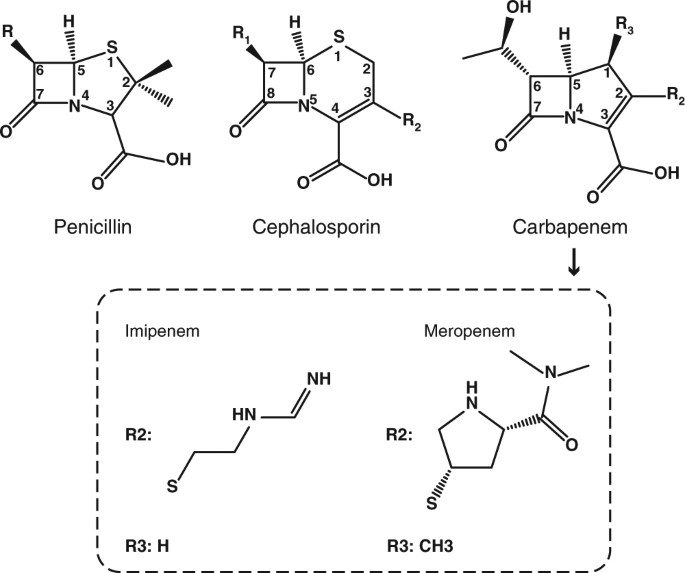
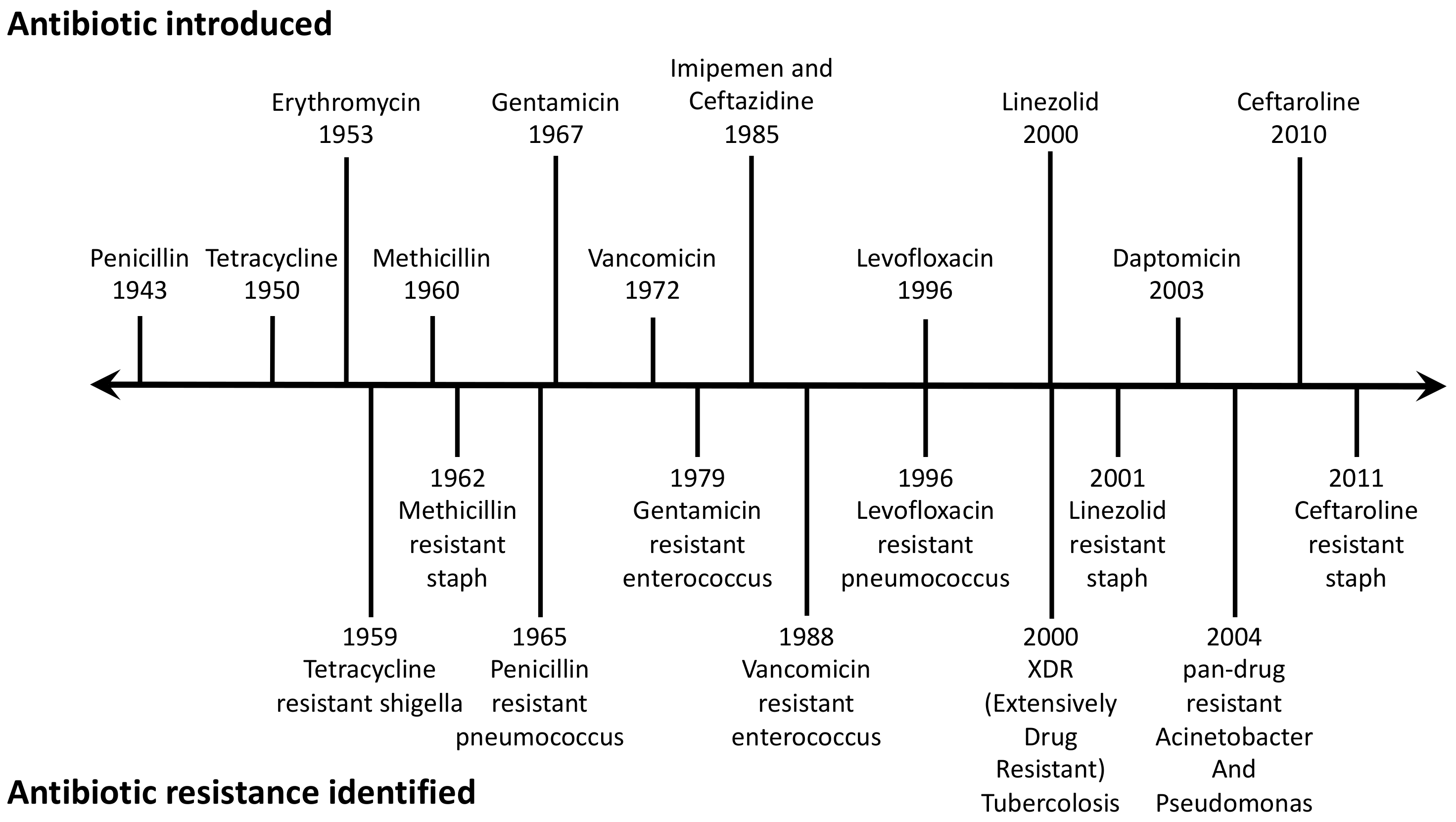
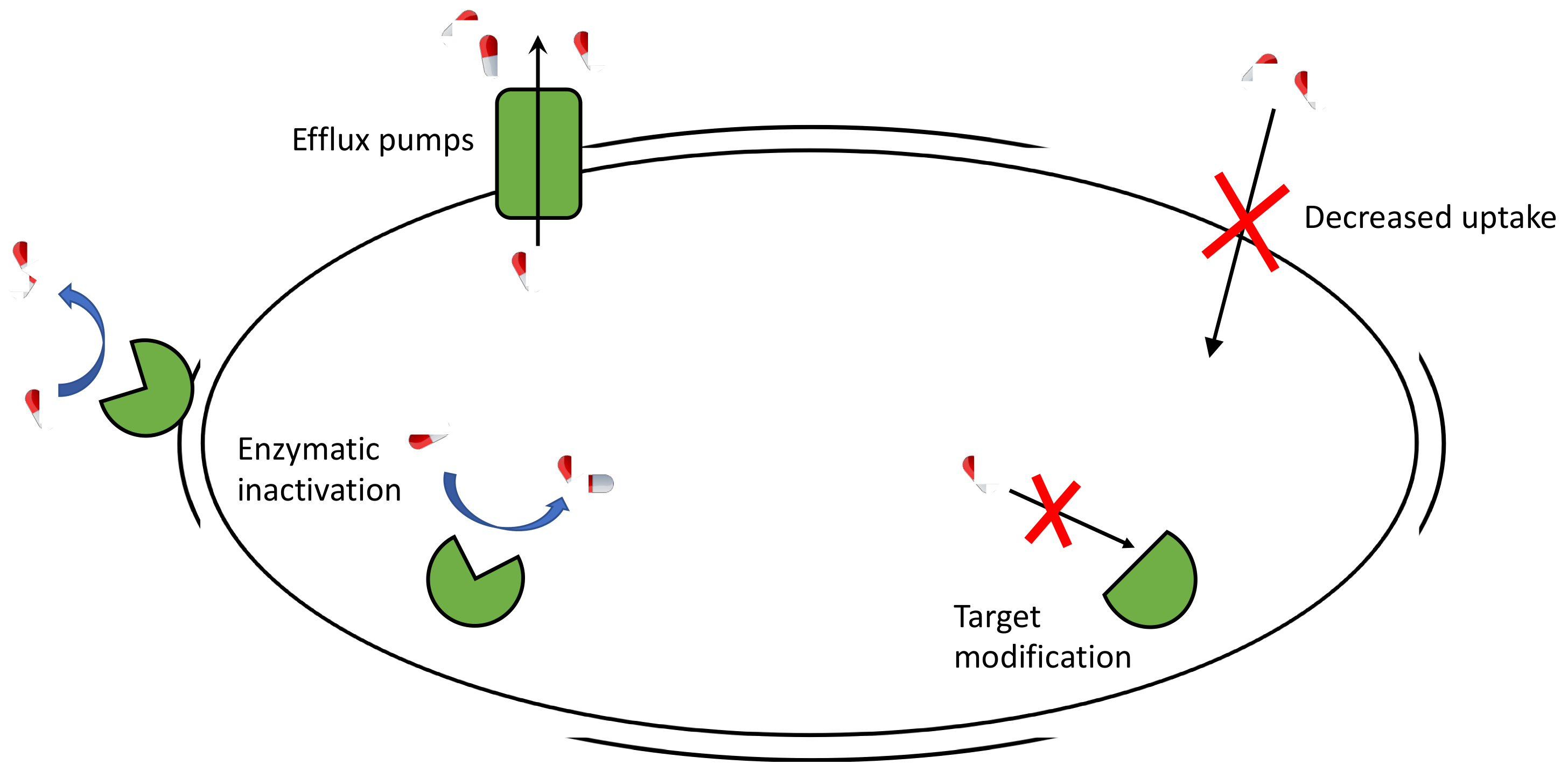
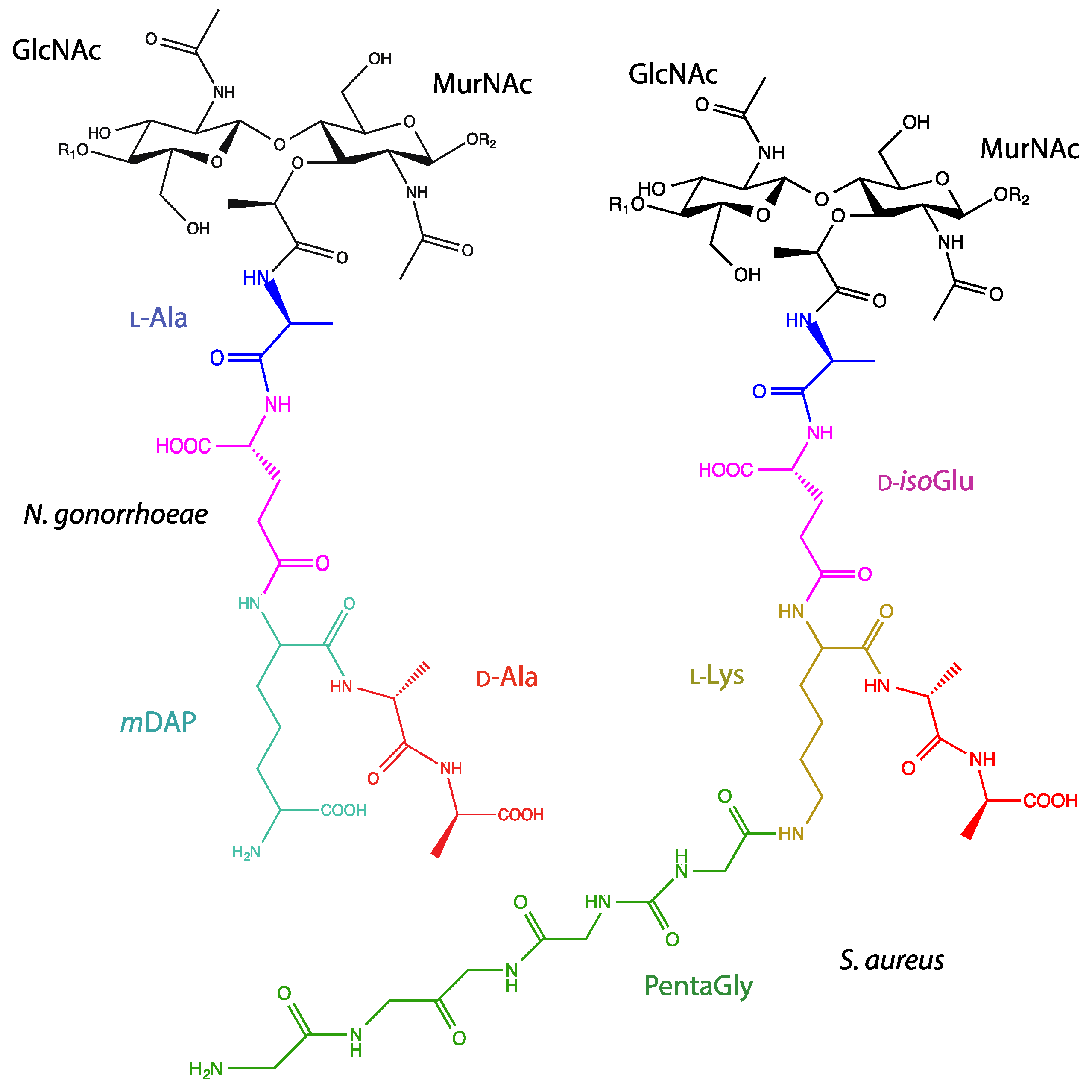
acid hydrolysis of penicillin mechanism
|
Hydrolysis of Penicillin G and Carbenicillin in Pure Water
It is reasonable that the hydrolysis occurred due to the protonation of nitrogen atom of β-lactam ring (acid mechanism) In spite the fact the hydrolysis |
What is the hydrolysis reaction of penicillin?
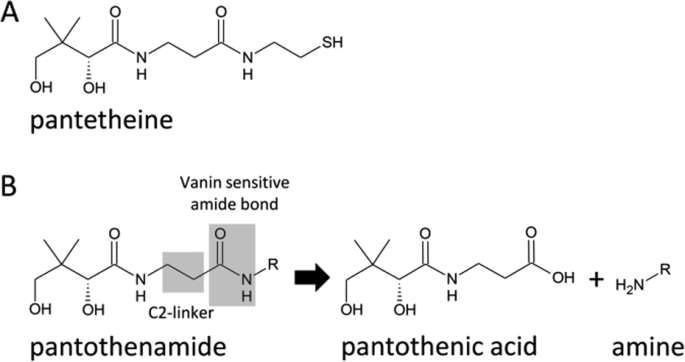
Enzymatic hydrolysis of penicillin G to produce 6-aminopenicillanic acid, key intermediate for the production of semisynthetic β-lactam antibiotics, is one of the most relevant example of industrial implementation of biocatalysts.
