acid anhydride hydrolysis mechanism
|
Reactions of Anhydrides
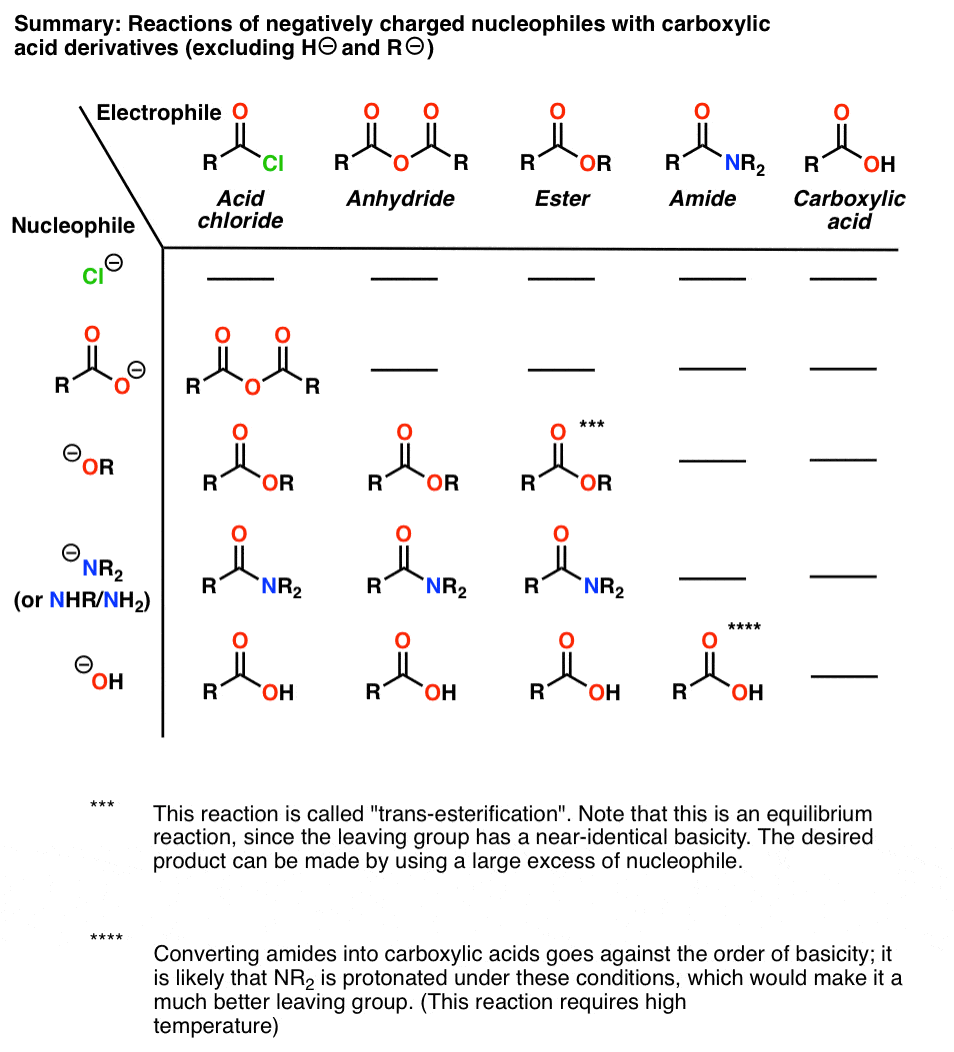
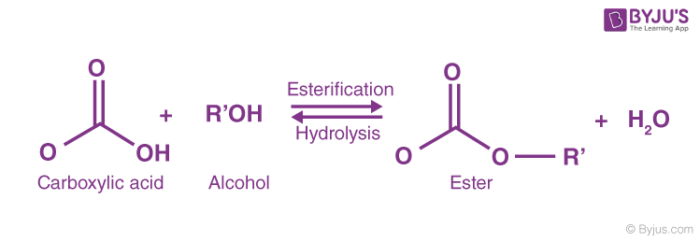
Esters are hydrolyzed with water in the presence of either acid or base to form carboxylic acids or carboxylate anions respectively Reactions of Esters • |
What does an acid anhydride on hydrolysis gives?
