acid anhydride amine mechanism
|
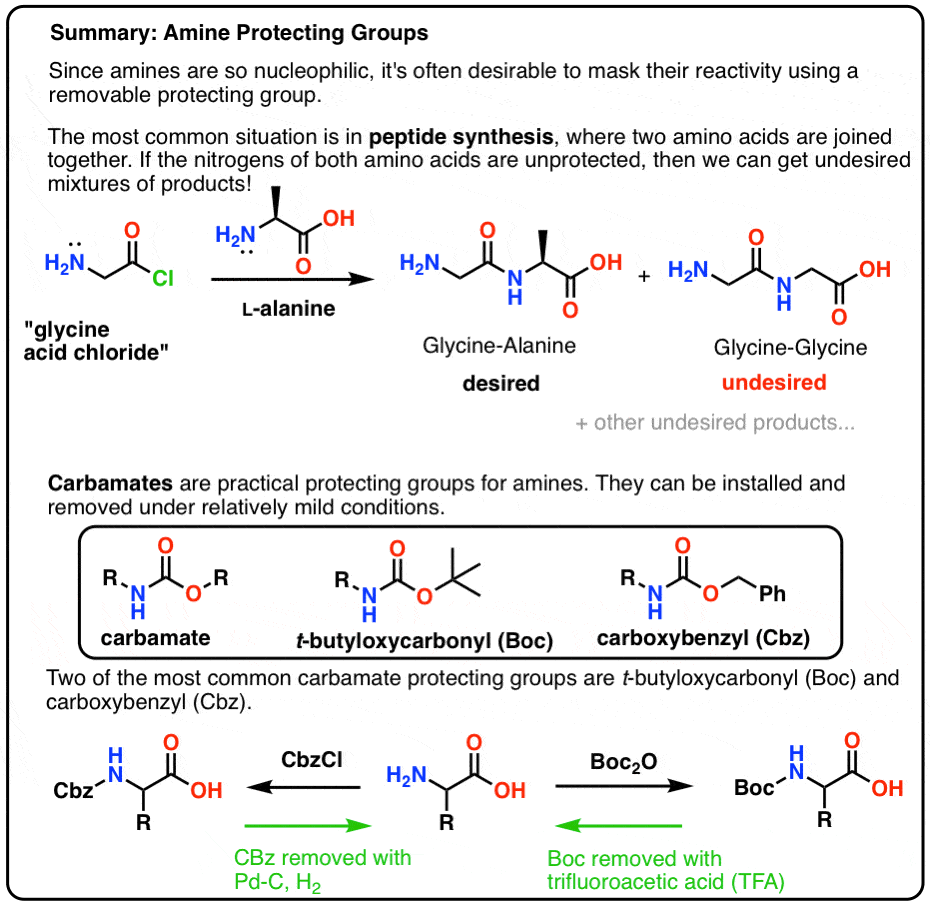
Reactions of Anhydrides
In acid the amine by-product is protonated as an ammonium ion whereas in base a neutral amine forms Reactions of Amides • The mechanism of amide hydrolysis |
Anhydrides of aromatic carboxylic acids are usually prepared by rereaction of powerful acylating agents (e.g., acyl chloride) with sodium salts of the same or another carboxylic acid or by treating acyl chloride with pyridine and decomposing the reaction mixture with water.
What is the mechanism of anhydride alcohol reaction?
Reacting an acid anhydride with an alcohol produces an ester and a carboxylic acid.
This is essentially the same reaction as with an acyl chloride (see Acyl Chlorides), but rather than releasing HCl, a carboxylic acid is formed.
How are amides formed from acid anhydrides?
Figure 1: Acid Anhydrides react with ammonia, 1o amines and 2o amines to form amides.
If ethanoic anhydride is added to concentrated ammonia solution, ethanamide is formed together with ammonium ethanoate.
Again, the reaction happens in two stages.
In the first stage, ethanamide is formed together with ethanoic acid.
