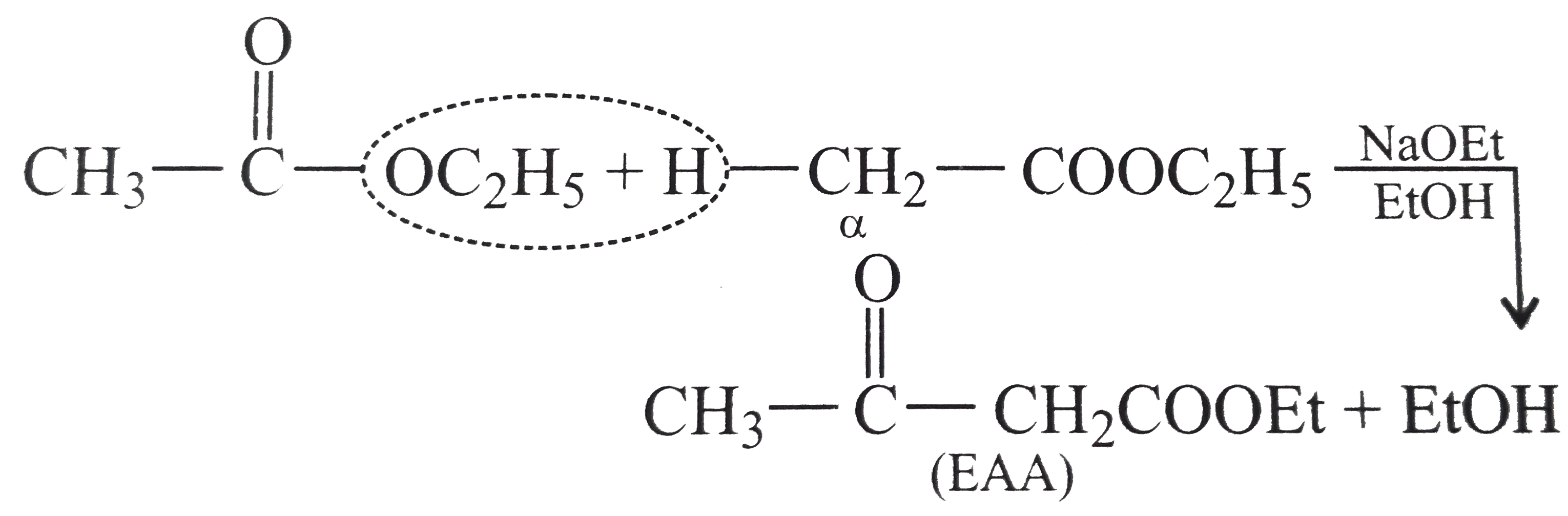
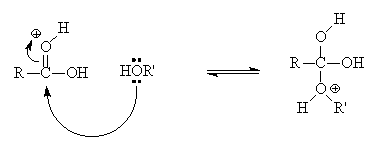
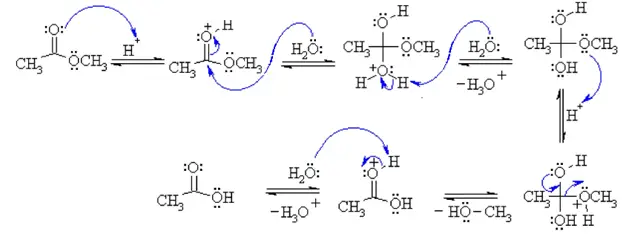
acid hydrolysis of ethyl ethanoate
What happens in acid hydrolysis?
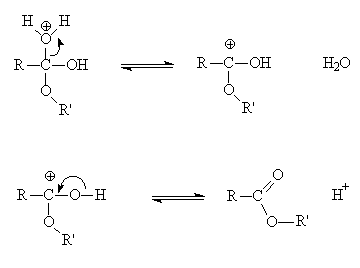
Acid hydrolysis is a hydrolysis mechanism in organic chemistry in which protic acid is used to catalyse the cleavage of a chemical bond by means of a nucleophilic replacement reaction with the addition of water elements (H2O).
