acid hydrolysis of nitriles conditions
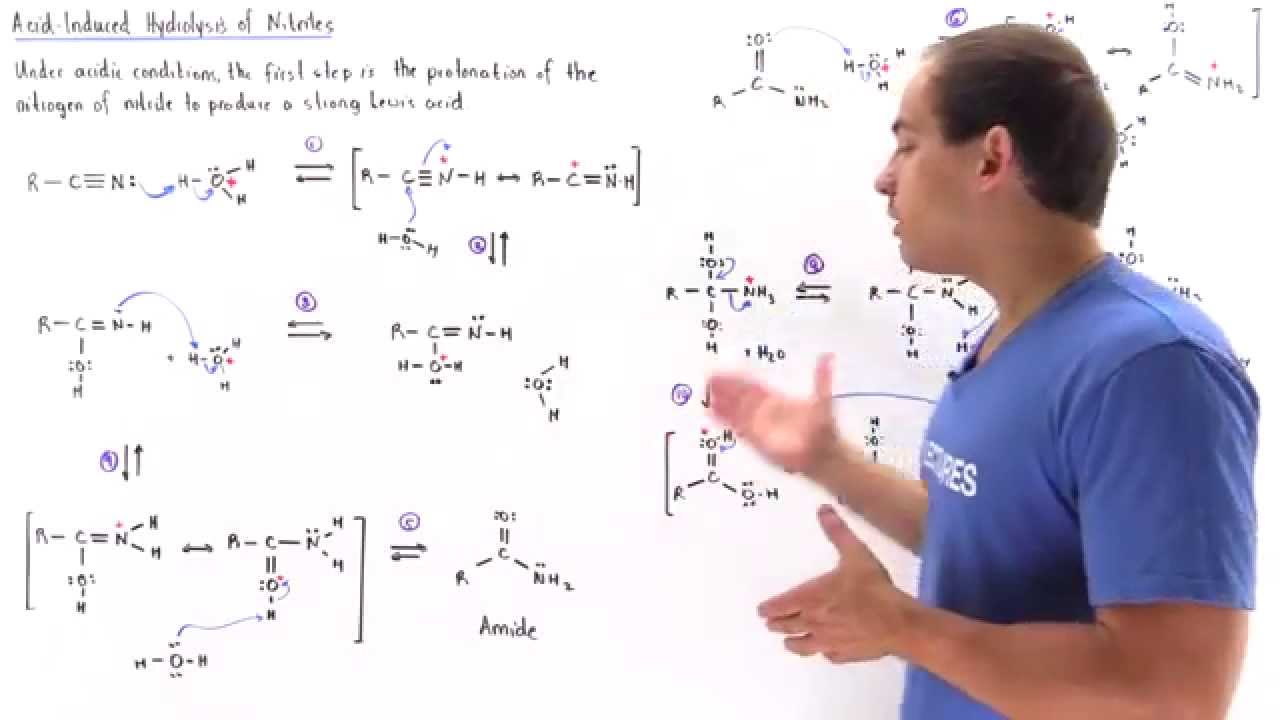
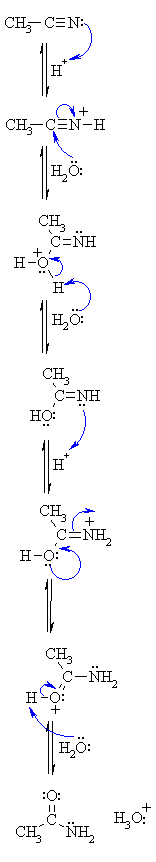
Hydrolysis under acidic conditions
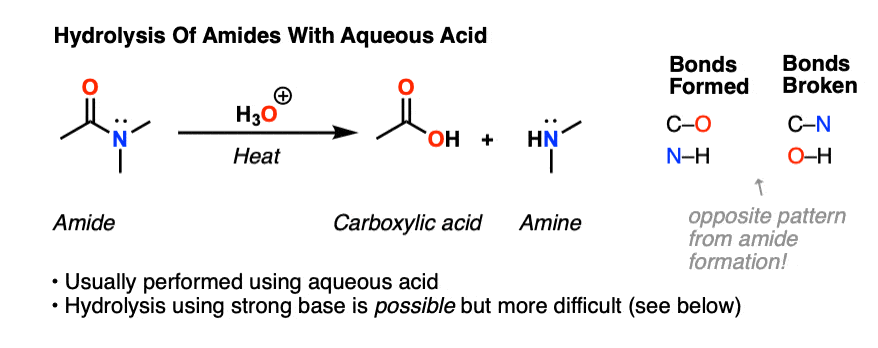
If ethanamide is heated with a dilute acid (such as dilute hydrochloric acid), ethanoic acid is formed together with ammonium ions.
