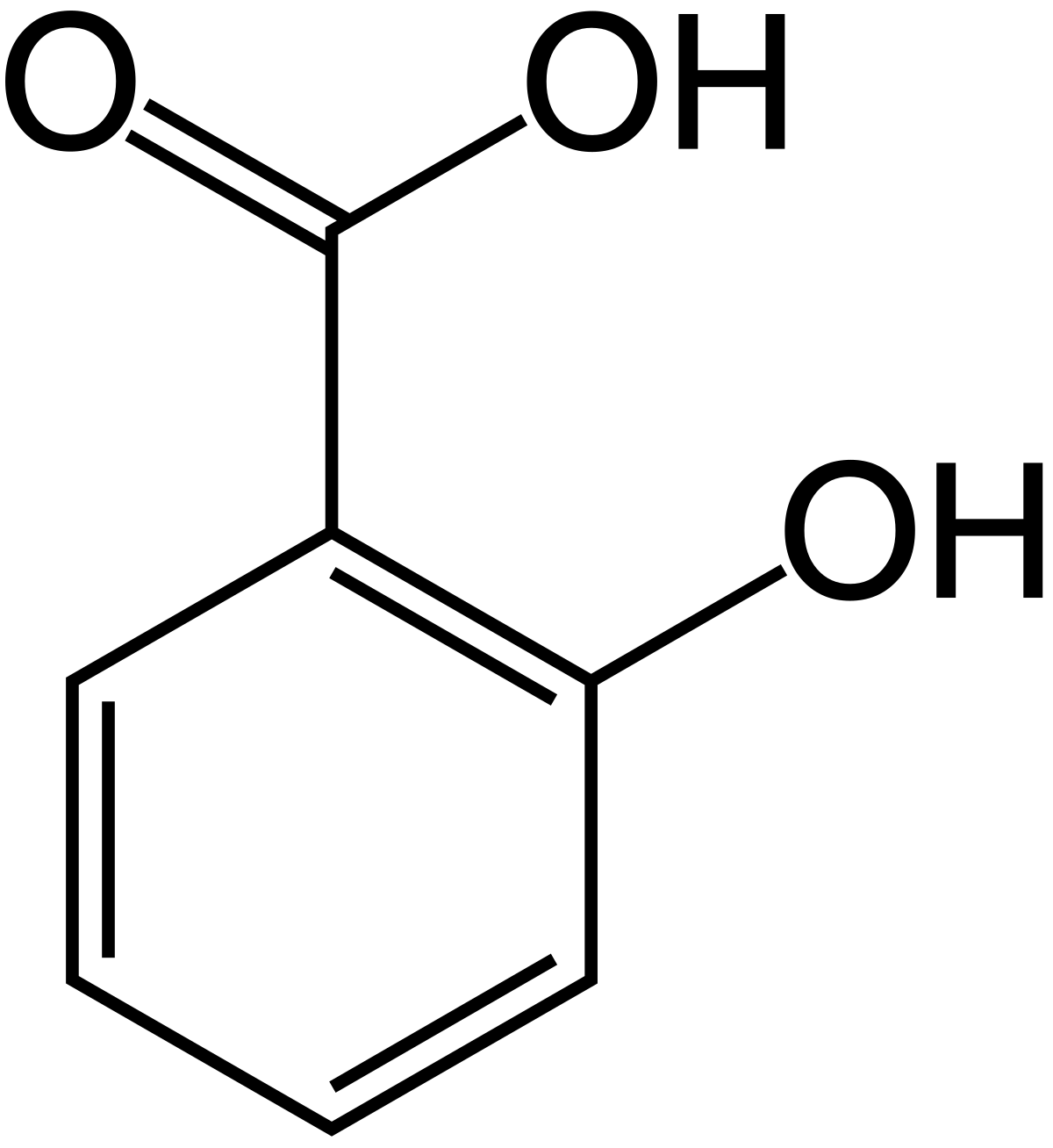
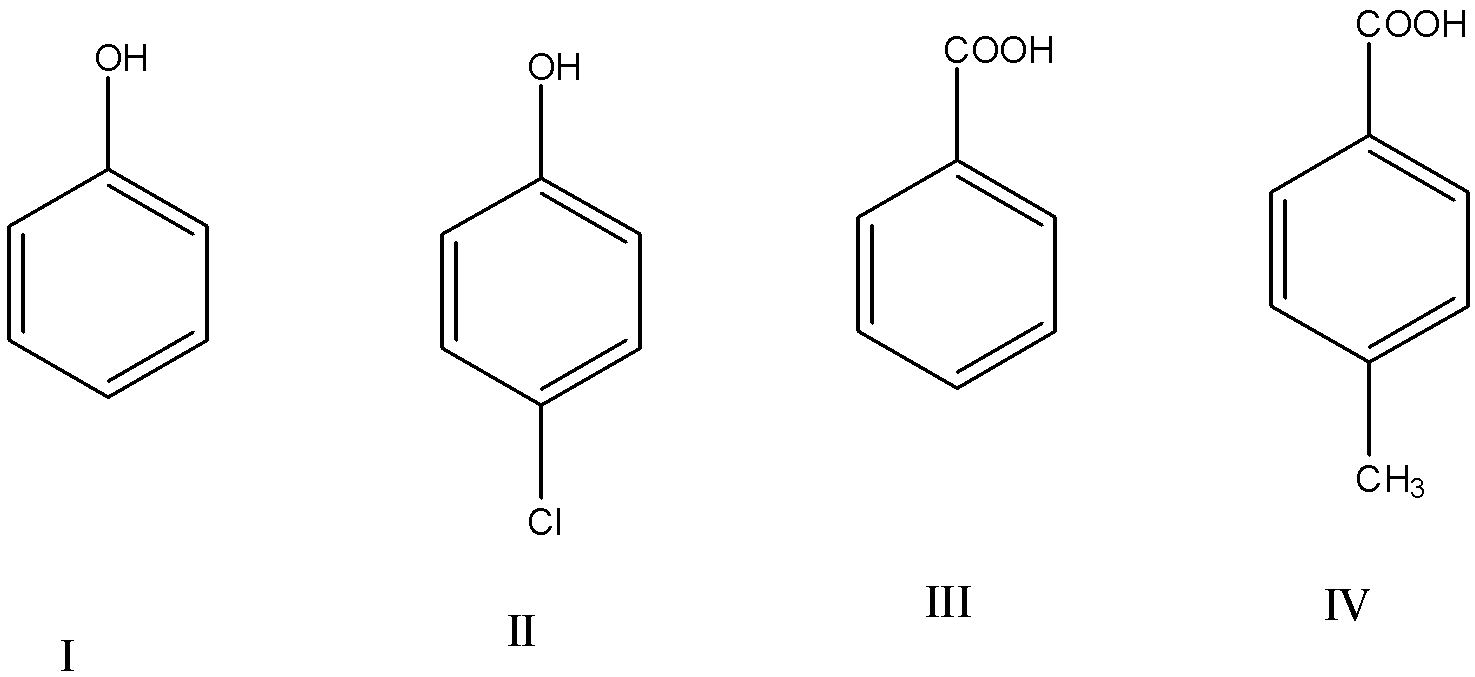
acidity of carboxylic acid and phenol
What is the difference between carboxylic acid and phenol?