In the conversion of cellulose or starch to glucose, for instance.What is the acid hydrolysis of an ester reaction?
The hydrolysis of ester takes place in an acidic or basic medium.
In an acidic medium, ester interacts with the water molecule to give a carboxylic acid and alcohol.
In contrast, ester interacts with the water molecule to give a carboxylate salt and alcohol in a basic medium.What is the acid hydrolysis of ethyl acetate?
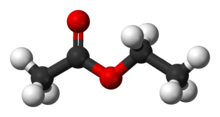
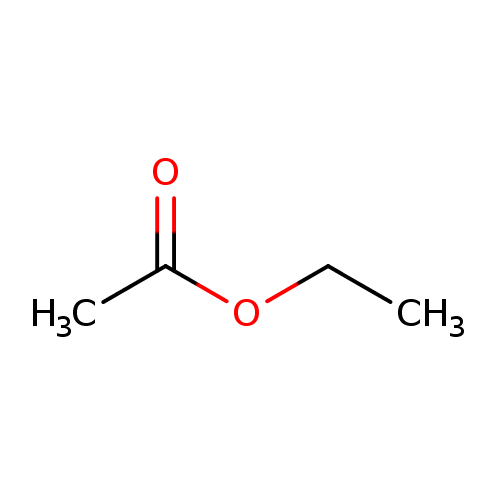
The hydrolysis of an ester such as ethyl acetate in the presence of a mineral acid gives acetic acid and ethyl alcohol.
Therefore, the kinetics of the reaction can be studied by taking a known quantity of ethyl acetate and mixing it with a relatively large quantity of HCl.The methyl ethanoate is an example of an ester compound.
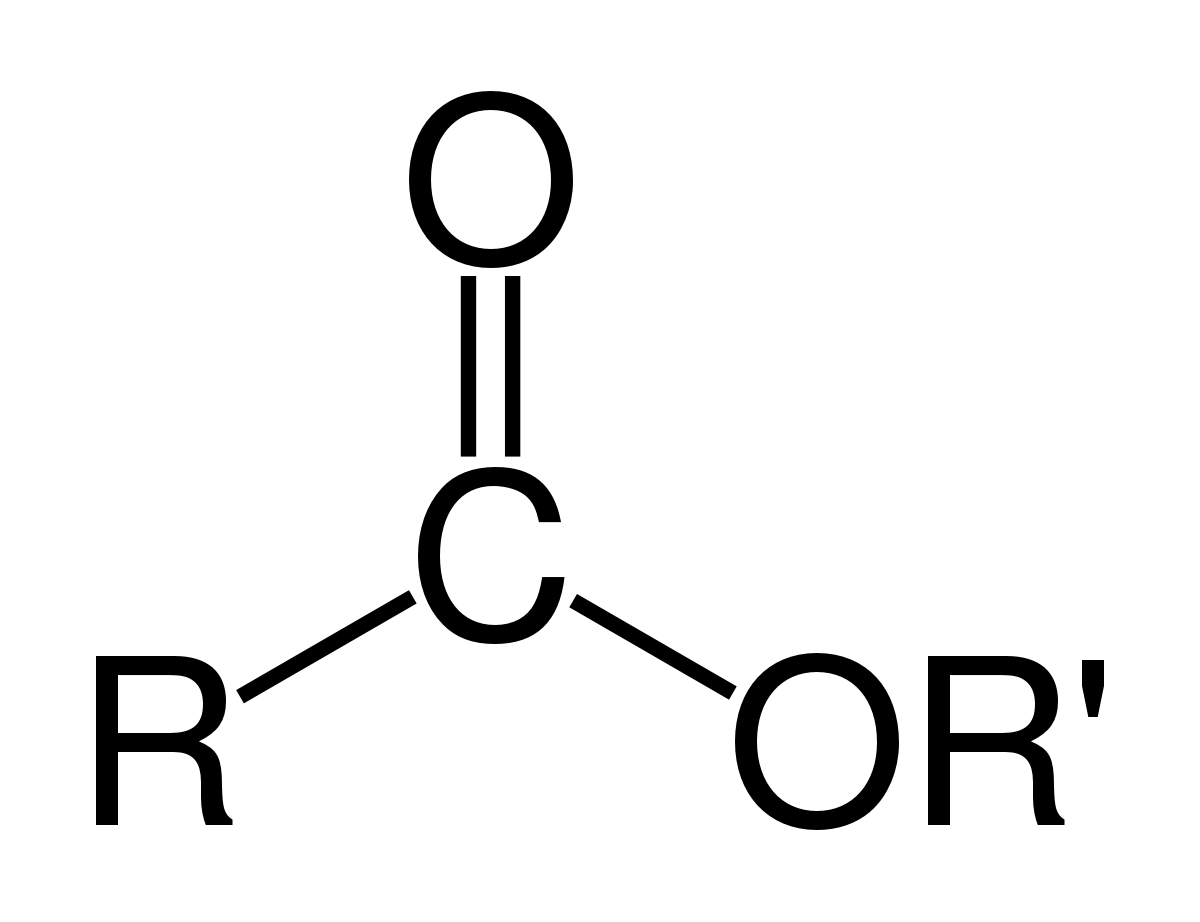
An ester compound releases an alcohol and a carboxylic acid upon hydrolysis.
The compound will release a methanol (an alcohol) and the ethanoic acid component (carboxylic acid).
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
Hydrolysis of an ester such as acetate in presence of a mineral acid: Hydrolysis is a chemical decomposition involving breaking of a bond and the addition |
|
Determination of the Expression Rate of Ethyl Acetate Hydrolysis
So the hydrolysis of ethyl acetate to produce the sodium salt of acetic acid (NaOAc) |
|
Concept laboratory preparation
https://fctemis.org/notes/487_Concept |
|
Researching Chemistry (SCQF level 7) Unit code: FE4J 13 Course
♢ Preparation of benzoic acid by hydrolysis of ethyl benzoate. ♢ Preparation of ethyl ethanoate. ♢ Preparation of cyclohexene from cyclohexanol. A video |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Determine the rate constant and the activation energy of the alkaline hydrolysis of ethyl 3 Pipette VHCl= 5 ml solution of hydrochloric acid (c = 0.04 mol dm- ... |