The correct Answer is:Two carboxylic acids.
What is the mechanism for acid anhydride with water?
Hydrolysis is the simplest answer.
Water hydrolyzes anhydrides into their corresponding carboxylic acids.
Note that when this happens, you get two carboxylic acids.
An example of this is water hydrolyzing acetic anhydride.What is the mechanism of formation of acid anhydride?
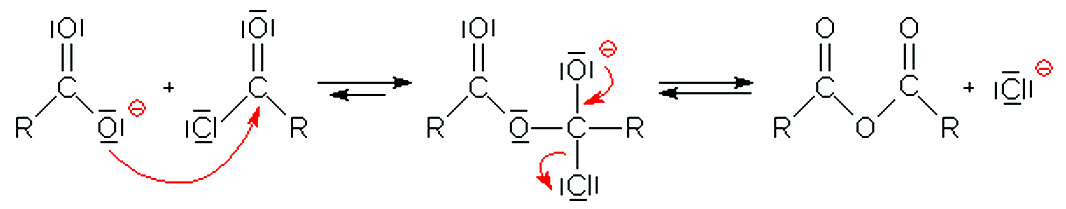
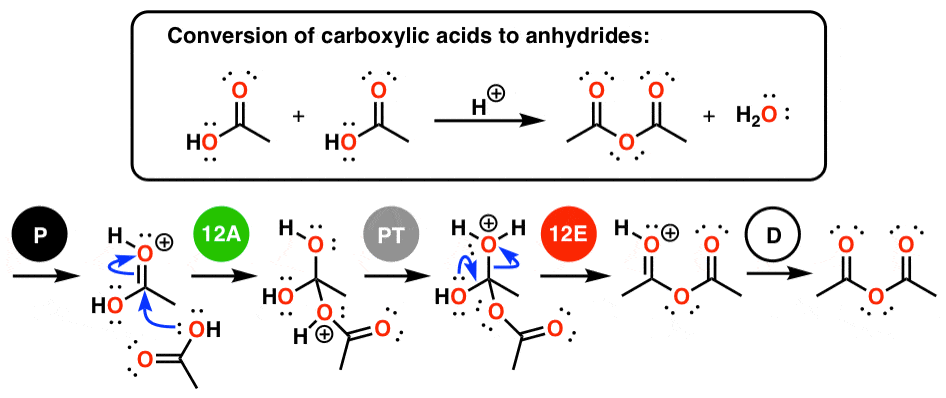
Acid anhydrides are typically prepared by nucleophilic acyl substitution reaction of an acid chloride with a carboxylate anion, as we saw in the previous section.
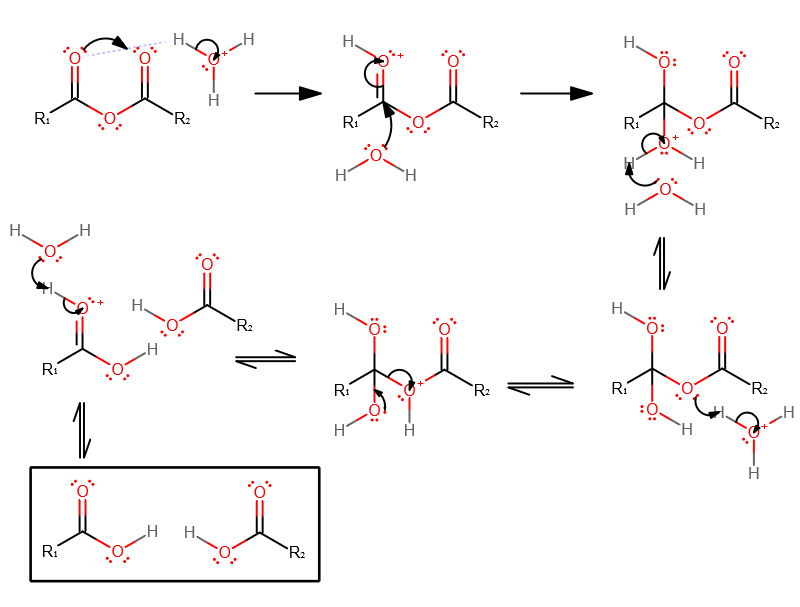
Both symmetrical and unsymmetrical acid anhydrides can be prepared in this way.It is i~sually accepted that the first step in the hydrolysis is the equilibrium transfer of a proton to the anhydride.