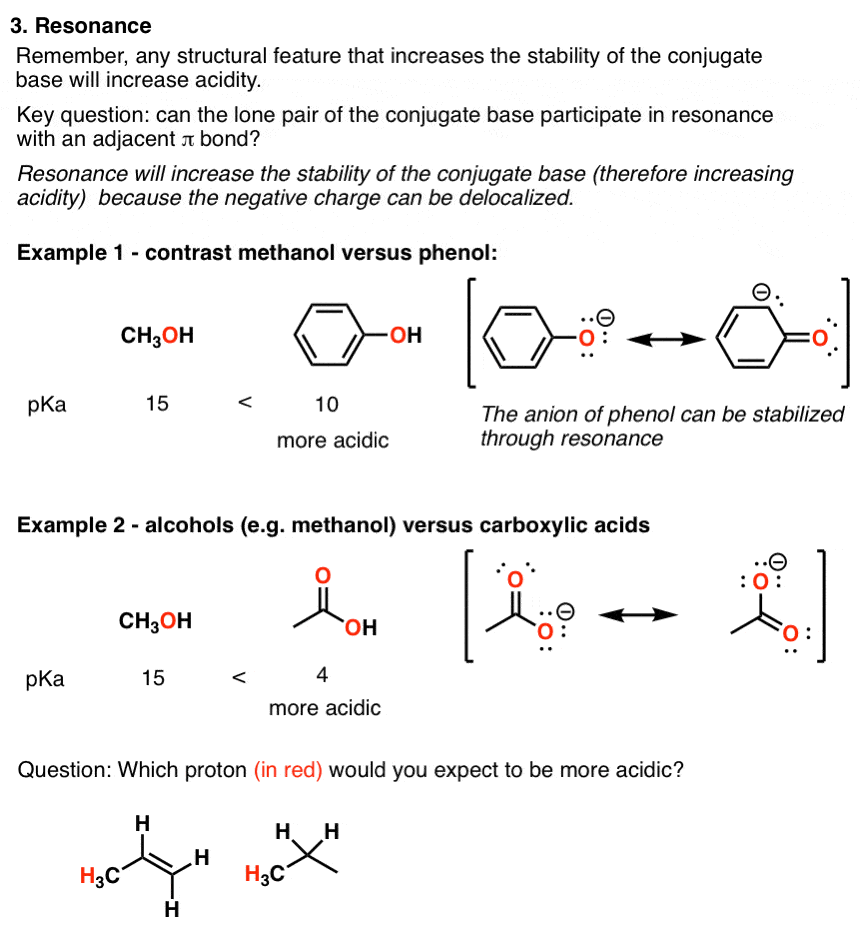
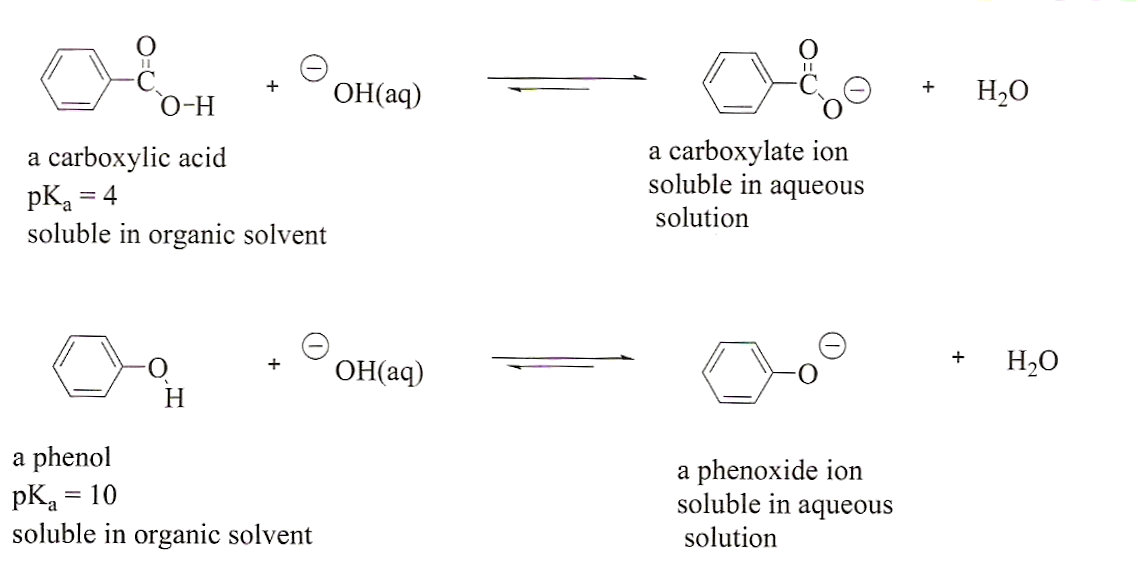
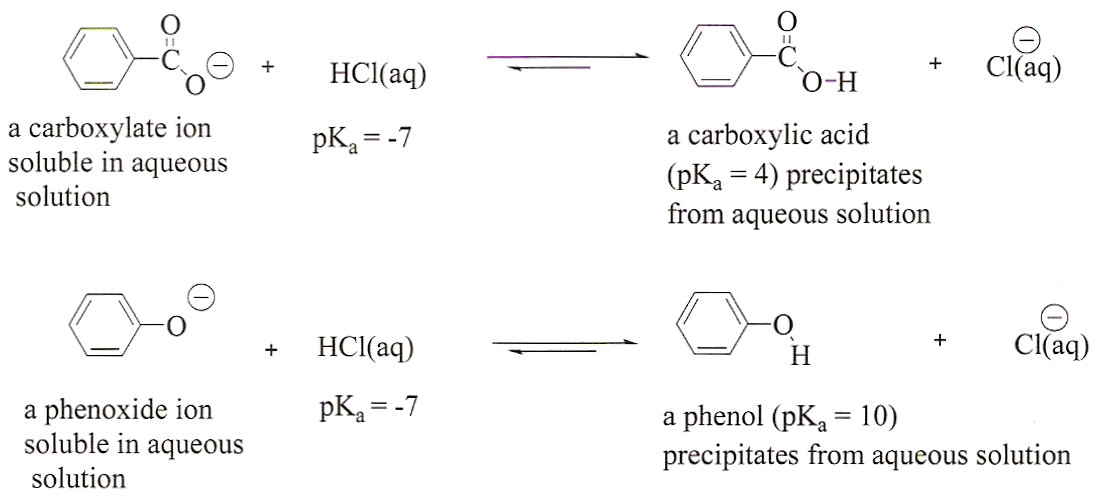
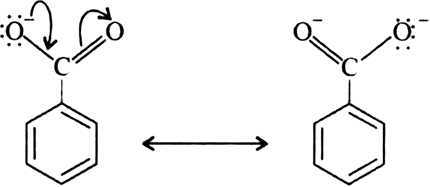
In contrast to phenols, carboxylic acids are more acidic.
The sodium bicarbonate test differentiates phenols from carboxylic acids with ease.
