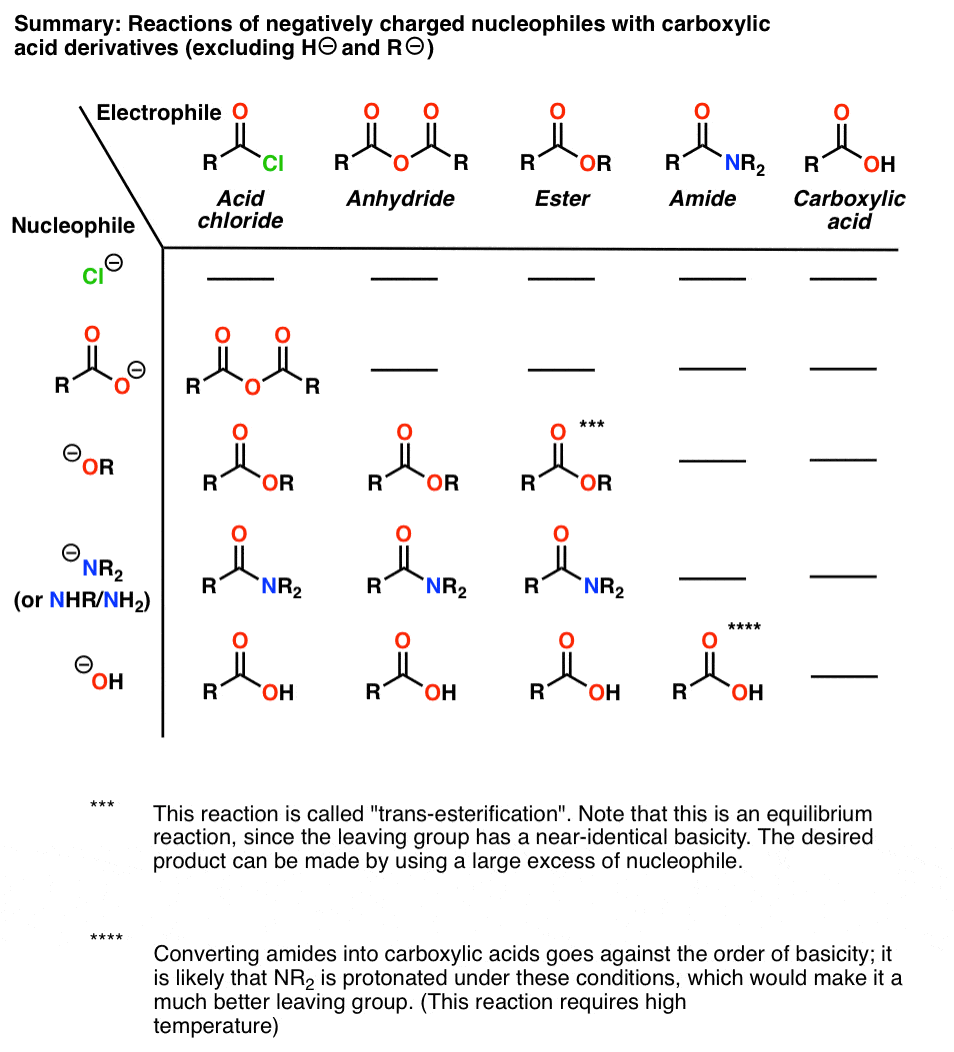
acid anhydride and alcohol conditions
How do acid anhydrides react with alcohols?
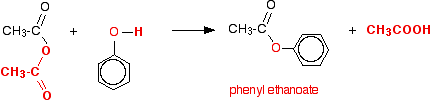
Anhydrides react with alcohols to form esters as the main product and a carboxylate as a side product.
The reaction is typically run with a base, such as NaOH or pyridine, to remove any acid produced.25 sept. 2022What are the conditions for an acid anhydride to become an ester?
