acid hydrolysis of methyl butanoate
What is the base hydrolysis of methyl butanoate?
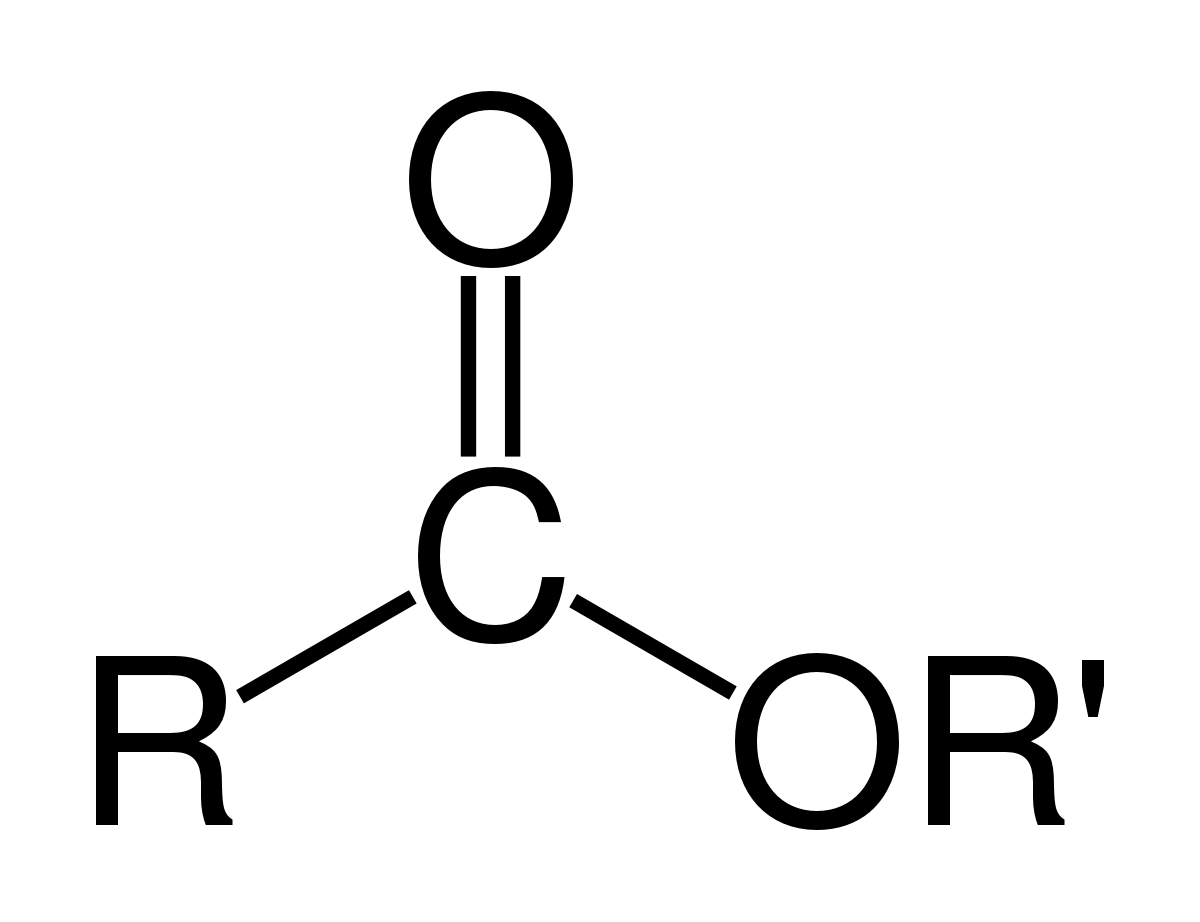
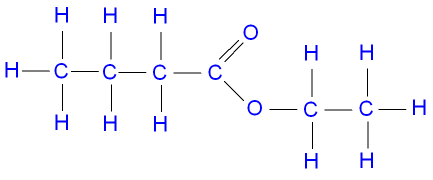
Methyl butanoate is an ester of butanoic acid (C3H7COOH) and methanol (CH3OH) and its formula is C3H7COOCH3.
If it is hydrolysed with an aqueous solution of sodium hydroxide (strong base), the products will be sodium butanoate and methanol.Methyl acetate undergoes hydrolysis, in the presence of an acid (HCl, for example), to give acetic acid and methyl alcohol.
In the presence of an acid, this reaction should be of second order, since two molecules are reacting.
But, it is found to be first order.