|
4.5 Carboxylic acids and their derivatives
ethanoic acid ethanol ethyl ethanoate. Acid chlorides. Prepared by the reaction between a carboxylic acid and phosphorus pentachloride PCl5. Dry conditions |
|
FACTFILE: GCE CHEMISTRY
For example ethanenitrile would produce ammonium ethanoate via ethanamide. Acid hydrolysis of nitrile ethyl ethanoate sodium ethanoate ethanol. H2O. CH3CN. |
|
1 Ethanoic acid and ethanol react together to form the ester ethyl
(f) (i) Complete the equation below for the alkaline hydrolysis of ethyl ethanoate using sodium hydroxide. State symbols are not required. (1). 3. CH COOCH. 2. |
|
Topic 3 – Chemical Structure and Bonding
The hydrolysis of ethyl ethanoate is a reversible reaction. The equation for He found that 2.0 mol of ethanoic acid had formed. The information in the ... |
|
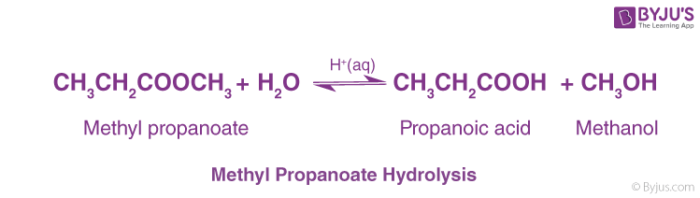
Chapter 5 Carboxylic Acids and Esters
– Ethanoic acid/acetic acid is the main ingredient in Esters may be broken apart under acidic conditions by water (a hydrolysis reaction) to form a carboxylic ... |
|
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
Exercise 6 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE 3 Pipette VHCl= 5 ml solution of hydrochloric acid (c = 0 04 mol dm-3) into a clean and |
|
IV SEMMESTER
KINETICS OF ACID HYDROLYSIS OF AN ESTER AIM: To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE: |
|
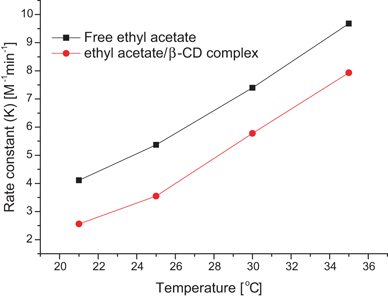
Temperature Dependence of Ester Hydrolysis in Water
The rate of the hydroxyl-promoted hydrolysis of ethyl acetate has been Arrhenius activation energy of the alkaline and acid hydrolysis shows an |
|
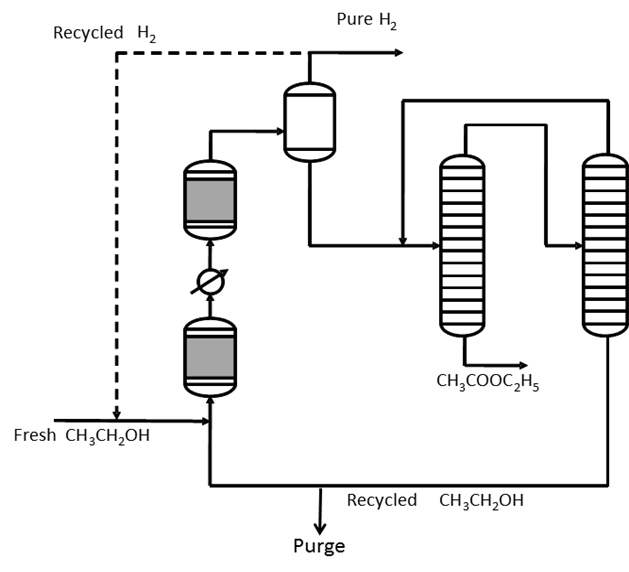
Effect of Ion Exchange Resin Catalyst on Hydrolysis of Ethyl Acetate
alternative heterogeneous catalyst for hydrolysis reaction of ethyl acetate The resin had to be washed using sulphuric acid to give an H+ charge on the |