Making esters using acid anhydrides
The reactions are slower than the corresponding reactions with acyl chlorides, and you usually need to warm the mixture.
In the case of a phenol, you can react the phenol with sodium hydroxide solution first, producing the more reactive phenoxide ion.What are the conditions for an acid anhydride?
Anhydrides of aromatic carboxylic acids are usually prepared by rereaction of powerful acylating agents (e.g., acyl chloride) with sodium salts of the same or another carboxylic acid or by treating acyl chloride with pyridine and decomposing the reaction mixture with water.
When primary alcohol is treated with a carboxylic acid in the presence of sulphuric acid a compound is formed.
This compound has a sweet smell.
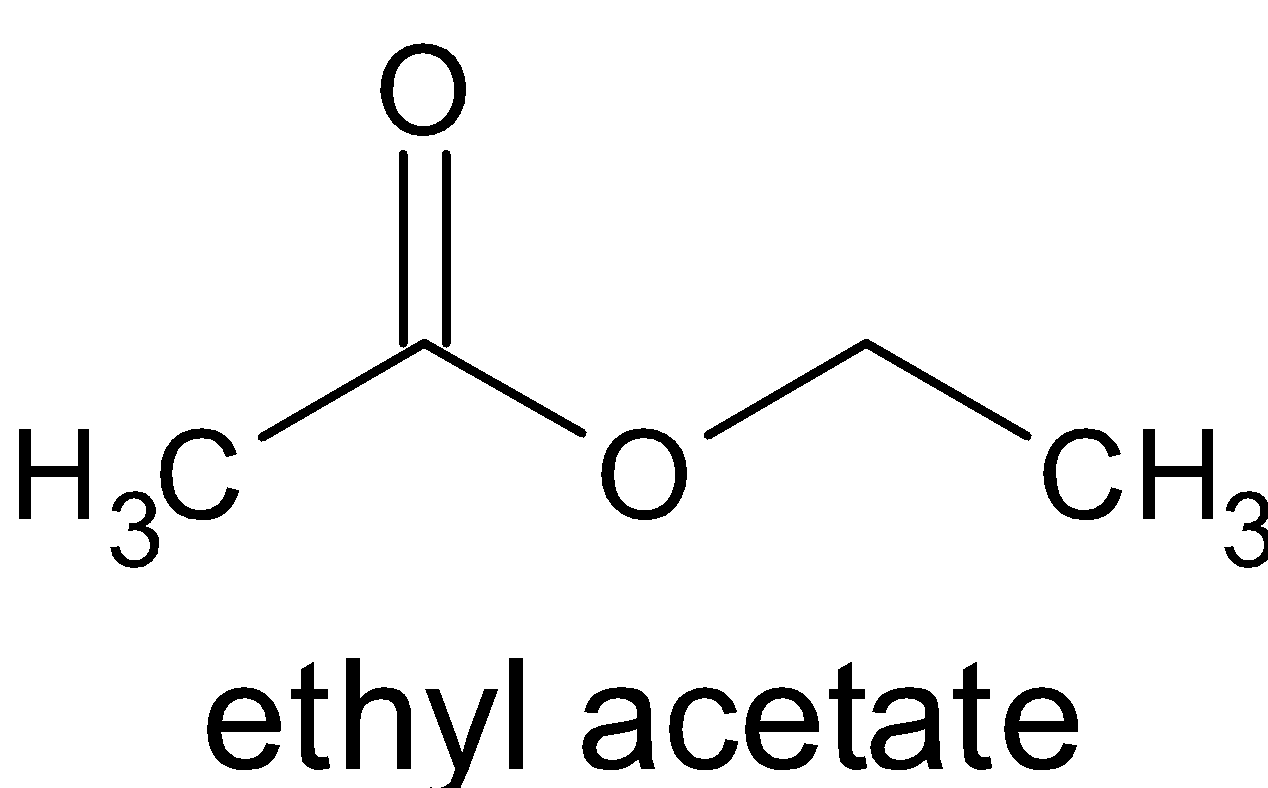
The compound obtained is called ester.
