acid dilution calculator percent
How do you dilute 10% to 6%?
A 10-6 dilution can be achieved by making three 1:100 dilutions, or six 1:10 dilutions, or a combination of 100-fold and 10-fold dilutions.
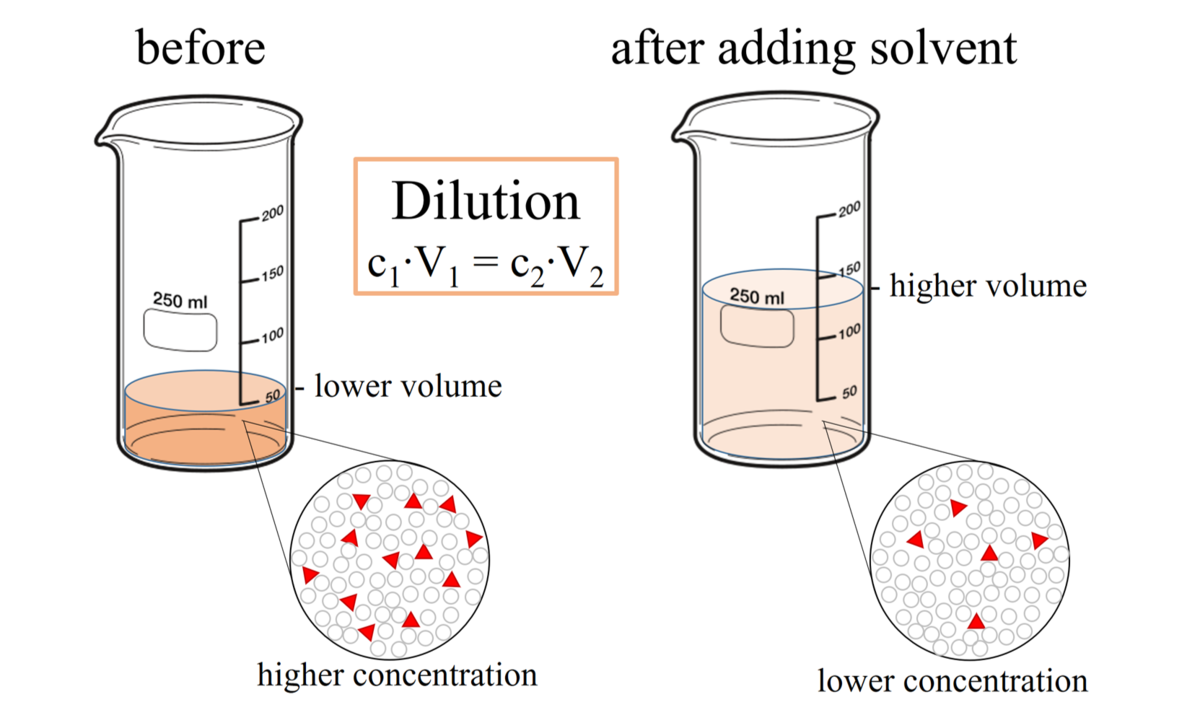
The formula for calculating a dilution is (C1) (V1) = (C2) (V2) where
The formula for calculating a dilution is (C1) (V1) = (C2) (V2) where
1C1 is the concentration of the starting solution.
2) V1 is the volume of the starting solution.
3) C2 is the concentration of the final solution.
4) V2 is the volume of the final solution.
How do you calculate 2% dilution?
You normally use the formula C1V1 = C2V2 to calculate dilutions:
1C1 is the concentrated starting stock.2) V1 is the volume of starting stock required.
3) C2 is the desired stock concentration.
4) V2 is the desired stock volume.
How do you make a 5% solution?
To make a 5% solution, take one part by weight of powder and add it to 19 parts by weight of solvent.
For example, dissolve 50 grams of sodium carbonate in 950 grams of water. 2.
In most circumstances, however, the water will not need to be weighed.
|
Laboratory Solution Preparation
Calculating Molarity from Percent Solutions Add solid to acid solution stir |
|
Citric Acid Dilution Chart
For example: a 2000 ml amount of 20 percent citric acid would be made from 436 grams (g) of dry powder citric acid and 1738 milliliters (ml) of RO water. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
However if you are asked to make a 1:3 dilution of acetic acid in ethanol |
|
TWEEN® 20 (P5927) - Product Information Sheet
pH of 1% aqueous solution: 5-7 1 sorbitol and 1 lauric acid as the primary fatty acid. Fatty acid constituents of this product are determined. |
|
Calculating Nucleic Acid or Protein Concentration Using the GloMax
7 Aug 2009 In addition buffer type |
|
Bellevue College
In this lab you will determine the percent purity of two commercially available The mass of oxalic acid |
|
Laboratory Solution Preparation
Calculating Molarity from Percent Solutions. To determine the molarity of Add solid to acid solution stir |
|
Triton™ X-100 (T8532) - Product Information Sheet
Critical micelle concentration (CMC): 0.22 to. 0.24 mM34 X-100 from solution |
|
Laboratory Math II: Solutions and Dilutions
calculate how to make any solution. media cell lysates |
|
Thrombin from human plasma (T6884) - Datasheet
The concentration of prothrombin in normal human acids. 2. Gly-Arg- |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
However, if you are asked to make a 1:3 dilution of acetic acid in ethanol, you would mix one unit below is a quick approach to calculating such dilutions where: mixed as dry mass (g) per volume where #g/100 ml = percent concentration |
|
Dilution and Concentration
Calculating the percentage or ratio strength of a solution made by diluting or con- centrating (by 30 mL of a folic acid dilution in a 100 mcg/mL concentration |
|
Citric Acid Dilution Chart
acid (CA) and reverse osmosis (RO) water required for your desired solution For example: a 2000 ml amount of 20 percent citric acid would be made from 436 |
|
Calculating Liquid Chemical Dilutions - RPC-Rabrenco
- 1 ml of purified water weighs 1 gram - 15 percent of the 1000 ml solution must be citric acid - 15 (0 15) x 1000 = 150 ml (or |
|
Concentrations and Dilutions INTRODUCTION
these calculations It is best to convert any ratio strengths to a percent We assume If you add 3 g of salicylic acid to 97 g of an ointment base, what is the final |
|
6 Percentage, Ratio Strength, and Other Expressionsppt [相容模式]
calculations of concentration for some dosage forms calculations involving percentage concentration l How many grams of aminobenzoic acid should be |
|
Laboratory Solution Preparation
Example: Prepare 100 mL of 1 0 M hydrochloric acid from Calculating Molarity from Percent Solutions Add solid to acid solution, stir, then add to water |
|
Lab Math Solutions, Dilutions, Concentrations and Molarity - APHL
25 avr 2016 · ➢Percent solutions (= parts per hundred) ➢Molar solutions However, to make 1:3 dilution of acetic acid in ethanol, Calculate molarity M= |
|
RSPT 1317 Calculating Drug Doses The Metric System The Metric
Calculating Drug Doses Percentages what is the percentage strength of a solution that 5 Prepare 1 L of 2 acetic acid using water and vinegar (5 ) |

















![92849518 Acetic Acid in Vinegar - [PDF Document] 92849518 Acetic Acid in Vinegar - [PDF Document]](https://i.ytimg.com/vi/AhZ7MKfl9hk/maxresdefault.jpg)










/volumetric-flask-containing-solution-of-potassium-dichromate--vi---k2cr2o7--and-other-flasks-of-transition-metal-salts--dry-chemicals-and-solutions--compounds-with-transition-metals-are-often-colored-702545755-5a579379eb4d520037b7a397.jpg)





