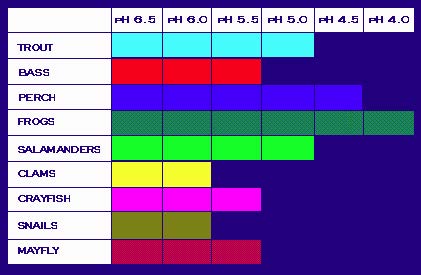
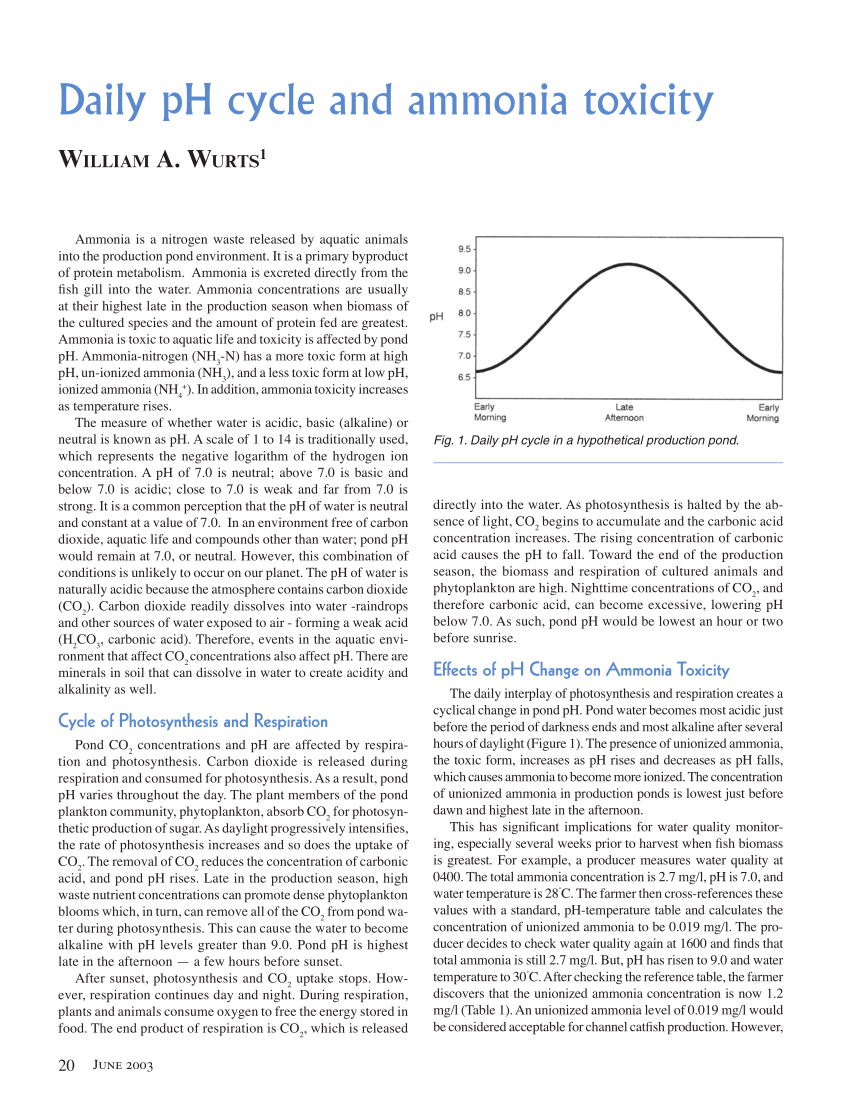
causes of acidity in drinking water
What is the cause of acidity of normal water?
The pH of pure water (H20) is 7 at 25 °C, but when exposed to the carbon dioxide in the atmosphere this equilibrium results in a pH of approximately 5.2 because CO2 in the air dissolves in the water and forms carbonic acid.
What happens if you drink acidic water everyday?
It is not recommended to drink acidic water because of the high concentration of heavy metals and potentially dangerous health effects such as tooth decay and bone loss.