The chemical reaction occurring in the formation of the ester is known as an esterification reaction.
|
ACETIC ANHYDRIDE HAZARD SUMMARY IDENTIFICATION
Breathing Acetic Anhydride can irritate the nose throat and mouth. worsen respiratory conditions caused by chemical exposure. |
|
Dimethyl sulfoxide-acid anhydride mixtures for the oxidation of
acetic anhydride is not necessary. Successful oxidations have been carried out with 3-5 moles of anhydride per mole of alcohol. Optimal conditions for the |
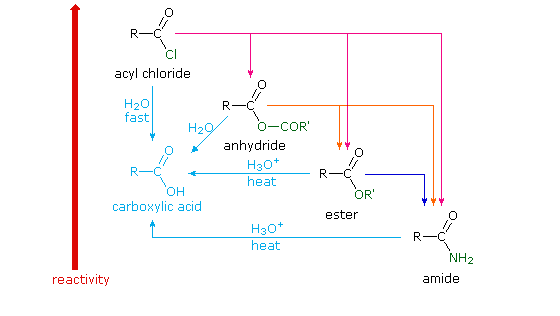
| II. CARBOXYLIC ACIDS AND DERIVATIVES One of the more |
|
Chapter 5 Carboxylic Acids and Esters
Esters may be broken apart under acidic conditions by water (a hydrolysis reaction) to form a carboxylic acid and an alcohol. • This is essentially the reverse |
|
Esterification of hydroxy compounds by fatty acid anhydrides
reaction conditions on the nature and extent of the reaction with partial glycerides has been determined. Alcohols also can be esterified by fatty acid |
|
Facile and Efficient Acetylation of Primary Alcohols and Phenols with
3 Des 2013 solvent-free conditions [34]. There have also been reports on the acylation of alcohols using acetic anhydride catalyzed by silica gel ... |
|
Widely Useful DMAP-Catalyzed Esterification under Auxiliary Base
6 Nov 2007 acid anhydrides under auxiliary base- and solvent-free conditions to give ... amounts of carboxylic acids and alcohols have been developed. |