Because carboxylic acids are more powerful acids than phenols, sodium bicarbonate is a weak base that reacts fast with carboxylic acids but not phenols.Which is more acidic between phenol and carbonic acid?
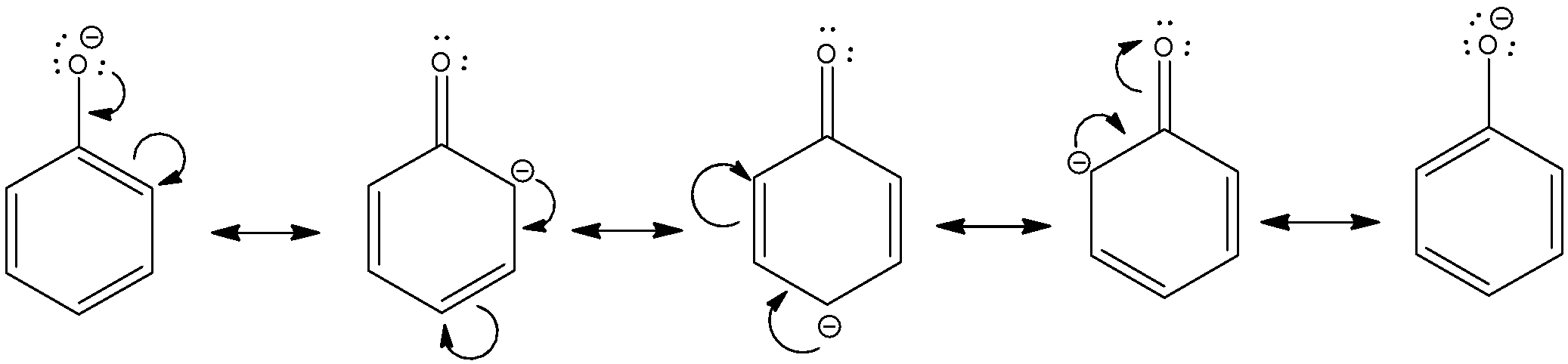
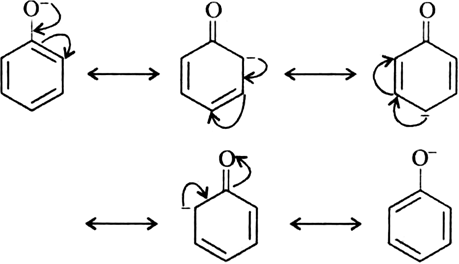
Carbonate ion is more resonance stabilized than phenoxide ion due to two equivalent resonance structures of carbonate ion.