The second step is most likely either a unimolecular decomposition of the conjugate acid (A-1 mechanism) or a bi- molecular addition of or substitution by a water ~nolecule (A-2 mechanism).
|
PicoSpin 45: Hydrolysis of Acetic Anhydride with Heavy Water (D2O)
The hydrolysis of acetic anhydride (Ac2O) to acetic acid (AcOH) serves as a Lowry T.H.; Richardson |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: V
The acid-catalyzed hydrolysis of acetic anhydride has been studied by Kilpatrick (I) and by Gold and I-Iilton (23). It is i~sually accepted that the first step |
|
Kinetic modeling using temperature as an on-line measurement
21 déc. 2021 Kinetic model for acetic anhydride hydrolysis taking into account autocatalytic ... 4 shows the reaction mechanism by the catalyzed route. |
|
Kinetics and Activation Energy Parameters for Hydrolysis of Acetic
The hydrolysis reaction of acetic anhydride in a water/acetone cosolvent systems forming acetic is pseudo-first order. The reaction mechanism begins by acetic |
|
Very fast zinc-catalyzed hydrolysis of an anhydride. Model for the
Received August 9 1974. Very Fast Zinc-Catalyzed Hydrolysis of an. Anhydride. A Model for the Rate and Mechanism of Carboxypeptidase A Catalysis. |
|
The Hydrolysis of Carboxylic Anhydrides. V.12 The Acid Hydrolysis
The acid hydrolysis of trimethylacetic anhydride follows an A-2 mechanism in both water and aqueous di- oxane and is slower than that of acetic anhydride. |
|
Solvent Isotope Effects in Catalyzed Hydrolysis of Carboxylic Acid
The latter mechanism is discarded for carboxylic acid derivatives with good leaving groups two mechanisms; benzoic anhydride hydrolysis. |
|
The Hydrolysis of Substituted Benzoic Anhydrides1
The mechanism of anhydride hydrolysis is discussed. Although the hydrolysis of aromatic esters acid chlorides and amides has been frequently studied. |
|
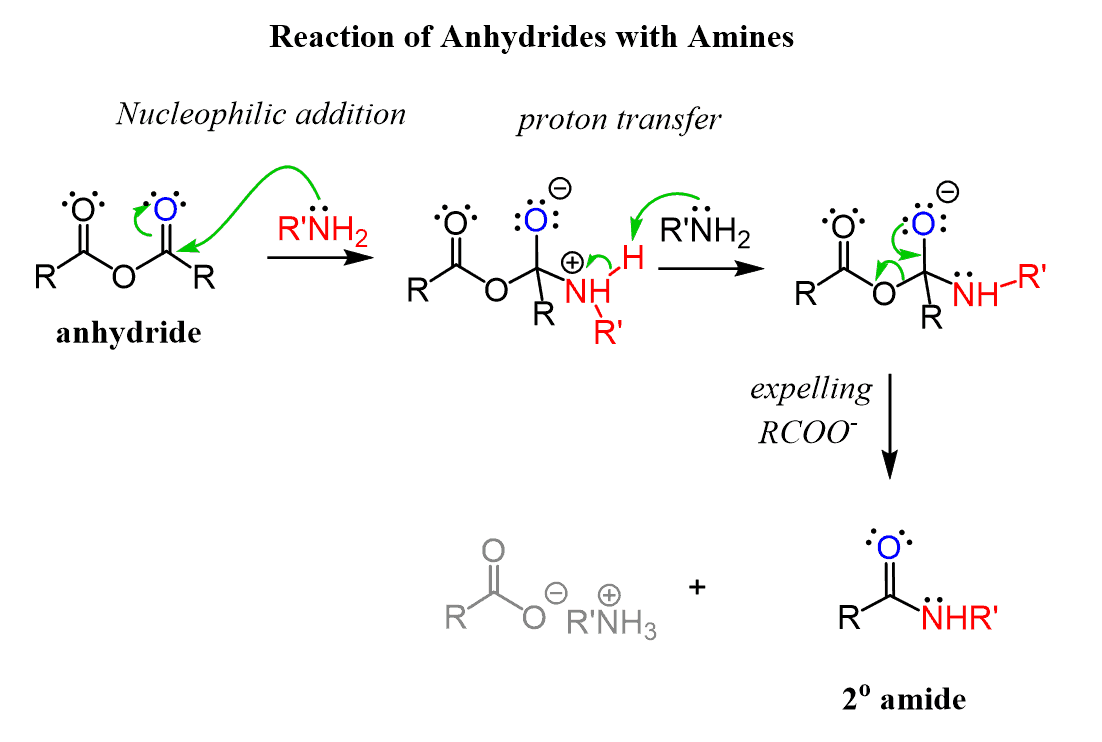
Reactions of Anhydrides Mechanism of Anhydride Substitution
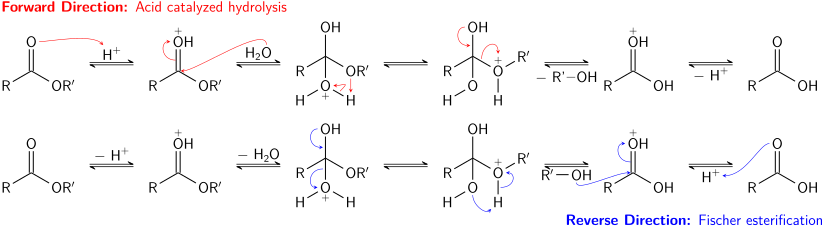
