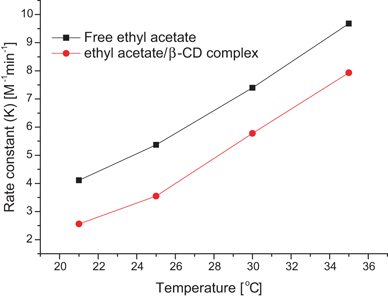
acid hydrolysis of ethyl acetate is which order
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
The half-life of a second-order reaction which depends on one second-order reactant is: 1 Transfer 50 ml of the solution of ethyl acetate (c = 0 04 mol dm-3) |
|
A Comparative Study of Alkaline Hydrolysis of Ethyl Acetate Using
ABSTRACT: Alkaline hydrolysis of ethyl acetate is essentially an irreversible and second order reaction Industrial importance of the reaction product |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
ethanol as the product from which second order rate constant can be calculated: CH3COOC2H5 + NaOH CH3COONa + C2H5OH Ethyl acetate sodium hydroxide sodium |
This reaction is a second order reaction because the rate of reaction is directly proportional to the both of the reactants.
This is called saponification of Ethyl acetate.
Formula used: \\[k = \\dfrac{1}{t}\\dfrac{{\\left( {{V_0} - {V_t}} \\right)}}{{{V_0}{V_t}}}\\]; k=rate constant for saponification of Ethyl acetate.
Why is hydrolysis of ethyl acetate pseudo first order?
As the [water] does not change significantly if it is in vast excess, the rate of reaction in this case is said to be 'pseudo first order' with respect to the [ethylacetate].
What is the order of hydrolysis of ester?
Assertion : Hydrolysis of ester in acidic medium follows first order kinetics Reason: Hydrolysis ofester is independent of the concentration of acid used.
CHEMISTRY.
What is the order of reaction of hydrolysis of ethyl acetate?
Assertion :Hydrolysis of ethyl acetate in presence of acid is a reaction of first order whereas in presence of alkali, it is a reaction of second order.
Reason: Acid acts as catalyst only whereas alkali acts as one of the reactant.
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
The effect is the second-order reaction of the hydrolysis of acetate (Kapoor. 2004). As a result of the reaction |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
concentration of reaction but depend upon order of reaction. Hydrolysis of an ester such as acetate in presence of a mineral acid: Hydrolysis is a chemical |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
The half-life of a second-order reaction which depends on one second-order reactant |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VII
For comparison the second-order rate constant for the hydrolysis of methyl acetate Acid-catalyzed hydrolysis of ethyl acetate in aqueous solution p/bar. loG ... |
|
Hydrolysis of ethyl acetate in hydrochloric acid solutions containing
Hydrolysis of ethyl acetate in 0.05 N-HCl at 25". 30" 35" |
|
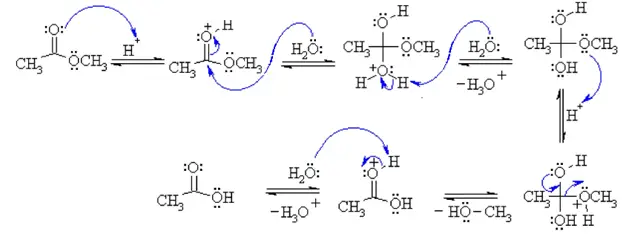
The Mechanisms of Acid Catalyzed Ester Hydrolysis Esterification
son8 hasshown that the hydrolysis of ethyl acetate is catalyzed by acetic acid acid catalyzed and first order in alcohol or water. (8) Berthelot and Pean ... |
|
Experiment 5
To determine the rate constant for the acid-catalyzed hydrolysis of methyl acetate. In the presence of an acid this reaction should be of second order |
|
Nucleophilic DisplacementReactions at the Thiol - Ester Bond. IV
in CHCls and in CHCls-acetic acid solutions in order to determine if theory of ester hydrolysis in acidic media.26. Further assuming a of —7.2 for the ... |
|
The Acid Catalyzed Hydrolysis of Ethyl Esters of Aliphatic Acids
saponificationand acid hydrolysis. Thus the change from acetate to isobutyrate has relatively little effect when compared to that be- tween isobutyrate |
|
Determination of rate of reaction and rate constant of the hydrolysis
In this hydrolysis of ethyl acetate with sodium hydroxide hydrochloric acid was used as catalyst to accelerate the reaction mixture. 1ml and 2ml of ethyl |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
concentration of reaction but depend upon order of reaction. Hydrolysis of an ester such as acetate in presence of a mineral acid: Hydrolysis is a. |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
which order of the reaction is the rate constant at different temperatures and also |
|
Electrolyte effects upon acid-catalyzed ester hydrolyses
of ethyl acetate. Consistently perchlorates have a greater salt effect than chlorides upon the acid hydrolysis of. /-butyl acetate |
|
The Mechanisms of Acid Catalyzed Ester Hydrolysis Esterification
son8 hasshown that the hydrolysis of ethyl acetate is catalyzed by acetic acid molecules chloroacetic benzoic acid and water is first order in hydro-. |
|
Acid-catalyzed Hydrolysis of Ethyl Acetate
water however |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Chemical reactions reaction rate. Chemical kinetics is the part of physical chemistry that studies reaction rates. The reaction. |
|
The Alkaline Hydrolysis of Ethyl Acetate from the Standpoint of Ion
hydrolysis of ethylene acetal by perchloric acid3 moles of sodiumhydroxide per mole of ethyl acetate re- ... strictly second order. |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VII
ethyl and t-butyl acetates in dilute aqueous acid and of ethyl acetate in the second-order rate constant for the hydrolysis of methyl acetate. |
|
Kinetics of the Hydrolysis of Ethyl Thiolacetate in Aqueous Acetone1
ethyl thiolacetate at ordinary temperatures was undertaken in order to compare thebehavior of The acid catalyzed hydrolysis of ethyl thiol- acetate at ... |
|
Ethyl trichloroacetate hydrolysis. I. Kinetic evidence for a common
Common Tetrahedral Intermediate in the Acid-Catalyzed and Water-Catalyzed Hydrolyses Pseudo-first-order rate constants for ester hydrolysis at 25.0°. |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide), HCl was used as catalyst to concentration of reaction but depend upon order of reaction the presence of a mineral acid gives acetic acid and ethyl alcohol |
|
IV SEMMESTER
To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE: This reaction follows pseudo first order kinetics |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
The exponents a and b are called reaction orders and depend on the reaction Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl 3 Pipette VHCl= 5 ml solution of hydrochloric acid (c = 0 04 mol dm -3 ) |
|
KINETICS OF HYDROLYSIS OF ETHYL ACETATE
hydrolysis of ethyl acetate which can be represented by the chemical equation: From these data, the order of the reaction, the rate constant and the enthalpy of See also the experiment, "Dissociation of Acids" in the Chem 366 lab |
|
Mechanism of the hydrolysis of ethyl acetate in aqueous solutions of
ionization of ethyl acetate (EA) have been investigated in the greatest detail in solutions of H2SO 4 ta mod- erately concentrated acid, the effective rate constant |
|
Hydrolysis of Methyl Acetate
A first-order reaction is one in which the rate of reaction is found by experiment to be produces one molecule of acetic acid, and the increase in acidity is a |













![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://i1.rgstatic.net/publication/303686508_Effect_of_Operating_Conditions_on_CSTR_performance_an_Experimental_Study/links/574d7ea208ae82d2c6bdc28a/largepreview.png)

















![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://www.mdpi.com/molecules/molecules-12-02396/article_deploy/html/images/molecules-12-02396-g002.png)