acidity of carboxylic acid slideshare
What is the order of acidity of carboxylic acid derivatives?
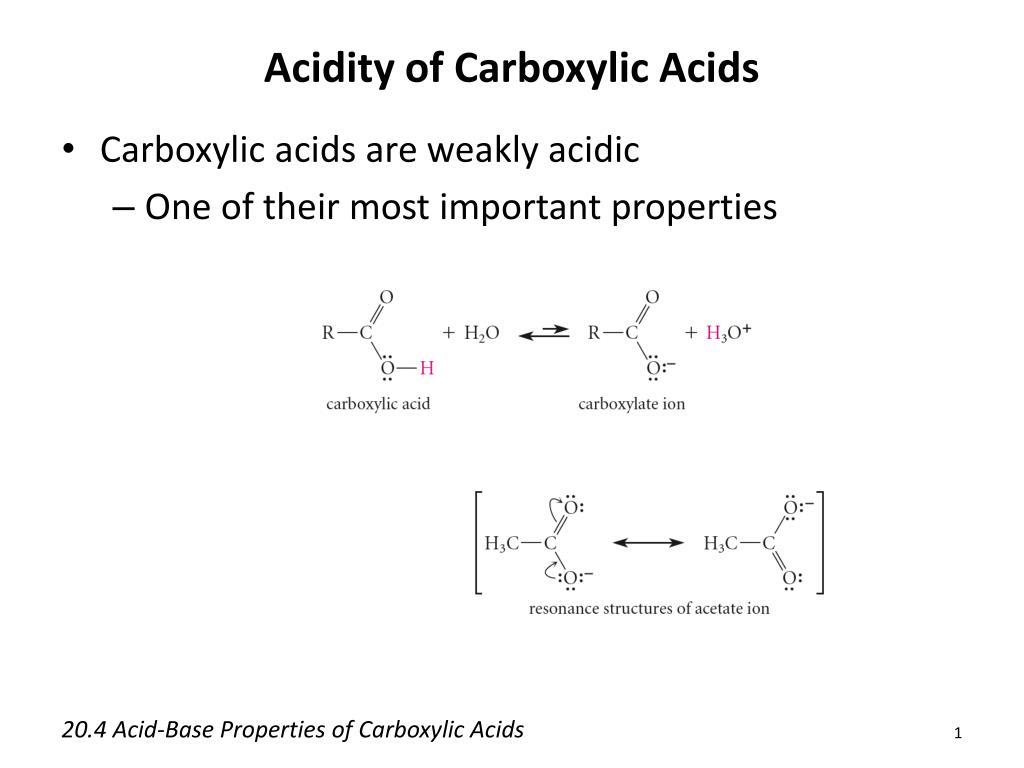
Using the definition of an acid as a "substance which donates protons (hydrogen ions) to other things", the carboxylic acids are acidic because of the hydrogen in the -COOH group.
In solution in water, a hydrogen ion is transferred from the -COOH group to a water molecule.What is acidity of carboxylic acid?
Acidity Of Carboxylic Acids.
Carboxylic acid is an organic compound containing a carboxyl group (COOH) attached to an alkyl or aryl group.
They react with metals and alkalis to generate carboxylate ions.
These reactions of carboxylic acids indicate their acidic nature.Why is COOH so acidic?
The common explanation for why carboxylic acids are more acidic than other molecules (such as alcohols) is that resonance delocalization of charge stabilizes the conjugate base anion relative to the reactant acid.