So, if you were using hydrochloric acid, the final solution would contain ammonium chloride and ethanoic acid.
What are the conditions for nitrile hydrolysis?
Nitriles can be hydrolyzed to carboxylic acids in acidic aqueous solutions, and to carboxylate salts with base-catalyzed hydrolysis: In both cases, the transformation consists of two main parts; conversion of the nitrile to an amide and hydrolysis of the amide to the corresponding carboxylic acid.
What are the conditions for nitrile to become a carboxylic acid?
Nitriles get hydrolysed with water to yield corresponding carboxylic acid or salt.
The reaction between water and nitrile doesn't occur easily.
Nitrile is heated with dilute acid or alkali for reaction to occur.
The end product will be a carboxylic acid, but its character may vary depending on the reaction conditions.
|
Dry hydrolysis of nitriles by sodium perborate and copper salts in
Nitriles react with sodium perborate or copper salts to yield carboxylic acids under mild acidic conditions. The multiphase reaction can be carried out in |
|
Hydrolysis of Nitriles
Conventional chemical conversion of nitriles suffers from several disadvantages including the requirement for highly acidic or basic reaction conditions |
|
Anchimeric?Assisted Spontaneous Hydrolysis of Cyanohydrins
Herein we report that cyanohydrins generated by the reaction of cyanide with ?-keto acids and ?-keto-alcohols |
|
Chapter 1 Introduction
highly active catalysts in nitrile hydrolysis under neutral conditions. The hydrolysis of nitriles to amides and carboxylic acids are very important. |
|
N. F. Jain and C. E. Masse Carboxylic acid derivatives can be
Hydrolysis of carboxylic acid esters amides |
|
Enzymatic Hydrolysis of Nitriles and Dinitriles
unfavourable ecological consequences. Clearly the hydrolysis of nitriles to carboxylic acids at room temperature and under neutral conditions would be |
|
Enzymatic hydrolysis of heterocyclic nitriles
requires rather strong acidic or basic conditions. Chemoselective hydrolysis of heterocyclic nitriles can be achieved by an easy to use immobilized bio-. |
|
Mild and Efficient Conversion of Nitriles to Amides with Basic Urea
The hydrolysis of nitriles is a method frequently used for hydration of nitriles apply usually strong acidic conditions espe-. |
|
20_18_20.html.ppt [Read-Only]
Hydrolysis of nitriles resembles the hydrolysis of amides. In basic solution the carboxylic acid product ... under the reaction conditions. |
|
Microbial Hydrolysis of Organic Nitriles and Amides
fungi. One pathway involves the sequential hydrolysis of the nitrile molecule to its corresponding carboxylic acid via an amide intermediate. This reaction. |
|
Hydrolysis of Nitriles
Hydrolysis of nitriles resembles the hydrolysis of amides The reaction is irreversible In basic solution the carboxylic acid product conditions of hydrolysis |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
The nitriles R–CN are anhydrides of the primary amides of carboxylic acids If the acidic or basic hydrolysis of a nitrile requires harsher reaction conditions than |
|
Theoretical study of nitrile hydrolysis by solid acid catalysts
1 jan 2000 · and it is a condition of accessing publications that users recognise and Subject headings: reaction mechanism; hydrolysis/ nitrile/ quantum |
|
Carbon bond formation and Synthesis - WordPresscom
Conditions: Ethanol solvent Nitriles also undergo hydrolysis to form carboxylic acids on heating with dilute acid alcohol + carboxylic acid (acid hydrolysis) |