The hydrolysis reaction is traditionally carried out in aqueous buffer at pH 7.5-8.The hydrolysis product, penicilloic acid or cephalosporoic acid, is biologically inactive.
FIGURE 8.4.
Is penicillin destroyed by acids?
Penicillin G degrades more easily by stomach acid and has less than 30% bioavailability; therefore, it is a parenterally administered drug.
Because of the short half-life, penicillin G is usually administered in divided doses 4 to 6 hours apart via the intravenous or intramuscular route.
How does penicillin degrade in acidic media?
It is well known that penicillin decomposes in acidic aqueous solution to form both penillic acid (V) and penilloic acid (111) with a maximum yield of the former at pH 2-3 (1).
It is also known that penicillenic acid (IV) is formed in acid solution and subsequently decomposes relatively rapidly (2).
|
Alkaline and acidic hydrolysis of the ?-lactam ring
hydrolysis has been studied through a BAC2 mechanism characterized by a of kinetic studies regarding penicillin and cephalosporin degradation in acidic. |
|
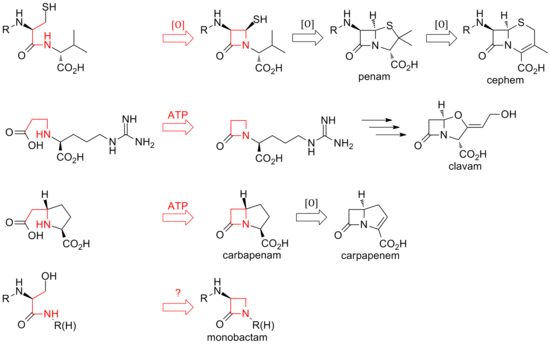
Structure Processing and Catalytic Action of Penicillin Acylase
importance primarily in the production of 6-aminopenicillanic acid (6-APA) penicillin G acylases are consistent with an acyl enzyme mechanism |
|
Mechanism of thiazolidine hydrolysis. Ring opening and hydrolysis
moderately acidic or neutral pH values with either penicillin derivatives or simpler thiazolidines. We have therefore |
|
The mechanism of action of penicillin. Penicillin acylates the active
May 10 2022 cally catalyze the penicillin-sensitive hydrolysis COOH-ter- ... PTH |
|
Comparative stability of cephalosporins in aqueous solution
Acidic Degradation Mechanism and Relative Stability of. Cephalosporins and Penicillins-The acidic degradation rates of penicillins are known to depend on the |
|
Mechanism of penicillin action: penicillin and substrate bind
and peptide composition and quantitationby amino acid analysis after 22-24 hr of acid hydrolysis (22) were as de- scribed. Peptide notation is as follows: |
|
Model catalysts which stimulate penicillinase. V. The
(amides and esters) and penicilloic acid and able to distinguish between them. The mechanism given by eq 5 for the BCD-catalyzed hydrolysis of penicillins |
|
Cupric Ion-Catalyzed Hydrolysis of Penicillins: Mechanism and Site
degradation of the penicillins studied to their corresponding penicilloic acids. This study was undertaken to further substantiate the reaction mechanism |
|
The Effect of the Carboxy Group on the Chemical and p-Lactamase
hydrolysis of esters and lactones of penicillins and cephalosporins. Synthesis of 7- Phenylactamidocephalosporanic Acid Methyl. Ester (5). |
|
Metabolism of Penicillins to Penicilloic Acids and 6
stable in the body, ampicillin was fairly stable, and benzyl penicillin was unstable ically inactive penicilloic acids by hydrolysis of by nonrenal mechanisms |
|
Penicillin Concepts Answer the following questions for the
the penicillins shown have at least one acidic carboxyl group with a pKa from 4- 5 and this route and mechanism of elimination important? (1 point) to acid hydrolysis, but not at the same rate as the beta-lactam ring of F Answers B, C and |
|
Cupric Ion-Catalyzed Hydrolysis of Penicillins: Mechanism and Site
degradation of the penicillins studied to their corresponding penicilloic acids This study was undertaken to further substantiate the reaction mechanism and to |
|
Penicillin Gs function, metabolites, allergy, and - Allied Academies
31 mai 2017 · simply by adding new side chains to 6-aminopenicillanic acid, The discovery of penicillin G was of the mechanisms behind their function Early evidence from the hydrolysis of penicillin G's amide function [36] In alkaline |
|
Theoretical studies of the hydrolysis of antibiotics - Repositori UJI
performed to explore the mechanisms of hydrolysis of two antibiotics, β- lactamases are known, classified into four groups,4 A-D, according to their amino acid |