Water that has a low pH can also damage your plumbing system, leaching metals into your water supply.How do you reduce the acidity of drinking water?
If your water is acidic (low pH), you can use a neutralizing filter containing calcite or ground limestone (calcium carbonate) or magnesia (magnesium oxide) to raise the pH.
Neutralizing filters must be backwashed periodically since they serve as mechanical filters to remove solid particles from the water.Stomach troubles develop more quickly from drinking acidic water.
You need to test your water if you experience symptoms like acid reflux, heartburn, and stomach pain.
Don't risk mixing stomach acid with bitter water; get your water system checked often.
|
Corrosion Control Program: Copper Pipes Protection
Metro Vancouver currently delivers water with a pH of 7.7 which is compliant with Health Canada's Guidelines for. Canadian Drinking Water Quality |
|
Mineral analysis for homeowners
At normal drinking water pH levels bicarbonate and decades) to low levels of arsenic in drinking water may cause certain types of cancer. |
|
The drop on water
This may lead to increased concentrations of these metals in drinking water which can cause health concerns. See our fact sheets on pH |
|
Commonly Found Substances in Drinking Water
At pH levels of less than 7.0 corrosion of water pipes may occur |
|
Using this fact sheet Interpreting Drinking Water Quality Results
Typical groundwater pH values in Wisconsin range from 6.5 to 8.5. Sources: Low values are most often caused by lack of carbonate minerals in the aquifer. |
|
Disinfection by-products (THMs and HAAs) - in Manitoba Water
All drinking water sources have potential to contain microorganisms such as bacteria |
|
Technical Support Document for Ontario Drinking Water Standards
parameters such as alkalinity |
|
Acid Rock Drainage in Southwest Nova Scotia
Acidic waterbodies can negatively impact potable and industrial water supplies and can cause excessive corrosion in metal and concrete infrastructure (Fox |
|
5 Chemicals in Drinking Water
chemicals found in Toronto's drinking water; advising that there are no A recent study also found that dichloroacetic acid (DCA) caused testicular. |
|
#06 Copper in Private Drinking Water Wells
Acidity or low pH is caused by natural geological conditions in the area. The optimal pH range for drinking water is 6.5 – 8.5 as established by the EPA. |
|
PH in Drinking-water - WHO World Health Organization
The pH of water is a measure of the acid–base equilibrium and, in most natural waters, is controlled by the carbon dioxide–bicarbonate–carbonate equilibrium system An increased carbon dioxide concentration will therefore lower pH, whereas a decrease will cause it to rise |
|
PH in Drinking-water - WHO World Health Organization
the effects on human health of the substances or contaminants examined in The update of pH in Drinking-water, Background document for development of |
|
PH in Drinking Water - Water Systems Council
What are the treatments for pH in drinking water? Two home treatment methods to adjust pH are acid neutralizing filters and chemical feed pump systems injecting |
|
PH of Well Water - The University of Rhode Island
What problems can low pH in well water cause? health concern, but over time, acidic well water can: Change how systems to treat substances in drinking |
|
What is Acidity
water and ozone to become sulfuric and nitric acid Section, has also provided significant funding for the treatment and clean-up of acid mine drainages |
|
Drinking Water Problems: Corrosion - Texas A&M
corrosion is not necessarily caused by water chemistry, but by for copper and lead (http://water epa gov/drink/ the water's pH, electrical conductivity, oxygen |
|
25 Physical and chemical properties 251 pH value The pH value of
Hard, alkaline waters can cause problems associated with scale formation Some hard waters may also be plumbosolvent The UK drinking water quality |
|
Drinking Water - SCDHEC
Iron and manganese cannot be easily removed from acidic water In this case, the acidity must be neutralized prior to the iron removal treatment TREATMENTS : 1 |























![Helpful Tips For Your Acid Reflux Relief - [PDF Document] Helpful Tips For Your Acid Reflux Relief - [PDF Document]](https://www.tyentusa.com/blog/wp-content/uploads/2019/02/PIN_7-Reasons-Why-Acidic-Water-Is-Bad-For-You.png)