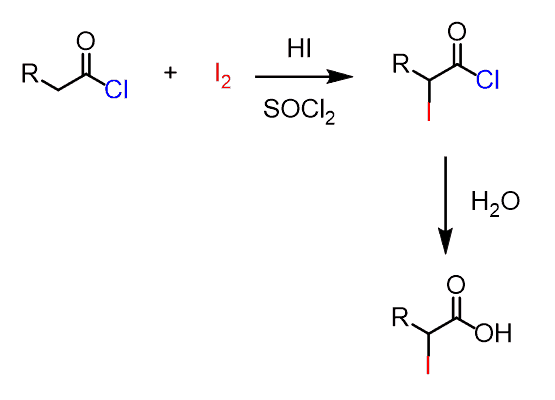
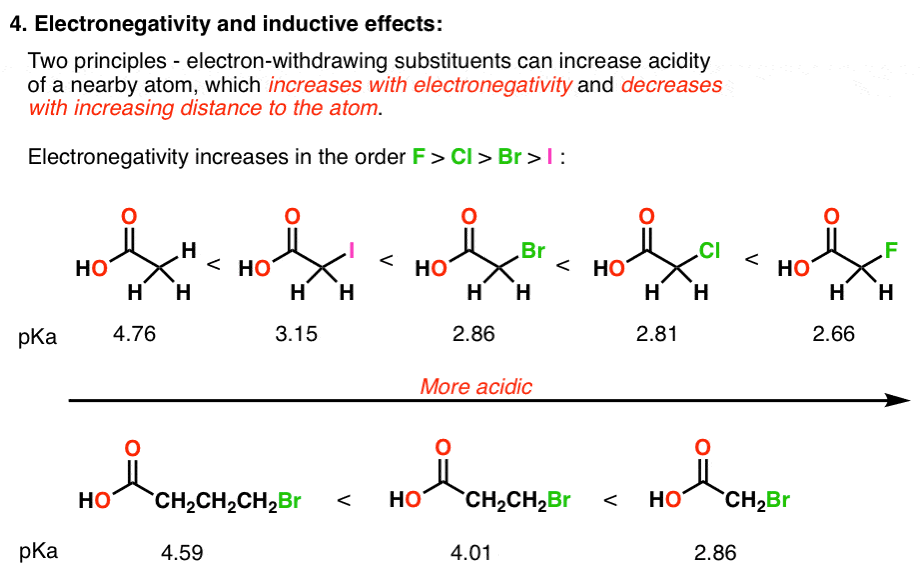
acidity of carboxylic acids with halogens
The acidic strength of hydrogen halide increases in the order of HF< HCl< HBr< HI.
What factors affect the acidity of carboxylic acids?
The presence of such groups near the COOH group of a carboxylic acid often has an effect on the acidity.
In general, electron-withdrawing groups increase acidity by increasing the stability of the carboxylate ion.
In contrast, electron-donating groups decrease acidity by destabilizing the carboxylate ion.
What is the effect of CL on the acidity of carboxylic acids?
C: Inductive effects
The presence of the chlorine atoms clearly increases the acidity of the carboxylic acid group, but the argument here does not have to do with resonance delocalization, because no additional resonance contributors can be drawn for the chlorinated molecules.
Do carboxylic acids react with halogens?
The silver(I) salts of carboxylic acids react with halogens to give unstable intermediates which readily decarboxylate thermally to yield alkyl halides.
