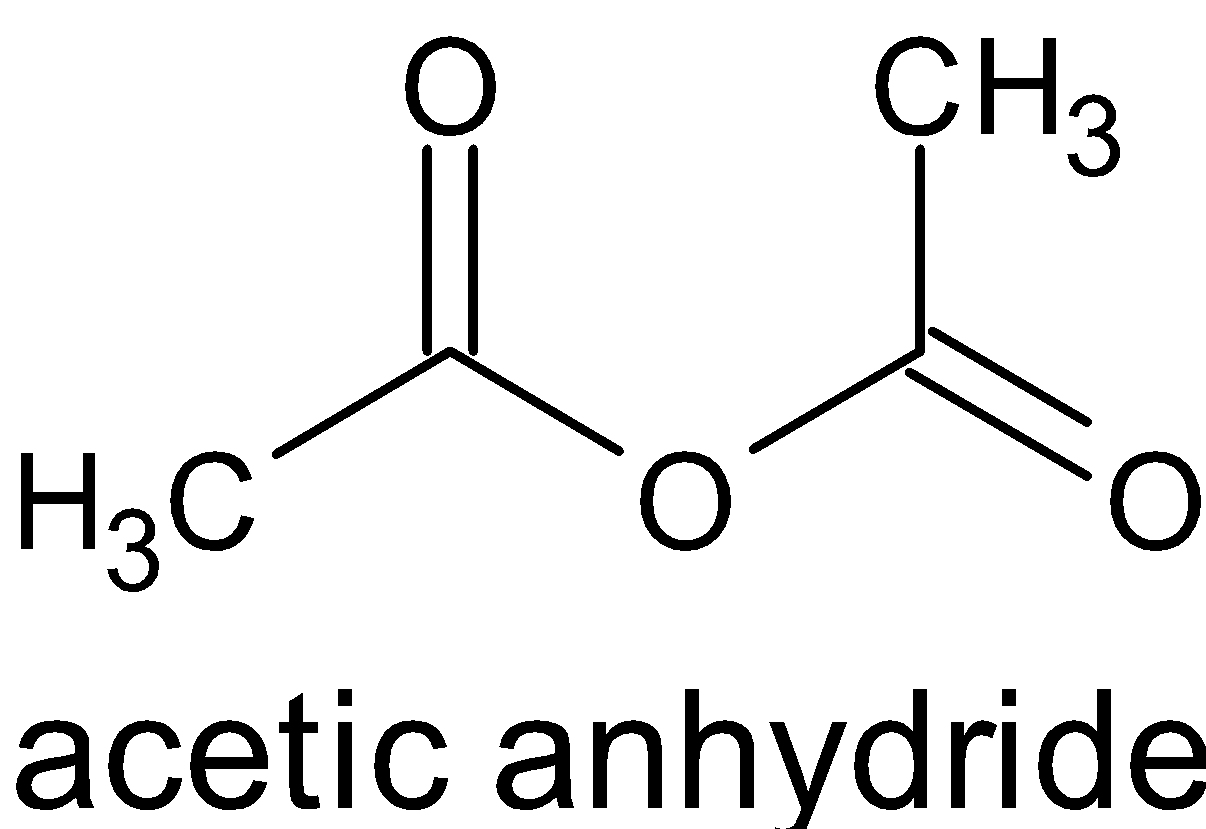
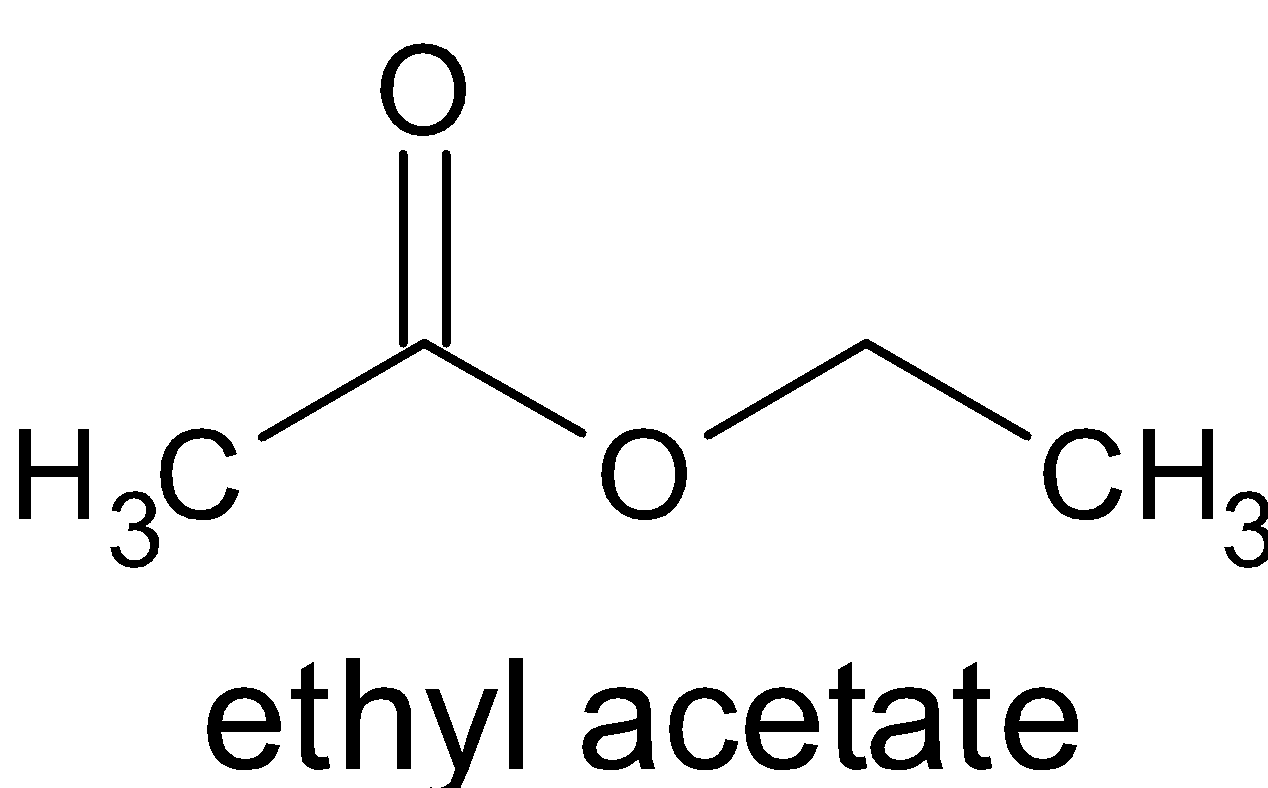
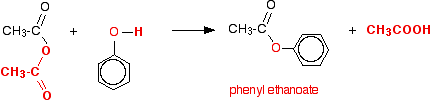
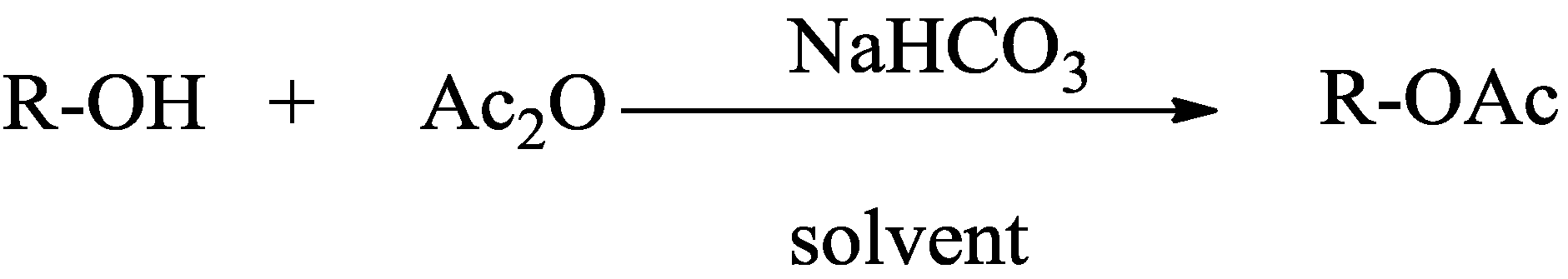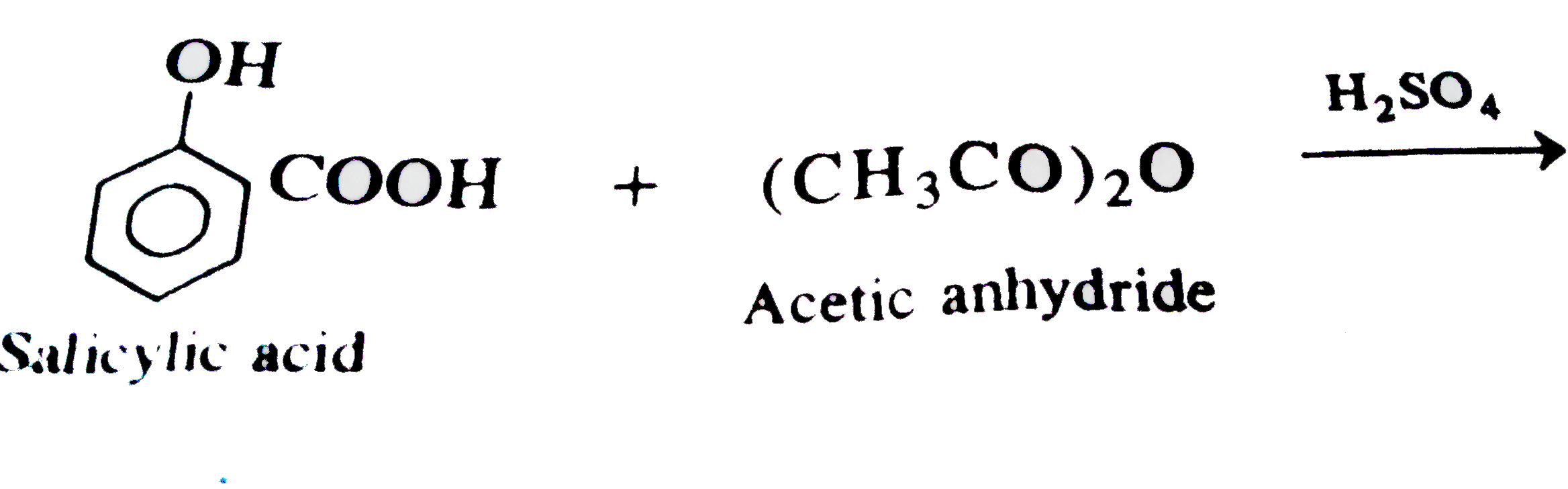acid anhydride plus alcohol
Do acid anhydrides react with alcohol?
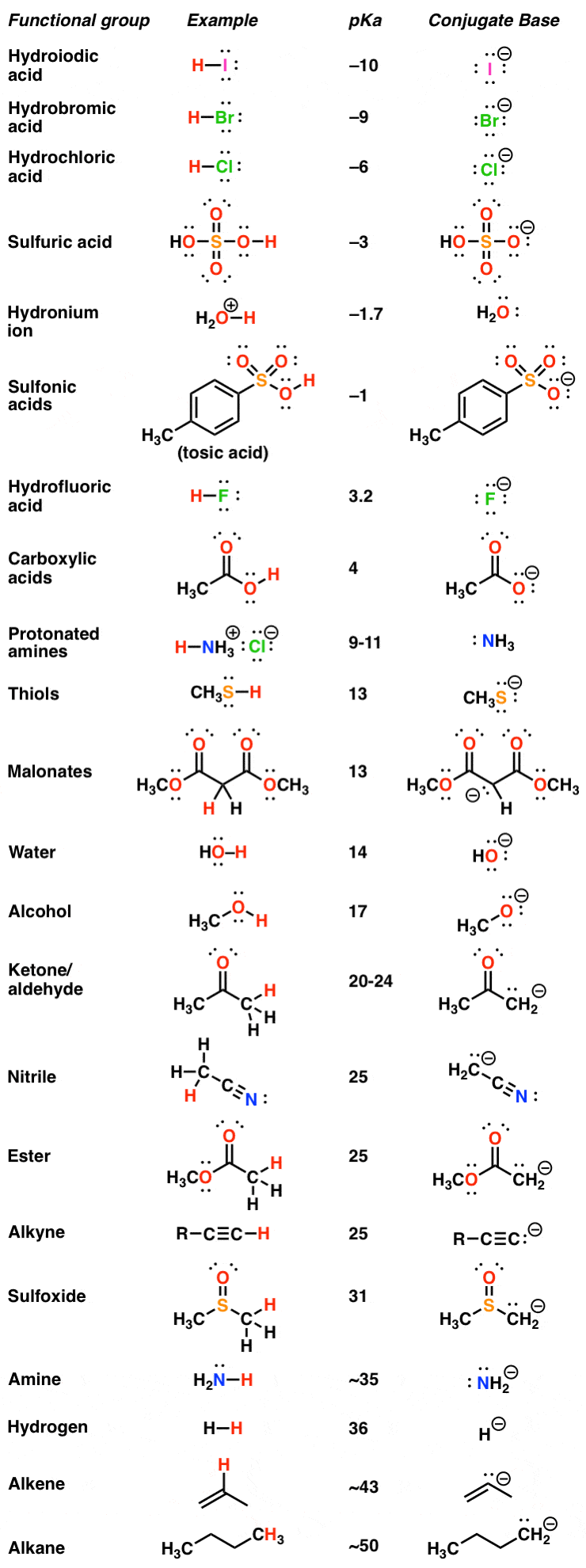
Reactions of Acid Anhydrides
Reacting an acid anhydride with an alcohol produces an ester and a carboxylic acid.
This is essentially the same reaction as with an acyl chloride (see Acyl Chlorides), but rather than releasing HCl, a carboxylic acid is formed.Remarkably, highly efficient phosphoric acid (1-3 mol %)-catalyzed acylation of alcohols with acid anhydrides was achieved and a 23 g scale synthesis of an ester was demonstrated.
Also, phosphoric acid catalyst was effective for synthetically useful esterification from carboxylic acids, alcohols, and acid anhydride.
What happens if alcohol reacts with acid?
The reaction, called Fischer esterification, is characterized by the combining of an alcohol and an acid (with acid catalysis) to yield an ester plus water.