|
Three Bond Technical News
In addition as in the case of other types of tertiary amine |
|
Formylation of Amines
10 jui. 2014 The amine and formic acid were heated to 80 °C until the reaction ... generated in situ from excess formic acid and acetic anhydride at ?20 ... |
|
100 Chapter 21. Carboxylic Acid Derivatives and Nucleophilic Acyl
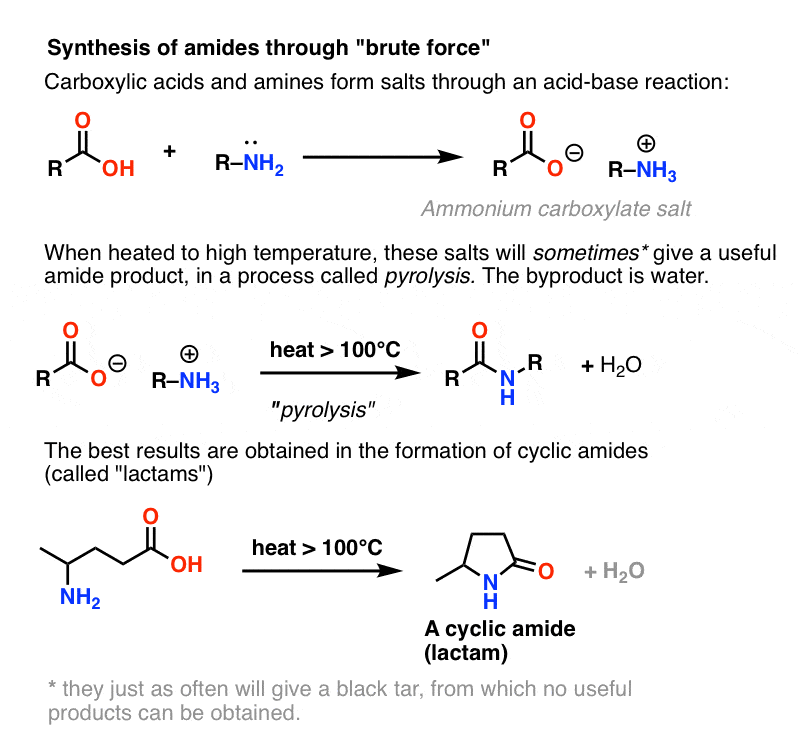
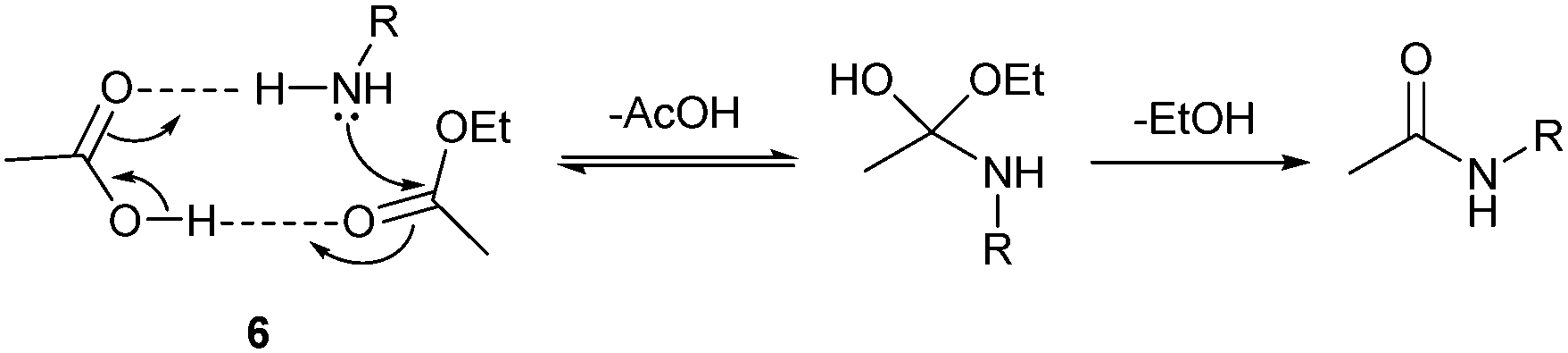
carboxylic acid ester amide acid chloride acid anhydride thioester acyl phosphate carboxylic acid and an amine is an acid-base reaction. |
|
Chapter 6 Amines and Amides
Amides are also formed when acid anhydrides react with 1° or 2° amines. N. R'. H. R. C. N. R'. |
|
Subject: Chemistry Year Group: 13 Carboxylic acid derivatives
chlorides with water alcohols |
|
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
acid anhydride. -oic anhydride. O. C. N. R. R'. R'' amide. -amide 20.3: General Mechanism for Nucleophilic Acyl Substitution. Mechanism occurs in two ... |
|
Aminolysis of acid anhydrides in water. I. Rate acceleration of
first order in both anhydride and amine neutral molecule concentrations when the sults are suggested to be consistent with mechanisms in which eitherthe ... |
|
Concerning the Paper of R. P. Mariella and K. H. Brown A Novel
describing the cleavage of some tertiary amines tions described by Mariella and Brown (1). by acetic anhydride inwhich a mechanism for the. |
|
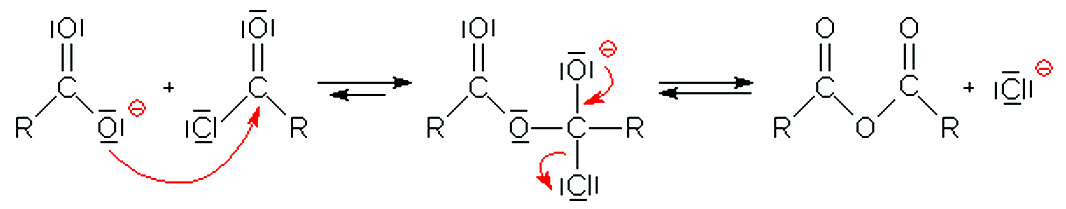
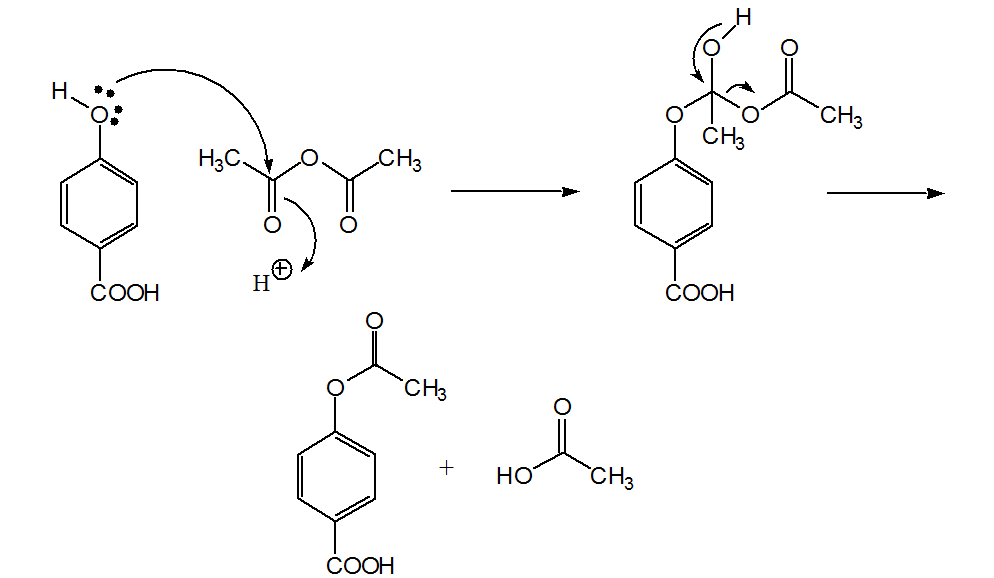
Synthesis of paracetamol by acetylation
The reaction mechanism can be devised in the following scheme for the general acetylation of an alcohol by acetic anhydride originating an ester and acetic acid |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to chlorides or acid anhydrides as starting materials. |
