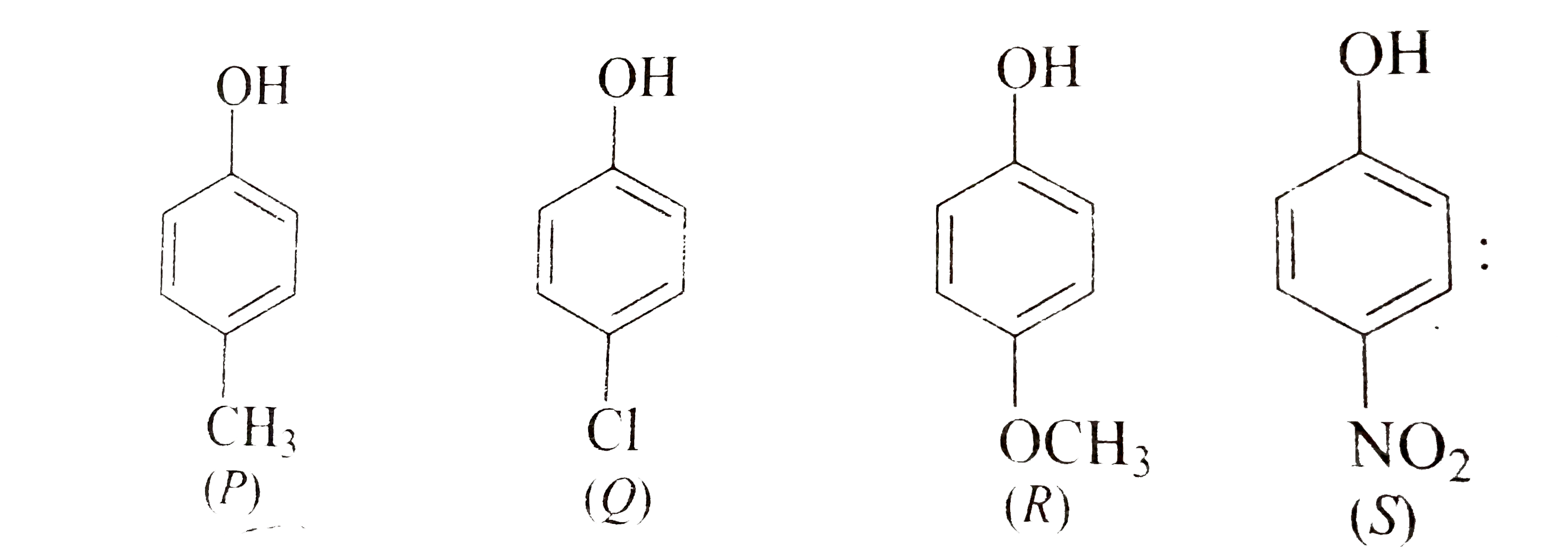acidity of phenol
|
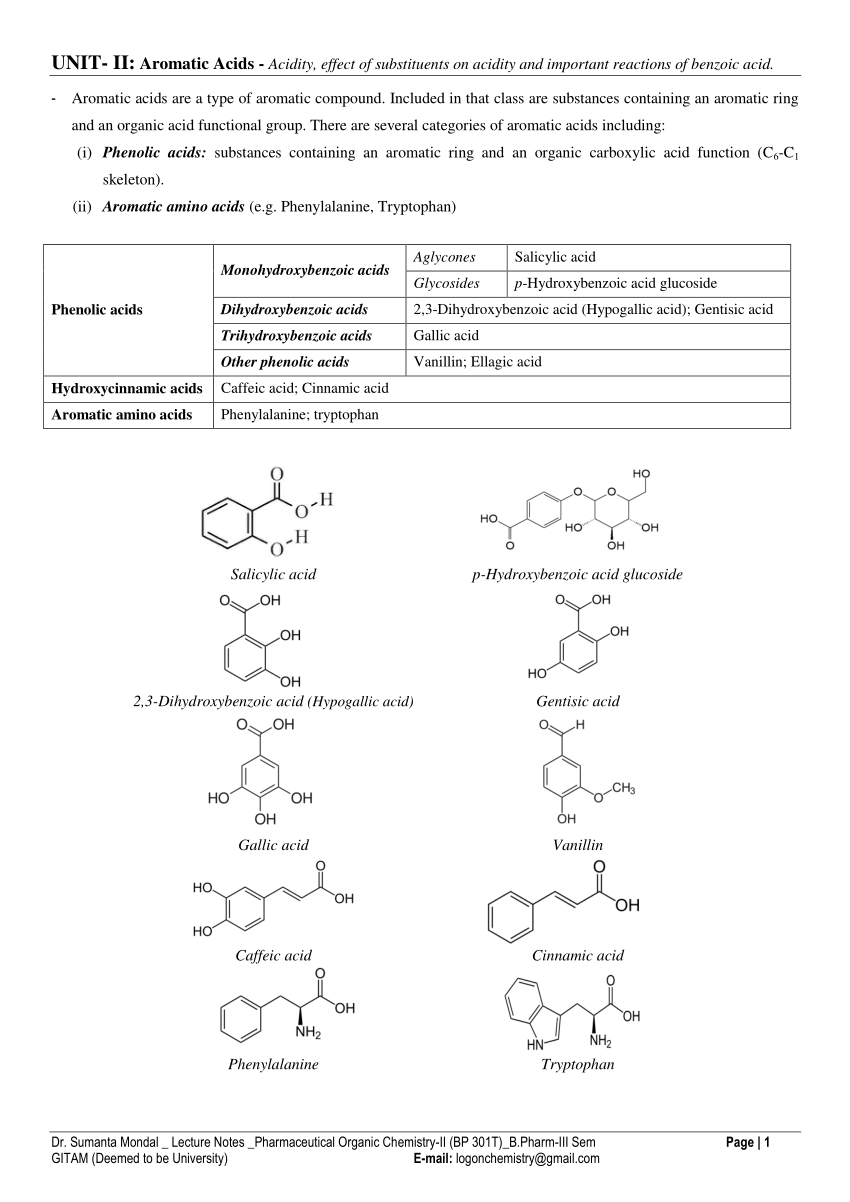
Acidity of phenols effect of substituents on acidity qualitative tests
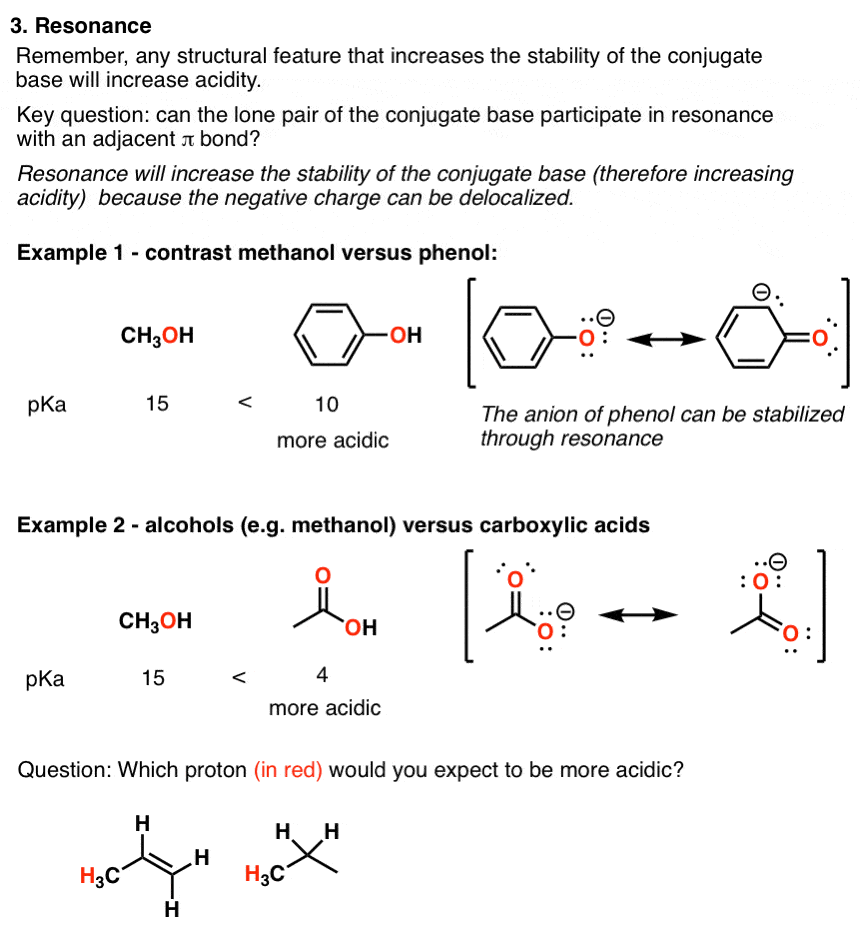
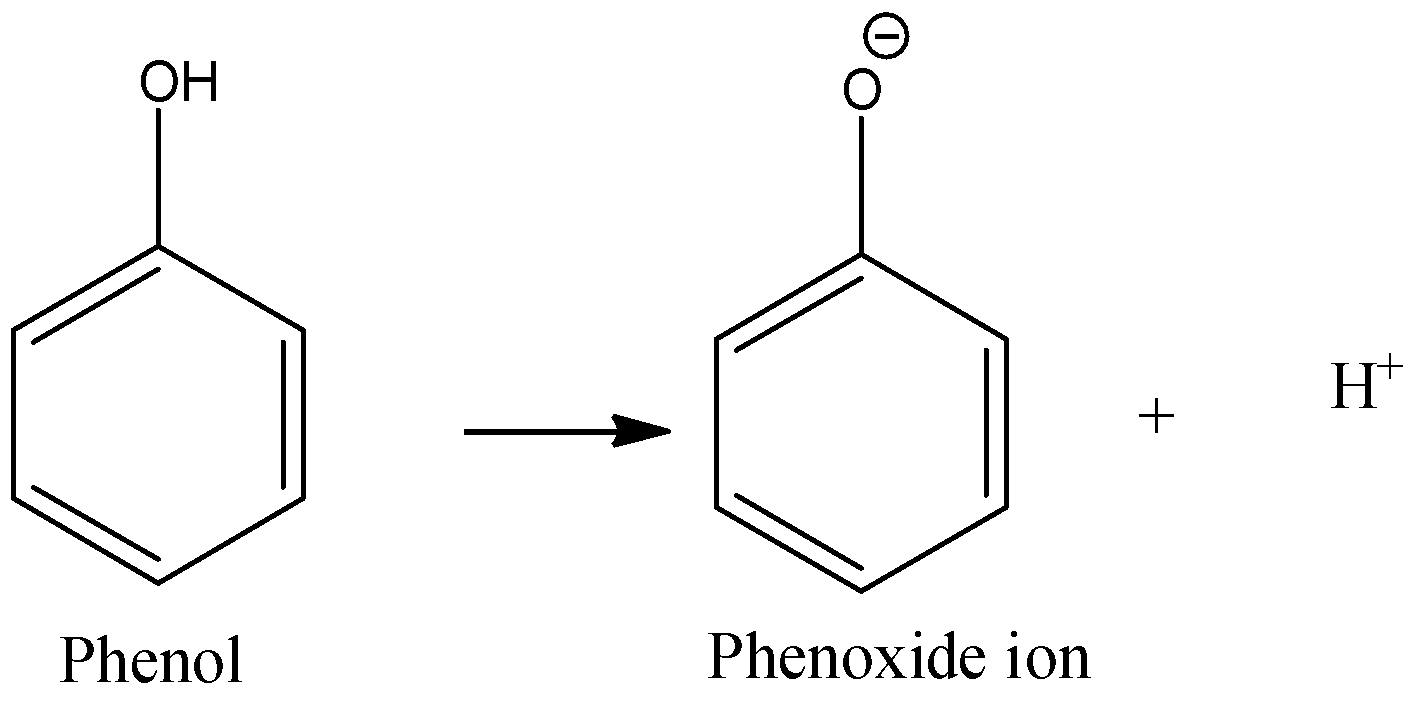
Compare to carboxylic acids phenols are weaker