The reaction is believed to involve homolysis of the C-C bond and a radical chain mechanism.
|
Reactions of the Halogens with the Silver Salts of Carboxylic Acids.
equivalent quantities of the silver salt of a carboxylic acid and halogen in an inert solvent produces a complex intermediate which may be thermally decom |
|
Formation of Carboxylic acid halides by the reactions of halogens
C6H5CH2 or C6H6 with the halogens(X2) chlorine |
|
HALOGENATION XX. THE REPLACEMENT OF SULFONIC ACID
THE REPLACEMENT OF SULFONIC. ACID GROUPS BY HALOGENS. By Rasik Lal Datta and. Jagadish Chandra Bhoumik. Received April 10 1920. |
|
THE REACTION OF CYSTINE AND ALANINE-3-SULPHINIC ACID
The mechanism of formation of the peptide bond is discussed and a sulphonic-carboxylic acid mixed anhydride is proposed as an intermediate. REACTIONS OF AMINO ... |
|
Identification of Organic Compound by Organic Qualitative Analysis
Carboxylic acid confirmed. (b) 0.1gm of substance +3 ml dilute. NaOH solution.Shake well. The substance dissolves. |
|
Derivatives of carboxylic acids
• carboxylic acid + ammonia (or amine) → amide + water. Page 13. 2.5. ACYLHALIDES R―CO―X X = halogen. • acyl name + halide. • e.g. acetyl chloride (= ethanoyl |
|
The Degradation Of Carboxylic Acid Salts By Means Of Halogen
general review of the reactions of silver salts of carboxylic acids with halogens. However his discussion is not comprehensive with respect to the |
|
Hydrogen bond donor solvents enabled metal and halogen-free
can be recovered and reused; 2) using halogen free starting material (carboxylic acids carboxylic acid anhydrides or even form carboxylic acid directly as ... |
|
Effects of halogen substituents on the intrinsic acidity of acetic acids
obtained for this intermediate. (8) Yield not optimized. (9) E. J. Corey and A. Venkateswarlu J. Amer.Chem. Soc. |
|
APPGCET -2022: SYLLABUS TEST NAME: 310 – Chemical Sciences
Group – 17: Inter halogen compounds and pseudo halogens Resonance or Mesomeric effect application to (a) acidity of phenol |
|
Formation of Carboxylic acid halides by the reactions of halogens
C6H5CH2 or C6H6 with the halogens(X2) chlorine |
|
Reactions of the Halogens with the Silver Salts of Carboxylic Acids.
equivalent quantities of the silver salt of a carboxylic acid and halogen in an inert solvent produces a complex intermediate which may be thermally decom-. |
|
HALOGENATION XX. THE REPLACEMENT OF SULFONIC ACID
REPLACEMENT OF SULFONIC ACID GROUPS BY HALOGENS. 303. [Contribution from carboxylic acid the replacement of sulfonic groups takes place by halo-. |
|
Reactions of Halogens with Compounds Containing the Carbonyl
In aqueous solution the acid reacts with bromine at a rate which group of a carboxylic acid differ in many respects from those of. |
