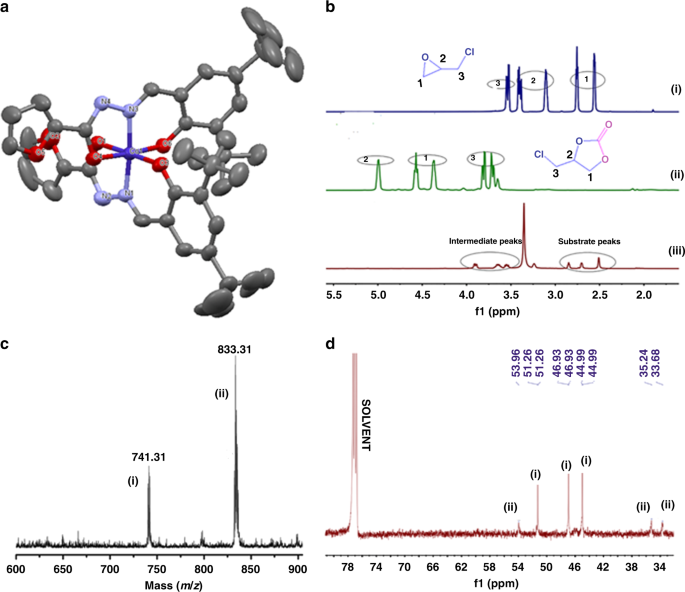
acid hydrolysis in octahedral complexes ppt
|
Ligand Displacement Reactions in Octahedral Complexes
Acid hydrolysis or aquation reactions may be defined as the reactions in which an aquo complex is formed due to the replacement of a ligand by water molecule |
What are the factors affecting acid hydrolysis in inorganic chemistry?
Factors influencing dilute acid hydrolysis include the degree of grinding of the cellulosic feedstock, liquid-to-solid ratio, reaction temperature, reaction time, acid concentrations, and co-catalyst type and concentration.
What is acid hydrolysis in octahedral complexes?
➢ Acid Hydrolysis
Acid hydrolysis or aquation reactions may be defined as the reactions in which an aquo complex is formed due to the replacement of a ligand by water molecule.
It has been observed that NH3, ammines like ethylene diamine or its derivatives coordinated to Co3+ are displaced at a very small rate.What are the mechanisms of hydrolysis of complexes?
Two types of mechanisms are followed for base hydrolysis in octahedral complexes. (i) SN2 or Associative mechanism. (ii) SN1CB or Dissociation mechanism. sulphoxide, can be explained by SN1CB mehanism but not by SN2.
|
Ligand Displacement Reactions in Octahedral Complexes- Acid
Some of the most prominent reactions in ligand substitution in six-coordinated complexes are discussed in detail. ➢ Acid Hydrolysis. Acid hydrolysis or |
|
BSc Chemistry
Acid hydrolysis of Octahedral Complex. 4. Factors affecting acid hydrolysis Base Hydrolysis of Octahedral Complexes: 7. Direct and indirect evidences in ... |
|
Substitution reactions in octahedral complexes
Whereas the acid hydrolysis is occurred for reactions of metal complexes with water at pH < 3 base hydrolysis occurs in basic solutions (at pH > 10). |
|
M.SC. INORGANIC CHEMSITRY
https://www.slideshare hydrolysis) – Acid catalyzed aquation reactions Anation reactions. Base hydrolysis |
|
M.Sc Semister I Paper I Inorganic Chemistry Department of
Acid hydrolysis reactions occur in neutral and acidic solutions. (pH<3) while observed and the acid hydrolysis of the divalent complexes like. Page 5. 5. |
|
Reaction Mechanism of Transition Metal Complexes – I
Q 2. Discuss the mechanism of acid hydrolysis taking the example of the octahedral complex of Co(III). Q.3 What do you understand by SN1CB |
|
Substitution reactions in octahedral complexes
Acid hydrolysis: The most common reaction of a metal complex is the reaction between the complex and the solvent water. The reactions of these complexes with |
|
School of sciences department of chemistry uttarakhand open
Discuss base hydrolysis of octahedral complexes in detail. 29. What the factors that influences the rate of acid hydrolysis of octahedral complexes. Discuss |
|
Acid Hydrolysis of Co(III) amine complexes
8.5.3 Examples of Substitution Reactions in octahedral complexes. Hydrolysis Reactions These are the substitution reactions in which a ligand is replaced by |
|
Stepwise and Overall Formation Constants and Their Interactions
❖ Ligand Displacement Reactions in Octahedral Complexes- Acid Hydrolysis Base Hydrolysis . ... ❖ Molecular Orbital Theory – Octahedral |
|
Ligand Displacement Reactions in Octahedral Complexes- Acid
Acid hydrolysis or aquation reactions may be defined as the reactions in which an aquo complex is formed due to the replacement of a ligand by water molecule |
|
Substitution reactions in octahedral complexes
Whereas the acid hydrolysis is occurred for reactions of metal complexes with water at pH < 3 base hydrolysis occurs in basic solutions (at pH > 10) |
|