|
Topic 13
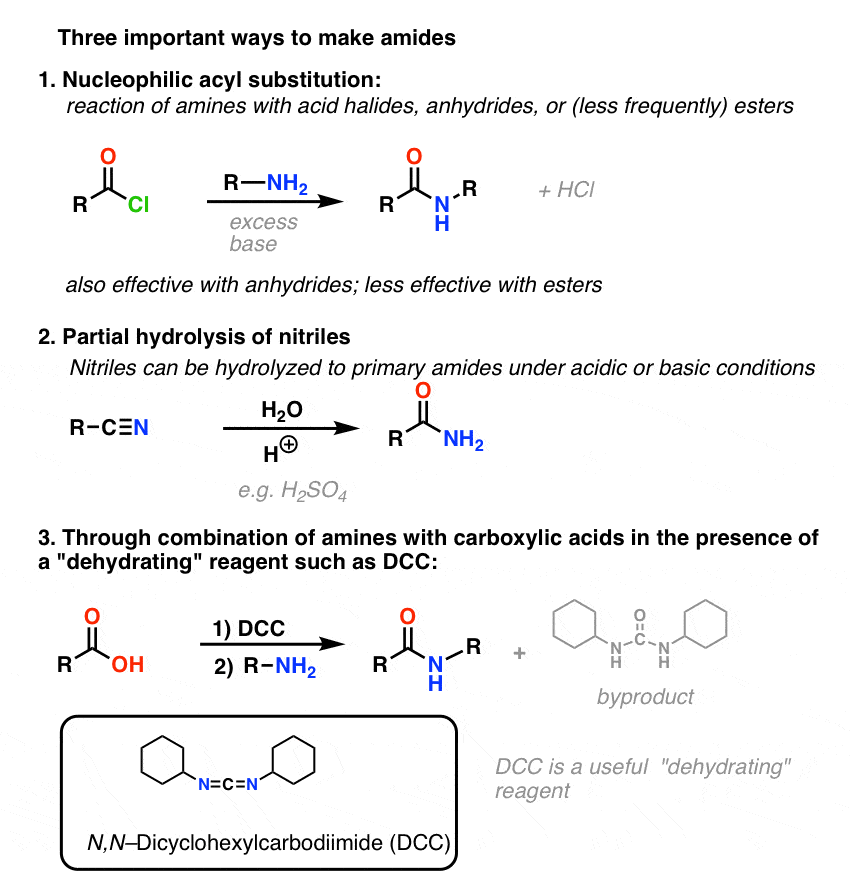
acyl chloride + alcohol ? ester conditions: room temperature equation: R1COCl + R2OH ? R1COOR2 + HCl. 31. acid anhydride + alcohol ? ester. |
|
Scandium Trifluoromethanesulfonate as an Extremely Active
great demand for acid catalysts to generate esters under mild conditions.4-6 We ... acid catalyst for acylation of alcohols with acid anhydrides or. |
|
EXPERIMENT 65 NOTES
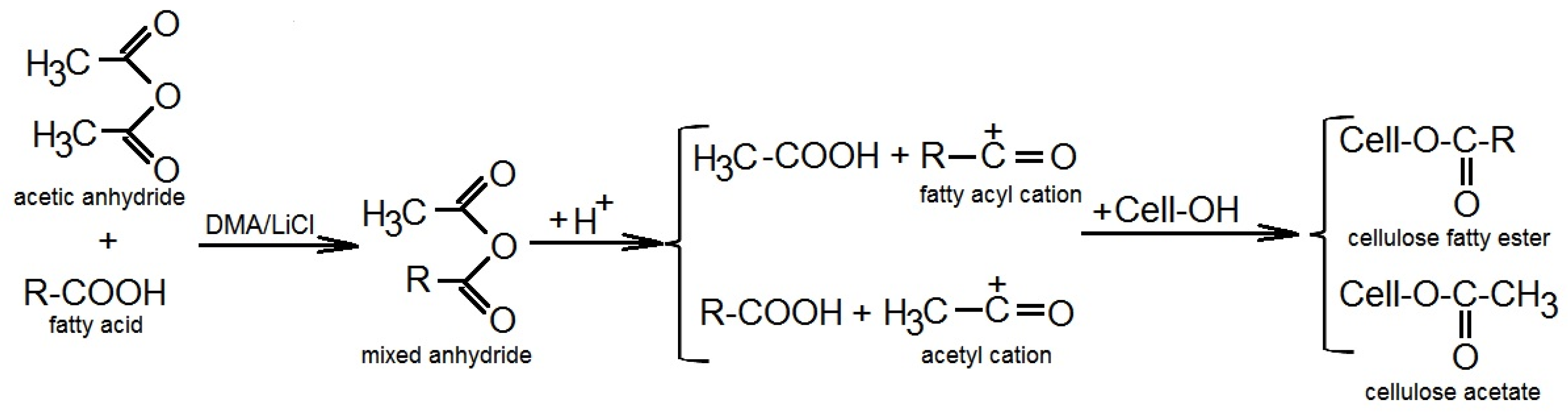
1. When an anhydride reacts with an alcohol (either under acidic or basic conditions) the anhydride plays the part of the electrophile |
|
II CARBOXYLIC ACIDS AND DERIVATIVES One of the more
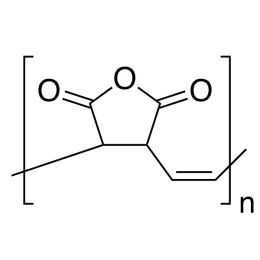
Terminal alkenes are converted to CO2 under these conditions Carboxylic acids can be reduced to primary alcohols with LiAlH4 or with BH3 followed by Simple acid anhydrides are named by replacing the ending "acid" with " anhydride" |
|
Esterification of succinic anhydride to di - CORE
the esterification of different carboxylic acids with p-cresol b Reaction conditions: p-cresol:succinic anhydride, 3:1 mol/mol; amount of catalyst, 0 5 g; reaction |
|
Reactions of Anhydrides Mechanism of Anhydride Substitution
The reaction is an equilibrium, so it is driven to the right by exploiting LeChatelier's Principle Fischer Esterification of Carboxylic Acids • The mechanism of acid- |
|