Under appropriate conditions, inorganic acids also react with alcohols to form esters.
What happens when you add alcohol to carboxylic acid?
When primary alcohol is treated with a carboxylic acid in the presence of sulphuric acid a compound is formed.
This compound has a sweet smell.
The compound obtained is called ester.
The chemical reaction occurring in the formation of the ester is known as an esterification reaction.
|
Vat Dyes of Acenaphthene Series. IV. Condensation of
Perylenetetracarboxylic. Acid Anhydride with o-Phenylenediaminem alcohol was added to the product stirred at 70° for 30 minutes |
|
Facile and Efficient Acetylation of Primary Alcohols and Phenols with
3 déc. 2013 Reaction of an alcohol with acetic anhydride in the presence of sodium bicarbonate. 2. Results and Discussion. |
|
Dimethyl sulfoxide-acid anhydride mixtures for the oxidation of
alcohol was employed; however such a large excess of acetic anhydride is not necessary. Successful oxidations have been carried out with 3-5 moles of |
|
Esterification of hydroxy compounds by fatty acid anhydrides
AS part of this process the alcohol present as stabilizer |
|
Some Fatty Acid Derivatives of D-Glucosamine
the acid anhydrides in anhydrous methanol or with the acid chlorides in water amide with acetic anhydride at low temperature1 and in aqueous methanol ... |
|
ACETIC ANHYDRIDE HAZARD SUMMARY IDENTIFICATION
Acetic Anhydride is a colorless liquid with a characteristic sharp odor. Use dry chemical CO2 |
|
Quantitative Determination of Anhydrides of Carboxylic Acids
on the titration ofhigher fatty acid anhydrides with alcoholic sodium hydroxide little use has been made of the above reaction in an analytical. |
|
AUTO-CATALYTIC EFFECT OF ACETIC ACID ON THE KINETICS
The exothermic esterification reaction of acetic anhydride with methanol is a widely used test process for safety-oriented investigations in the field of |
|
Preparation of l-Leucyl-l-glutamic Acid Anhydride and its Behavior