|
UNIT- II: Aromatic Acids - Acidity effect of substituents on acidity and
➢ Why Benzoic acid will not undergo Friedel-Craft Reaction? - Because -COOH group present in aromatic carboxylic acids is an electron withdrawing group causing. |
|
Chapter 3 Alcohols Phenols
https://www.angelo.edu/faculty/kboudrea/index_2353/Chapter_03_2SPP.pdf |
|
ORGANIC CH B. Sc. II YEAR ORGANIC CHEMISTRY HEMISTRY-II
6.3 Nomenclature of carboxylic acids. 6.4 Structure and bonding. 6.5. Physical properties. 6.6 Acidity of carboxylic acids and effect of substituents on acid |
|
PRACTICAL LAB MANUAL
The general formula for an aliphatic carboxylic acid is RCOOH and for an aromatic carboxylic acid is ArCOOH. mixture until it becomes acidic ad benzoic acid ... |
|
SAMPLE PREPARATION FUNDAMENTALS FOR
recovered by elution with a non-polar acidic solvent such as hexane/EtOAc with 1% acetic acid. carboxylic acid (RCOOH) from the donor side by first ... |
|
Nomenclature of Carboxylic Acids
Esters may be broken apart under acidic conditions by water (a hydrolysis reaction) to form a carboxylic acid and an alcohol. • This is essentially the reverse |
|
Synthesis and Chemistry of Indole
Step 3: Cyclization to indole‐2‐carboxylic acid. Step 4: Decarboxylation. Page 4. By Dr. Divya Kushwaha. 2.4 Bartoli Indole Synthesis: ➢ Efficient and |
|
Chemistry/xii-(2020-21)
Carboxylic Acids: Nomenclature acidic nature |
|
PCI Syllabus: B.Pharm UNIT –V: Carboxylic Acids The carboxyl
to silver mirror and Fehling's solution to red ppt. and itself gets Since conjugation of carboxylic acid to aryl rings is known to increase the its acidity. |
|
Chapter 1 Organic Compounds: Alkanes Organic chemistry
– Many functional groups contain oxygen atoms such as alcohols |
|
CARBOXYLIC ACIDS
Substituents mainly exert their influence on the acidity of aliphatic carboxylic acid through the inductive effect. Since the inductive effect operates through |
|
UNIT- II: Aromatic Acids - Acidity effect of substituents on acidity and
There are several categories of aromatic acids including: (i) Phenolic acids: substances containing an aromatic ring and an organic carboxylic acid function |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
The carbonyl group may be further oxidized to carboxylic acids Addition of water to carbonyl compounds under acidic conditions is analogous. |
|
14: Substituent Effects
Chapter 14. 14: Substituent Effects. Substituents and Their Effects. Carboxylic Acid Acidity. SN1 Reactions. Electrophilic Aromatic Substitution Reactions. |
|
Mitsunobu Reaction
carboxylic acids in the presence of diethyl azodicarboxylate. (DEAD) [A0705] and triphenylphosphine (TPP) Furthermore the p?a of the usable acidic. |
|
Lab Manual - Pharmaceutical Organic Chemistry
Functional group test: test for carboxylic acid 3- Aq. solution + CaCl2: A white ppt. of Ca oxalate is separated ... 2- It gives Acidity test +ve. |
|
Chapter 21: Carboxylic Acid Derivatives
all derivatives of carboxylic acids: carboxylic acid ester anhydride acyl halides amides ... basic or acidic conditions the latter being more common. |
|
AMINO ACIDS CLASSIFICATION AND PROPERTIES
Acidic amino acids: have carboxyl group in their side chain. Eg: Aspartic and Glutamic acid. • Basic amino acids: contain amino group in their side chain. |
|
Synthesis and Chemistry of Indole
Step 3: Cyclization to indole?2?carboxylic acid Reaction involves acidic treatment of 2-arylamino-ketones (produced from a 2–halo-ketone. |
|
211 Introduction Carboxylic Acids 211 Introduction Carboxylic
25 avr 2012 · Copyright 2012 John Wiley Sons, Inc Klein, Organic Chemistry 1e 21-8 • How does the pKa value for a carboxylic acid |
|
Carboxylic Acids
Acidity Carboxylic acids can dissociate in aqueous solution into carboxylate ions and protons Both alcohols and carboxylic acids are acidic since their respective O-H bonds can be broken heterolytically, giving a proton (See next slide) |
|
CARBOXYLIC ACIDS
Any factors that stabilize the excess charge on the carboxylate anion will enhance the acid dissociation constant and hence the acidity of the carboxylic acid |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine The pKa of butanoic acid is about 4 8 previous slide are not identical |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
The carbonyl group may be further oxidized to carboxylic acids H3C C H CH3 Compare pKa values of the conjugate acids of aldehyde/ketones with those for product of this step is a halomagnesium alkoxide (see previous slide) Step 2 |
|
PCI Syllabus: BPharm UNIT –V: Carboxylic Acids The carboxyl
Carboxylic acids: Acidity of carboxylic acids, effect of substituents on acidity, to silver mirror and Fehling's solution to red ppt and itself gets oxidised to CO2 |
|
19_06_12htmlppt [Read-Only]
standard of comparison is acetic acid (X = H) Substituent Effects on Acidity X CH 2 COH O K a = 1 8 x 10-5 pK a = 4 7 |
|
Chapter 17: Alcohols and Phenols
76 Chapter 17: Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol Carboxylic acids are simply deprotonated by Grignard reagents and |
|
Acidity of phenols, effect of substituents on ac - Yengage
Phenols are less acidic than Carboxylic acids (pKa ≈ 5) COMPARISON OF ACIDITY OF PHENOLS AND ALCOHOLS • Phenol exists as resonance hybrid of |
|
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides
A carboxylic acid contains a carboxyl group, which is found on carbon 1 in carboxylic acids O CH 3 — C—OH Acidity of Carboxylic Acids Carboxylic |