The mechanism is similar to acid-catalyzed esterification/hydrolysis. • Esters can also undergo trans-esterification reactions by reacting with. |
|
Structure–property effects in the generation of transient aqueous
of benzoic anhydrides generated from the corresponding acids and EDC The reactions can be described by a simple mechanism that merges known behavior. |
|
Reactions of Anhydrides Mechanism of Anhydride Substitution
Esters are hydrolyzed with water in the presence of either acid or base to form carboxylic acids or carboxylate anions, respectively Basic hydrolysis of an ester is |
|
Hydrolysis of Acetic Anhydride with Heavy Water - Thermo Fisher
In a hydrolysis reaction, a chemical bond is broken by the addition water The hydrolysis of acetic anhydride (Ac2O) to acetic acid (AcOH) serves as a model example of the hydrolysis reaction Acetic anhydride rapidly hydrolyzes in the presence of water, alcohol and catalyzing acid, in this case water |
|
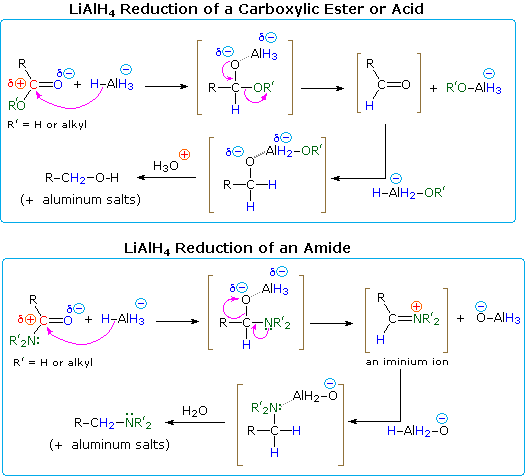
85 Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl
derivatives (acid chlorides and anhydrides) carboxylic acid amide acid chloride acid anhydride Mechanism of the base-promoted hydrolysis, Fig 20 4, p 851 |
|
Carboxylic acid deri
General mechanism: acid ester amide acid chloride acid anhydride thioester acyl phosphate acid Mechanism of the base-promoted hydrolysis (Figure 21 9) |
|
Carboxylic Acid Derivatives Addition/Elimination
The Mechanism of formation starting with an anhydride: addition/elimination again The products for anhydride hydrolysis are two carboxylic acids, the extent to |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
The mechanism of nitrile hydrolysis in acidic solution involves, first, protonation of the ni- nitriles < amides < esters, acids |
|
Carboxylic acid Derivatives
The word anhydride literally means without water, and an acid anhydride is the The basic mechanism is similar to that for hydrolysis of an ester, with hydroxide |