What are the products of the hydrolysis reaction of methyl butanoate with Koh?
The substances formed are butanoic acid (C3H7COOH) and methanol (CH3OH), which are respectively the parent acid and alcohol of the ester.20 nov. 2020
What is the acid hydrolysis of ethyl butanoate?
The hydrolysis of an ester compound will give an alcohol and a carboxylic acid as the products.
Since the given ester compound is an ethyl butanoate, it will give off ethyl alcohol as the alcohol product and butanoic acid as the carboxylic acid product.
|
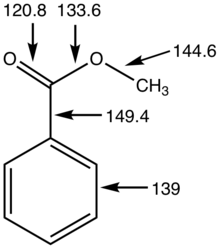
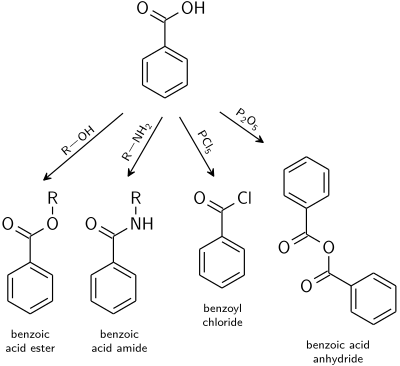
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
The Acid Catalyzed Hydrolysis of Ethyl Esters of Aliphatic Acids
acid hydrolysis of aliphatic esters appear in recent literature. In particular there is the methyl ester. Acetate. 9 |
|
Hydrolysis of chlorogenic acid in apple juice using ap?coumaryl
2019. 7. 20. Hydrolysis of chlorogenic acid in apple juice ... health effects.1011 For chlorogenic acid hydrolysis |
|
Chapter 21 The Chemistry of Carboxylic Acid Derivatives
(c) isopentyl 3-methylbutanoate (common: isoamyl isovalerate) Leaving-group basicities: In the acid-catalyzed hydrolysis of an ester the leaving group ... |
|
Linear Free Energy Relationships from Rates of Esterification and
and k/ko therelative rate for such an ester (or acid) in a given reaction series. 5 Hydrolysis of methyl esters in water |
|
Carboxylic acid ester hydrolysis rate constants for food and
2016. 4. 5. hydrolysis and matrix pH when modelling the evolution of sensory characteristics for foods and beverages with carboxylic acid. |
|
The Effects of Alkali and Temperature on the Hydrolysis Rate of N
1 and Table 1 illustrated that the biggest possibility was that the NMP hydrolysis to produce N-methyl-4-aminobutyric acid in the alkaline environment |
|
Estimation of hydrolysis rate constants of carboxylic acid ester and
Therefore methyl substituents |
|
MDMB-4en-PINACA
2020. 12. 7. Risk assessment report on a new psychoactive substance: methyl 3 ... ester hydrolysis which gives rise to the formation of an acidic ... |
|
Estimated carboxylic acid ester hydrolysis rate constants for food
2011. 9. 28. carboxylic acid ester groups that may be amenable to hydrolysis in various food and beverage products. ... ethyl 3-methylbutanoate. |
|
Estimated carboxylic acid ester hydrolysis rate constants for - CORE
28 sept 2011 · carboxylic acid ester groups that may be amenable to hydrolysis in various food and beverage products Acid- (kA) ethyl 3-methylbutanoate |
|
47 HYDROLYSIS OF ESTERS Hydrolysis of esters is just the
Hydrolysis of esters is just the reverse of esterification: Ethyl butanoate is a major component of peach flavor Pentyl propanoate is a major component of apricot flavor Methyl and ethyl acetate The carboxylic acids are responsible for the |
|
Unit 1 homework answers - WordPresscom
(d) The hydrolysis of methyl butanoate in the presence of sodium hydroxide solution produces the sodium salt of butanoic acid and methanol The equation |
















![Chapter 5 Carboxylic Acids and Esters - [PDF Document] Chapter 5 Carboxylic Acids and Esters - [PDF Document]](https://media.springernature.com/lw685/springer-static/image/art%3A10.1186%2Fs13104-019-4232-1/MediaObjects/13104_2019_4232_Fig2_HTML.png)






![PDF] Enzymatic hydrolysis of castor oil: An approach for rate PDF] Enzymatic hydrolysis of castor oil: An approach for rate](https://d10lpgp6xz60nq.cloudfront.net/physics_images/KSV_CHM_ORG_P2_C14_S01_020_S03.png)