|
Gas phase acidities of aliphatic carboxylic acids based on
These include halogen OH |
|
Simple Chemical Segregation Guidelines
acids aldehydes |
|
Effects of halogen substituents on the intrinsic acidity of acetic acids
the extract underwent clean acid-catalyzedcyclization mostly aliphatic carboxylic acids. ... introduction of a halogen substituent leads to a large. |
|
Reactions of Silver Salts of Carboxylic Acids with Halogen
of concentrated sulfuric acid in 290-ml. of water. When all of the solid material had disappeared after two and one-half hours the ether layer was. |
|
THE REACTION OF CYSTINE AND ALANINE-3-SULPHINIC ACID
THE REACTION OF CYSTINE AND ALANINE-3-SULPHINIC ACID WITH. HALOGENS sulphonic-carboxylic acid mixed anhydride is proposed as an intermediate. |
|
Compatibility Guide
acids aldehydes |
|
Carboxylic Acid Structure and Chemistry
The carboxylic acid moiety is considered to be a highly polar organic carbon atom nearer the reaction site (OH) is more acidic that the acid where the chlorine |
|
Carboxylic Acid Structure and Chemistry
As a result of their relatively acidic nature, carboxylic acids will ionize if Replacement of the carboxylic acid OH with a halogen as in acid chlorides (and other |
|
Acidty of carboxylic Acid - JNMPG COLLEGE
pKa 's of some typical carboxylic acids are listed in the following table Additional halogen substituents have an additive influence, and moving the substituent |
|
CARBOXYLIC ACIDS
The common name of a carboxylic acid (R-COOH) is derived by adding the suffix halogen is another manifestation of the effectiveness of charge dispersal in |
|
Derivatives of carboxylic acids
of an acid 1 1 HALOGEN DERIVATIVES (= 2-hydroxypropanoic acid or α- hydroxypropionic acid) carboxylic acid + ammonia (or amine) → amide + water |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
of -NH2 moiety or to halogens are called amides and acyl halides acids [ Section 12 6 1] by replacing the ending –ic of acid with aldehyde At the same time, |
|
36 RELATIONSHIP OF STRUCTURE TO ACIDITY
acid more acidic Electronegative substituent groups such as halogens increase the acidities of carboxylic acids by stabilizing their conjugate-base carboxylate |
|
204 ACID–BASE PROPERTIES OF CARBOXYLIC ACIDS
3 6C), that halogen substitution within the alkyl group of a carboxylic acid enhances acidity by a polar effect Trifluoroacetic acid, commonly abbreviated TFA, is |
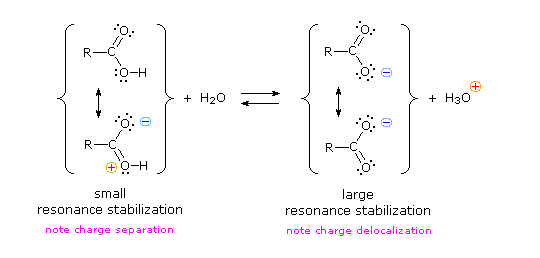

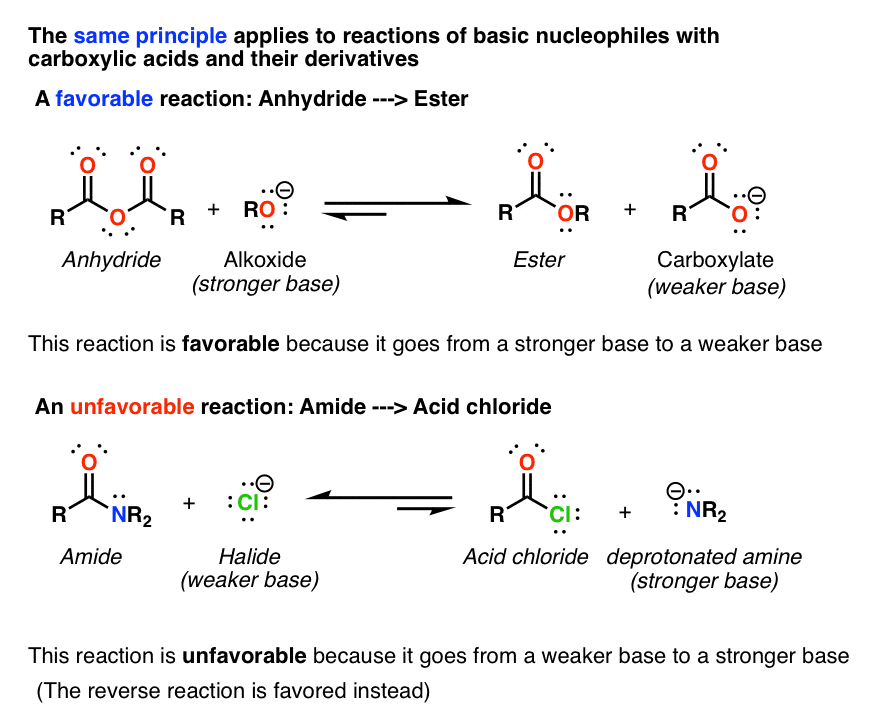






![Carboxylic Acid \u0026 Nitriles - [PDF Document] Carboxylic Acid \u0026 Nitriles - [PDF Document]](https://ars.els-cdn.com/content/image/3-s2.0-B0080447058002223-gr53.jpg)