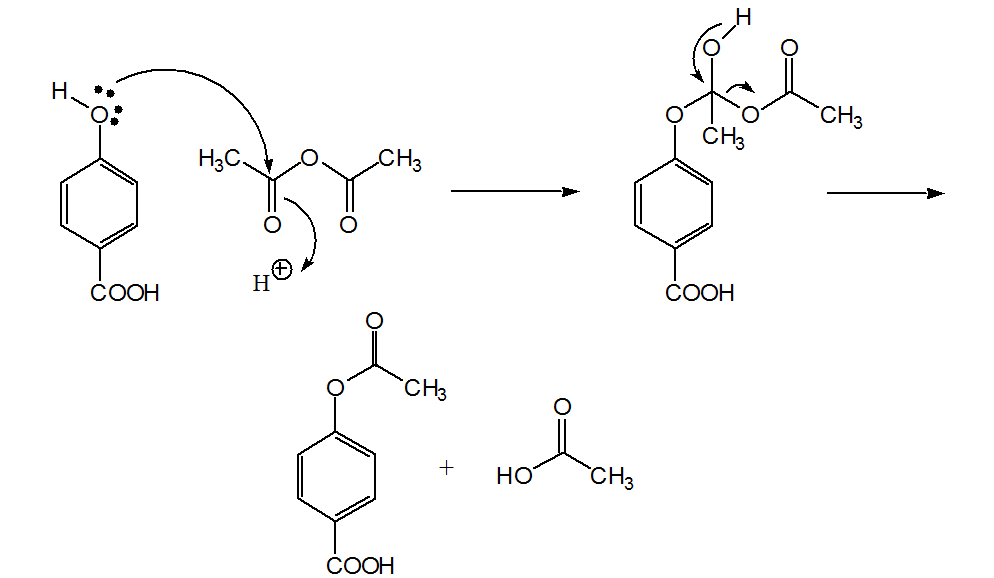
Synthesis of paracetamol by acetylation
Acetic anhydride. Tetrahedral intermediate. Ester. Acetic acid. H+. H. + H+. +OH. O. O. R-OH. Scheme SM 3.1.5.1 – General acetylation reaction mechanism of |
|
ACYLATION OF THE ENZYMATIC SITE OF δ-CHYMOTRYPSIN BY
deacylation reactions; measurements of the acylation reaction are based could also be readily acylated by acid anhydrides and acid chlorides of. |
|
The Acylation of Amides
Reaction between an acid amide and the corresponding acid anhydride leads on the one hand |
|
Esterification of hydroxy compounds by fatty acid anhydrides
procedure acetylation is brought about using acetic anhydride with perchloric acid as the catalyst. The reaction takes place rapidly at room temperature. |
|
A convenient synthesis of phosphatidylcholines: acylation of sn
erate amounts of fatty acid anhydride ( 2 mol eq. of fatty acid anhydride per mol of OH) in a At the end of the reaction the phosphatidylcholine can be. |
|
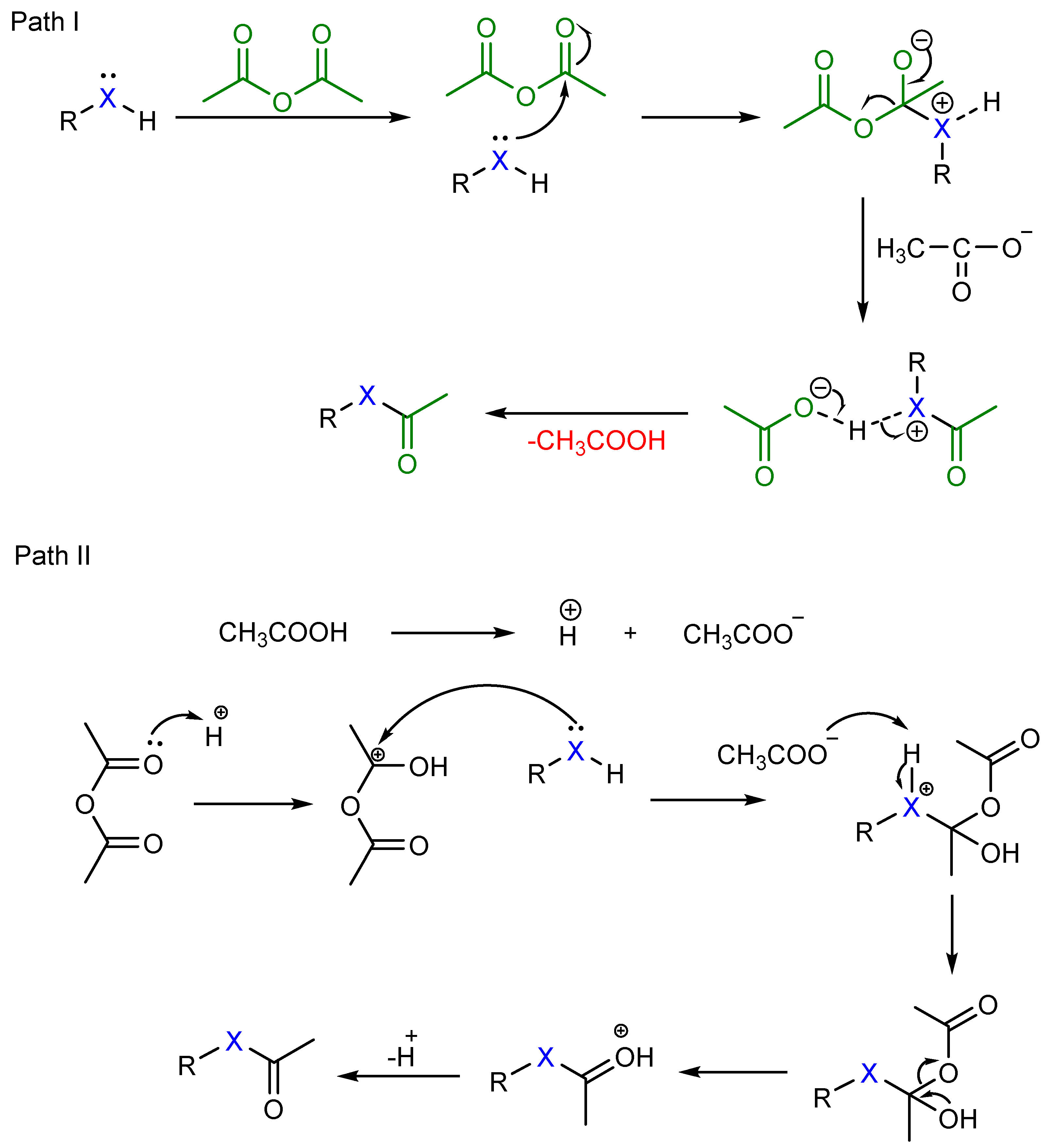
Insight into the Mechanism of the Acylation of Alcohols with Acid
15-Mar-2021 Also phosphoric acid catalyst was effective for synthetically useful esterification from carboxylic acids |
|
Kinetics and mechanism of imidazole-catalyzed acylation of - CORE
12 oct 2012 · 5 mL of a solution of acid anhydride plus imidazole in pure DMAC The final LiCl concentration was 4 , or 0 943 mol L−1 b CHM, CHD, and the |
|
Acylation by acetyl tririuoroacetate and trifluoroacetic anhydride A
The mechanism of this acid catalysis was discussed The acylation of phenols in carbon tetrachloride by acetic anhydride, trifluoroacetic anhydride and acetyl |
|
Friedel–Crafts acylation and related reactions - ResearchGate
uses acyl chlorides or acid anhydrides as acylating The acylation of anisole with acetic anhydride mechanisms of the proton generation in heteropoly |
|
Acetic Anhydride - Sigma-Aldrich
Acylation may improve the stability of a compound by protecting unstable groups, and may increase volatility Acetic anhydride can be used with a basic catalyst, such as pyridine Pyridine acts as an acceptor for the acid by-product formed in the reaction |