|
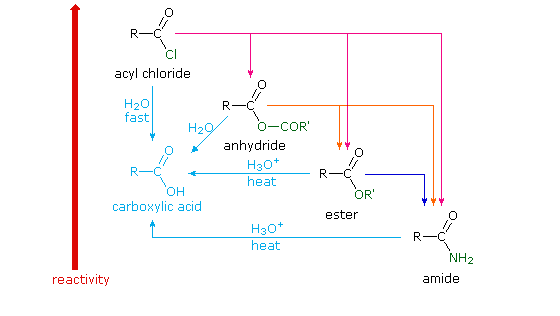
Reactions of Anhydrides Mechanism of Anhydride Substitution
Amides are hydrolyzed in acid or base to form carboxylic acids or carboxylate anions In acid, the amine by-product is protonated as an ammonium ion, whereas in base, a neutral amine forms The mechanism of amide hydrolysis in acid is the same as the mechanism of ester hydrolysis in acid |
|
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
20 3: General Mechanism for Nucleophilic Acyl Substitution amide acid anhydride ester amide ester amide 168 The reactivity of the acid derivative is related |
|
Carboxylic acid deri
O O carboxylic acid ester amide acid chloride acid anhydride thioester acyl phosphate Reaction of a carboxylic acid with thionyl chloride (SOCl2) R OH C O |
|
218 REACTIONS OF CARBOXYLIC ACID DERIVATIVES WITH
Reaction of an acid chloride with a primary amine (an amine of the form RNH2) gives a sec- Anhydrides react with nucleophiles in much the same way as acid |
|
Carboxylic Acid Derivatives Addition/Elimination
O R' OH O R'CO2H ROH ROH HNR2 HNR2 H3O+ SOCl2 HNR2 acid amide ester anhydride acid chloride H3O+ H3O+ H3O+ * important * reaction |
|
Propanephosphonic Acid Anhydride (T3P®) - A Benign Reagent for
Abstract: Propanephosphonic acid anhydride (T3P®) is a prevail- ing coupling and products of the coupling reaction are soluble in water and do not offer any difficulty then attacked by the amine 13 to result in the peptide 14 (Scheme 3) |
|
“Acyl-Transfer Reactions” I Introduction Resonance contribution by Z
5 oct 2012 · acid halide* acid anhydride ester thioester amide note: R could be "H" (2) The mechanism for the acid-catalyzed esterification [Commonly |