Thus, phenol is weaker acid than carbonic acid (H2CO3).Which is more acidic phenol or carboxylic acid?
(b) Carboxylic acids are more acidic than phenols.9 jan. 2020
The pH of a typical dilute solution of phenol in water is likely to be around 5 - 6 (depending on its concentration).
That means that a very dilute solution isn't really acidic enough to turn litmus paper fully red.
Litmus paper is blue at pH 8 and red at pH 5.
|
Secondary Deuterium Isotope Effects on the Acidity of Carboxylic
2007?3?15? NMR titration method applicable to a mixture of isotopologues. Deuteration definitely decreases the acidity of carboxylic acids and phenols ... |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to carboxylic acid with an alcohol or phenol (plus an. |
|
Chapter 17: Alcohols and Phenols
Phenols are much more acidic than aliphatic alcohols: Carboxylic acids are simply deprotonated by Grignard reagents and do not give addition products. |
|
Untitled
Molecular adsorptions of phenols and carboxylic acids on crosslinked poly(4-vinylpyridine) were acidic using Methyl Red as the indicator (in the adsorp-. |
|
20.4 ACID–BASE PROPERTIES OF CARBOXYLIC ACIDS
reaction with litmus or pH paper. Carboxylic acids are more acidic than alcohols or phenols other compounds with OLH bonds. The acidity of carboxylic acids |
|
Identifying an Unknown Compound by Solubility Functional Group
Phenols are less acidic than carboxylic acids and do not react with NaHCO3 to form Solubility in NaOH indicates either the carboxylic acid or phenol. |
|
Experiment 3: Extraction: Separation of an Acidic a Basic and a
When separating mixtures of organic compounds that have acidic or basic functional groups such as carboxylic acids |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
You already know why phenols are more acidic than alcohols. The higher acidity of carboxylic acids as compared to phenols can be understood similarly. The |
|
Carboxylic Acid Structure and Chemistry
However, phenols are generally less acidic than carboxylic acids based on the electronic nature of atoms over which the charge of the conjugate base is |
|
Chapter 17: Alcohols and Phenols
Phenols are much more acidic than aliphatic alcohols: a benzene ring is Carboxylic acids are simply deprotonated by Grignard reagents and do not give |
|
120 Chapter 24: Phenols Alcohols contain an OH group bonded to
24 5: Substituent Effects on the Acidity of Phenols Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance effect) |
|
Phenols - Patna Science College
Acidity of phenols Phenols are much more acidic than alcohols but less so than carboxylic acids or even carbonic acid Phenols are acidic due to the formation of stable phenoxide ions in aqueous solutions For example, phenol itself gives phenoxide ion on dissociation |
|
Are Benzoic Acids Always More Acidic Than Phenols? The Case of
18 fév 2015 · Keywords: Acidity / Carboxylic acids / Mass spectrometry / Phenols / Protomers To address the title question, the relative intrinsic acidities of |
|
Extraction of aromatic carboxylic acids and phenols by strongly
of an aqueous phase of the acidity desired in a separation funnel for 10 minutes The solutes (an aromatic carboxylic acid or a phenol) were dissolved in the |
|
8 - MSU chemistry
necessary to ensure that the pH was above the pKa of benzoic acid (4 2)? How would So the carboxylic acid but not the phenol will be ionized at pH 7, they |