acid - Hence hydroxy group bonded to benzene ring is much more acidic than hydroxy group to alcohol This is |
|
Chapter 24: Phenols Alcohols contain an OH group bonded
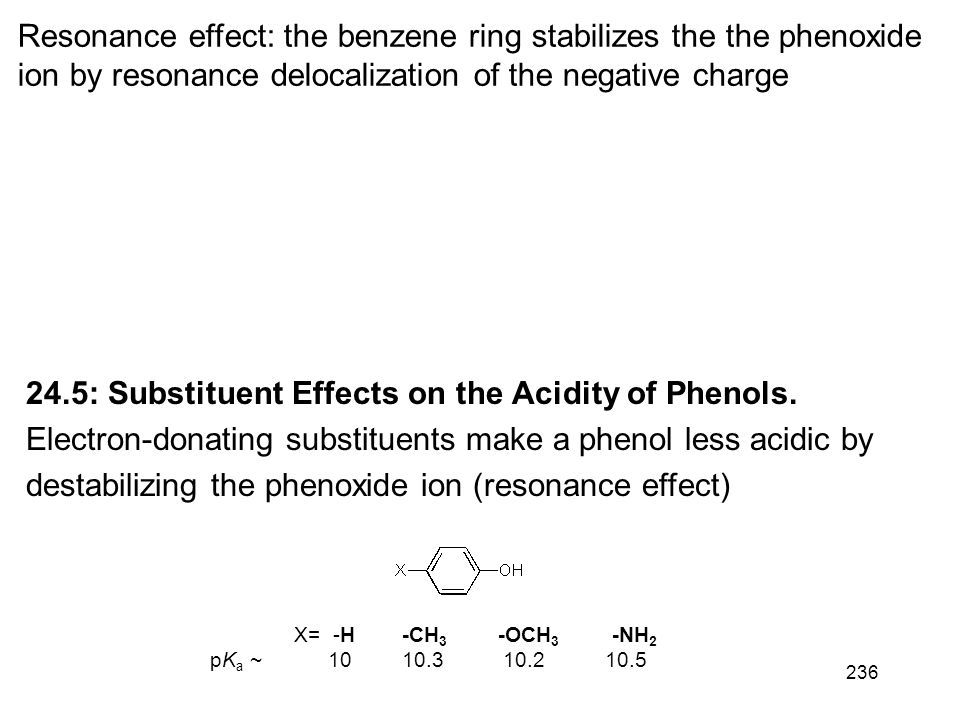
Electron-withdrawing substituents make a phenol more acidic by stabilizing the phenoxide ion through delocalization of the negative charge and through inductive |
What is the strongest phenol acid?
Typical aliphatic (i.e. “alkyl”) alcohols such as ethanol, isopropanol, and t-butanol have a pKa of about 16-18, making them slightly more acidic than water.
Alcohols that are in conjugation with a pi bond or aromatic ring will be more acidic since the conjugate base is resonance-stabilized.What is the pKa of phenol?
Due to the presence of strong electron- withdrawing group - NO2(−I,−R), p-nitro phenol is the strongest acid.
Which is the most acidic phenol?
m-Nitrophenol. 2,4-Dinitrophenol.