|
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
The hydrolysis of an ester such as ethyl acetate in the presence of a mineral acid gives acetic acid and ethyl alcohol As the hydrolysis proceeds, there will be proportional increase in the concentration of acetic acid formed |
|
Mechanism of the hydrolysis of ethyl acetate in aqueous solutions of
ionization of ethyl acetate (EA) have been investigated in the greatest detail in solutions of H2SO 4 ta mod- erately concentrated acid, the effective rate constant |
|
IV SEMMESTER
KINETICS OF ACID HYDROLYSIS OF AN ESTER AIM: To determine the rate constant of the hydrolysis of Ethyl acetate using an acid as a catalyst PRINCIPLE : |
|
Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE
1 Exercise 8 KINETICS OF THE HYDROLYSIS OF ETHYL ACETATE Theory CHEMICAL KINETICS Chemical reactions, reaction rate Chemical kinetics is the |

![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://i1.rgstatic.net/publication/332622731_Reaction_rate_and_rate_constant_of_the_hydrolysis_of_ethyl_acetate_with_sodium_hydroxide/links/5cc08bda299bf120977d614d/largepreview.png)




















![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid\u003d31272\u0026t\u003dl)

![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](https://reader017.staticloud.net/reader017/html5/2019111209/553dfe5d5503466f328b488b/bg1.png)
![CSTR Design for Ethyl Acetate Production - [PDF Document] CSTR Design for Ethyl Acetate Production - [PDF Document]](https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/0-malonic-ester-synthesis-example-and-acetoacetic-ester-synthesis-example.gif)








![PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange PDF] Catalytic Hydrolysis of Ethyl Acetate using Cation Exchange](http://www.chem.ucalgary.ca/courses/350/Carey5th/Ch20/esterh3omech.gif)