/-Leucyl-Z-glutamic Acid Anhydride and Proteinases. 1103 obtained from oxidations allowed to tone plus petroleum ether then from 25 ml. of 50% ethanol. |
|
The Esterification Rate of Dibasic Acid Anhydrides with Primary
The reaction of dibasic acid anhydrides with primary alcohols at room temperature (25-27°); I phthalic anhydride in methanol; II |
|
II CARBOXYLIC ACIDS AND DERIVATIVES One of the more
rapidly reduced to give the primary alcohol Carboxylic As with acid halides, reduction of anhydrides with LiAlH4 results in the addition of two moles of hydride |
|
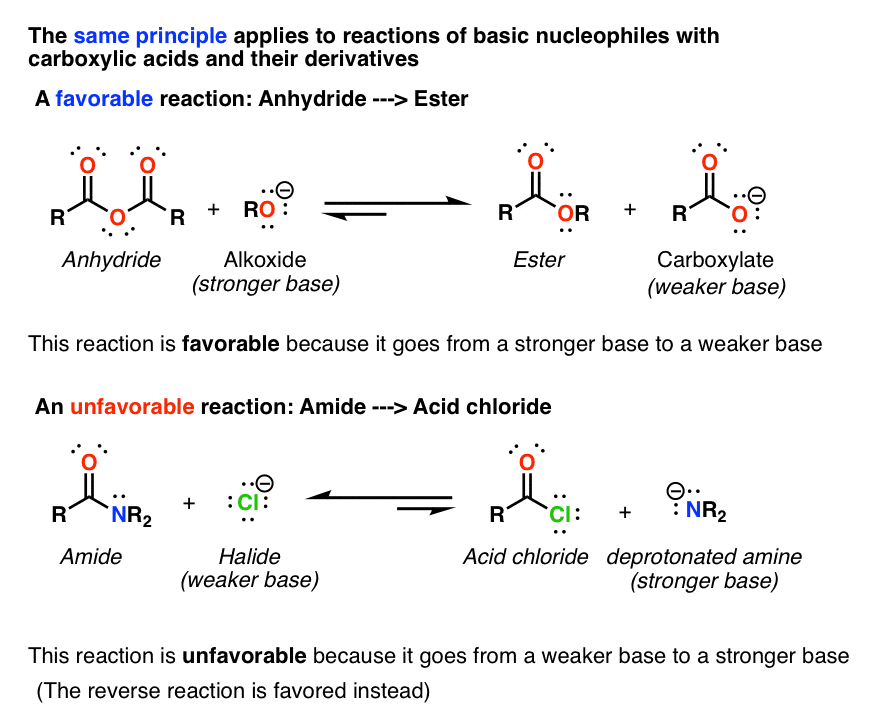
Reactions of Anhydrides Mechanism of Anhydride Substitution
Dehydration of Carboxylic Acids Treatment of a carboxylic acid with an alcohol in the presence of an acid catalyst forms an ester This reaction is called a Fischer esterification The reaction is an equilibrium, so it is driven to the right by exploiting LeChatelier's Principle |
|
Making esters from acid anhydrides - Chemistry Department - Weebly
This reaction is both slow and reversible E g ethanoic acid with methanol Write an equation for butanoic acid reacting with ethanol |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
A carboxylic acid and an alcohol react in the presence of an acid catalyst to form an ester and water as shown in equation 1 This reaction, termed Fischer |
|
The Institute of Paper Chemistry - SMARTech
An unsymmetrical anhydride of TMA and another acid could react with an alcohol to give esters of either or both carboxylic compounds depending on the nature of |
|
Carboxylic Acid Derivatives Addition/Elimination
IUPAC priority: acid > anhydride > ester > acid halide > amide > nitrile > aldehyde > ketone > alcohol > amine > alkene > alkyne > halide • Priority increases with |
|
100 Chapter 21 Carboxylic Acid Derivatives and Nucleophilic Acyl
except the word acid is replaced with anhydride H3C C O C O 3° alcohol + + H3C MgX CH3 CH3 MgXCl Grignard reagent (two equiv ) a) THF b) H3O+ |