1515586718CHE_P3_M25_etextpdf - BSc Chemistry
Acid hydrolysis of Octahedral Complex hydrolysis reactions; the water containing complex is obtained in acidic solution while the hydroxo (HO-) complex |
|
Mechanism of Hydrolysis reaction - LS College
two types (a) when an aqua complex is formed by the replacement of a ligand by H2O molecules are called acid hydrolysis or equation reactions |
|
HYDROLYSIS
Hydrolysis reactions are generally enhanced by both acids and bases and three independent reaction mechanisms account for neutral acid and base hydrolysis |
|
Ligand Substitution - CORE
2 mai 2006 · 3-14 Acid Hydrolysis and Anation of Cr(lI1) and the approximately octahedral six-coordinate complexes and the approxi- |
|
UNIT: III Thermodynamic and Kinetic Aspects of Metal Complexes
CFAE value of HS or LS octahedral complex undergoing ligand substitution reaction through is called acid hydrolysis (or aquation) of the complex |
|
Chapter 20 Coordination chemistry: reactions of complexes
12-4 Experimental Evidence in Octahedral substitution 12-5 Stereochemistry of Reactions 12-6 Substitution Reactions of Square-Planar Complexes |
|
BSc III YEAR INORGANIC CHEMISTRY-III
To predict the stability of a chemical bond using the hard-soft acid base theory covalent in nature and outer orbital octahedral complexes with sp |
|
Ligand Field Calculations without Group Theory
dependence of multiplet energies of an octahedral complex (Tanabe-Sugano The metal ions are Lewis acid catalysts in hydrolysis and |
|
Ligand Displacement Reactions in Octahedral Complexes- Acid
Acid hydrolysis or aquation reactions may be defined as the reactions in which an aquo complex is formed due to the replacement of a ligand by water molecule It |
|
Reaction Mechanism in Transition Metal Complexes
B Mechanism of Acid Hydrolysis when the inert ligand is a pi donor Hence the aquation of octahedral complexes would prefer to follow dissociative S N |
|
BSc Chemistry - e-PG Pathshala
Metal-Ligand Equilibria and Reaction Mechanism of Transition Metal Complexes ) Module No 25: Acid and Base hydrolysis, Reactions without metal ligand |
|
Kinetics of the acid hydrolysis (aquation) - CORE
Elleman, Thomas Smith, "Kinetics of the acid hydrolysis (aquation) and isotopic exchange of choride with plied this distinction to octahedral complexes and has corre lated the of the amount of pPt(NHg)Cl2(H20)3 formed during the first |
|
Ligand Substitution - CORE
2 mai 2006 · metal complexes should provide sufficient background Thus, we hope that this book 3-14 Acid Hydrolysis and Anation of Cr(lI1) and Rh(II1) |
|
Reaction Mechanisms of Inorganic Compounds C21J, 4 Lectures
The divalent first row transition metal ions (except for V2+, Cr2+ Cu2+) Aquation or acid hydrolysis larger when Y and the metal complex are of opposite |
|
Chapter 20 Coordination chemistry: reactions of complexes
12-4 Experimental Evidence in Octahedral substitution 12-5 Stereochemistry of Reactions 12-6 Substitution Reactions of Square-Planar Complexes Pt(II) is a soft acid, so soft ligand react hydrolysis of esters, amides, and peptides |
|
Substitution reactions
Reaction types of transition metal complexes The incoming ligand adds to the complex, and an intermediate with a increased Acid hydrolysis: square |
|
Coordination compounds - School Learning Resources
1 3 Factors affecting the labile/inert nature of complexes 2 2 1 Aquation (acid hydrolysis) reaction 3 Substitution reactions in square planar complexes |
|
MSc Chemistry 2015_16
Reaction Mechanism of Transition Metal Complexes: Energy profile of a kinetics of octahedral substitution, acid hydrolysis, factors affecting acid hydrolysis |