acid hydrolysis of sucrose
|
Side reactions of acid hydrolysis of sucrose
In condensed solutions of sucrose hydrolysis takes place along with numerous side reactions resulting from interaction between suhstrates and reaction products |
|
Application of the acid hydrolysis of sucrose as a temperature
The hydrolysis of sucrose in an acid medium was used as a temperature indicator to measure holding temperatures in a con- tinuous thermal-processing unit From |
What is the process of the hydrolysis of sucrose?
Hydrolysis of sucrose to give glucose and fructose is called inversion. hydrolysis of sucrose brings about a change in the sign of rotation, from dextro (+) to laevo (–) and the product is named as invert sugar.
Yes, the acidic hydrolysis of sucrose is called inversion reaction.
Also the product sugar is known as invert sugar.
The hydrolysis of sugar is carried out in an acidic medium.
Due to this reason the reaction is said to be acidic hydrolysis of sucrose.15 nov. 2020
What happens when sucrose is hydrolysed with dilute acids?
Sucrose is dexlrorotatory but after hydrolysis , it gives dextrorotatory glucose and laevorotatory fructose.
Does sucrose hydrolyze with HCl?
Reaction 2: Sucrose is protonated by HCl then undergoes a hydrolysis reaction to yield glucose and fructose as the final products.
Reaction 1: Sucrose is broken down by invertase into glucose and fructose via a hydrolysis reaction.
Reaction 3: Glucose is phosphorylated by ATP using hexokinase.
|
Application of the acid hydrolysis of sucrose as a temperature
The hydrolysis of sucrose in an acid medium was used as a temperature indicator to measure holding temperatures in a con- tinuous thermal-processing unit. |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
Acid-Catalyzed Hydrolysis of Sucrose. A student study of a reaction mechanism. H++~GLUCOSE. = a GLUCOSE. Figure 1. For dilute solutions of acids the most |
|
SIDE REACTIONS OF ACID HYDROLYSIS OF SUCROSE
Key words: sucrose fructosylglucose |
|
THE HYDROLYSIS OF SUCROSE IN THE HUMAN STOMACH
Ferris and Lusk. (8) studied the hydrolysis of sucrose by hydrochloric acid in concentra- tions corresponding to those of normal gastric juice and decided that. |
|
Sucrose hydrolysis catalyzed by auto-immobilized invertase into
Acid and enzymatic hydrolysis have been identified as chemical and biochemical ways to sucrose inversion. (disaccharide) into glucose and fructose (soluble. |
|
Kinetic solvent effects on acid-catalyzed hydrolysis of sucrose in
Rate constants of acid-catalyzed hydrolysis of sucrose (S) to D-glucose and L-fructose have been determined at 25OC by optical. |
|
Activities and the Hydrolysis of Sucrose with Concentrated Acids
Results by two methods of extrapolation have been discussed. New Haven Conn. Received August 14 |
|
Autocatalyzed Hydrolysis of Sucrose by Acid
Autocatalyzed Hydrolysis of Sucrose by Acid. By Lawrence J. Heidt F. William Southam and. Edward A. Sullivan1. Received February 4 |
|
Comparison of economic indicators of the sucrose acid inversion or
Inverted syrups are sucrose hydrolysis products of equivalent sweetness which are composed of mix- tures of sucrose |
|
Hydrolysis of Sucrose over Sulfonic Acid Resins
Hydrolysis of Sucrose over Sulfonic Acid Resins. Christoph Buttersack*[a |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
Acid-Catalyzed Hydrolysis of Sucrose A student study of a reaction mechanism H++~GLUCOSE = a GLUCOSE Figure 1 For dilute solutions of acids the most |
|
Application of the acid hydrolysis of sucrose as a temperature
The hydrolysis of sucrose in an acid medium was used as a temperature indicator to measure holding temperatures in a con- tinuous thermal-processing unit |
|
SIDE REACTIONS OF ACID HYDROLYSIS OF SUCROSE
Key words: sucrose, fructosylglucose, acid sucrose hydrolysis By-ptoducts formation chanism [2, 4] In acidic environment fructose undergoes condensation to |
|
Inversion of Sugar : The hydrolysis of sucrose by boiling with a
The hydrolysis of sucrose by boiling with a mineral acid, or by the enzyme invertase, produce a mixture of equal molecules of D-glucose and D-fructose H+ |







![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](http://www.scielo.br/img/revistas/bjps/v46n3/a22fig03.jpg)










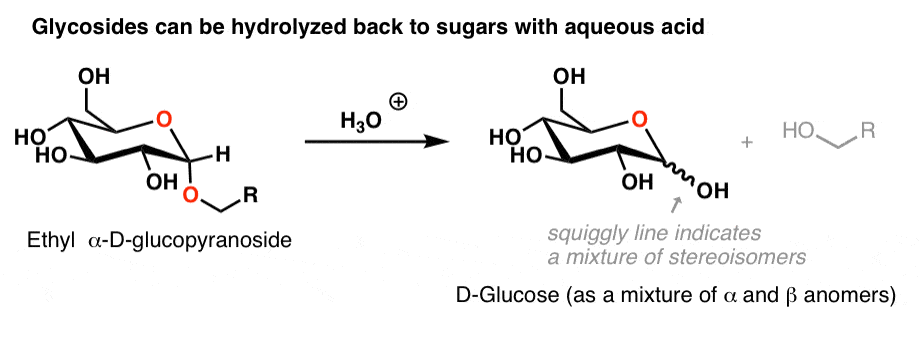







![Structural elucidation of sucrose - [PDF Document] Structural elucidation of sucrose - [PDF Document]](https://cdn.britannica.com/55/2555-050-50E2D75D/Pathways-utilization-carbohydrates.jpg)