The Institute of Paper Chemistry - SMARTech
The Unsymmetrical Anhydride of Trimellitic and Acetic Anhydrides 31 Preparation of unsymmetrical anhydride with an excess of alcohol led to the opening of the conditions favoring ionization, the ester of the weaker acid will be prepared |
|
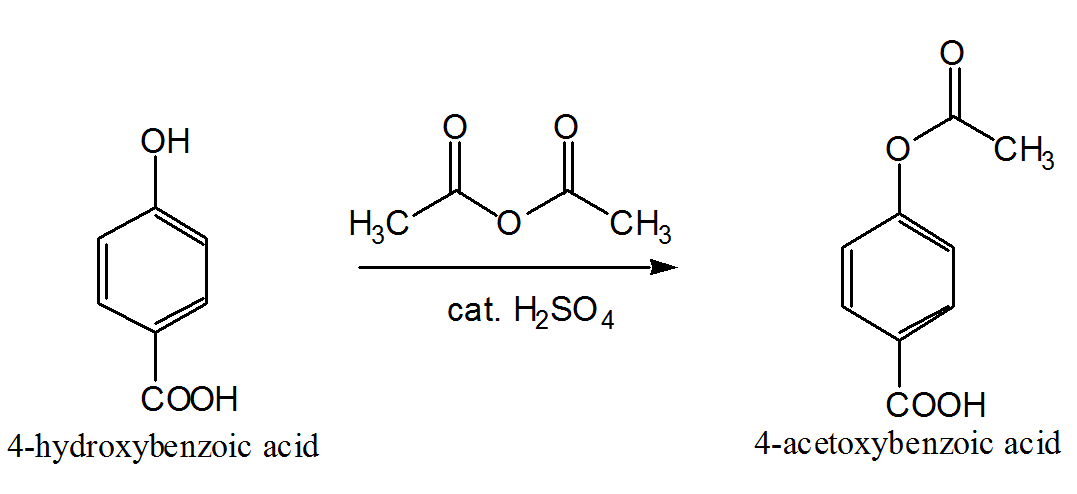
Reactions of alcohols in acetic anhydride-mineral acid mixtures
In acetylation reactions of alcohols with mixtures of an inorganic acid and acetic anhydride two general types of mechanism' are to be anticipated The first |

















![Piasticizers Made from Phthalic Acid Esters - [PDF Document] Piasticizers Made from Phthalic Acid Esters - [PDF Document]](https://ars.els-cdn.com/content/image/3-s2.0-B9780080925554500098-u05-08-9780080925554.gif)
![PDF] Facile and Efficient Acetylation of Primary Alcohols and PDF] Facile and Efficient Acetylation of Primary Alcohols and](https://media.cheggcdn.com/study/2e1/2e1f3139-9166-41ce-8bab-2653d9e93c2c/image.png)

![PDF] Acetylation of alcohols phenols and salicylic acid by PDF] Acetylation of alcohols phenols and salicylic acid by](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid\u003d7918\u0026t\u003dl)



![PDF] Acetylation of alcohols phenols and salicylic acid by PDF] Acetylation of alcohols phenols and salicylic acid by](https://www.mdpi.com/catalysts/catalysts-03-00954/article_deploy/html/images/catalysts-03-00954-g001.png)