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
24.5: Substituent Effects on the Acidity of Phenols. Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance |
|
Acidity of phenols effect of substituents on acidity
https://www.sips.org.in/wp-content/uploads/2021/08/Phenols.pdf |
|
A Theoretical Investigation of Excited-State Acidity of Phenol and
Abstract: We present an ab initio study of the first few singlets of the acid/base couples phenol/phenolate and cyanophenols/cyanophenolates in both gas and |
|
Alcohols Phenols and Ethers
From the above data you will note that phenol is million times more acidic than ethanol. Arrange the following compounds in increasing order of their acid |
|
Alcohols Phenols
https://ncert.nic.in/ncerts/l/lech202.pdf |
|
A Theoretical Investigation of Excited-State Acidity of Phenol and
Abstract: We present an ab initio study of the first few singlets of the acid/base couples phenol/phenolate and cyanophenols/cyanophenolates in both gas and |
|
Safety Data Sheet
19 Mar 2015 108-95-2 Phenol |
|
Determination of the Acidity Constants of some Phenol Radical
Phenoxyl radicals were generated in solutions containing from 0 to 75 % sulphuric acid. The changes observed in the e.s.r. spectra over this range are shown |
|
PREPARATION OF YEAST RNA BY EXTRACTION WITH HOT
Yeast RNA can be isolated efficiently and directly from intact cells by extraction with acidic phenol. (pH 5) and SDS at 65°C. Because this procedure does not |
|
Leep511.pdf
Explain why p-nitrophenol is more acidic than phenol. 54. Explain why alcohols and ethers of comparable molecular mass have different boiling points? 55. The |
|
18.7 ACIDITY OF PHENOLS
Thus phenol is approximately 107 times more acidic than an alcohol of similar size and shape. Recall from Fig. 3.2 |
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
24.5: Substituent Effects on the Acidity of Phenols. Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance |
|
A Theoretical Investigation of Excited-State Acidity of Phenol and
Abstract: We present an ab initio study of the first few singlets of the acid/base couples phenol/phenolate and cyanophenols/cyanophenolates in both gas and |
|
A Theoretical Investigation of Excited-State Acidity of Phenol and
Abstract: We present an ab initio study of the first few singlets of the acid/base couples phenol/phenolate and cyanophenols/cyanophenolates in both gas and |
|
Determination of the Acidity Constants of some Phenol Radical
of phenol radical cations. In favourable cases plots of the e.s.r. parameters against acidity enabled us to determine the pKa of the radical cations |
|
Chapter 17: Alcohols and Phenols
For phenols follow benzene nomenclature and use phenol as the parent name. 17.3: Properties of alcohols and phenols: acidity and basicity:. |
|
Alcohols Phenols and Ethers
(iii) Acidity of phenols: The reactions of phenol with metals (e.g. sodium |
|
Steric Hindrance and Acidity
Apparent molal volumes for some phenols and their sodium salts in methanol have been evaluated from density data. Volumes of ionization for the phenols are |
|
Conjugative Effects of Methylsulfonyl and Methylthio Groupings1
CHSOi group on the acidity of phenol and the anilinium ion -than for its effect on the acidity of benzoic acid is interpreted. |
|
120 Chapter 24: Phenols Alcohols contain an OH group bonded to
24 5: Substituent Effects on the Acidity of Phenols Electron-donating substituents make a phenol less acidic by destabilizing the phenoxide ion (resonance effect) |
|
Chapter 17: Alcohols and Phenols
76 Chapter 17: Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol 17 3: Properties of alcohols and phenols: acidity and basicity: Like water |
|
Phenols - Patna Science College
Phenols are much more acidic than alcohols but less so than carboxylic acids or even carbonic acid Phenols are acidic due to the formation of stable phenoxide |
|
Acidity of phenols, effect of substituents on ac - Yengage
Aromatic compounds containing one or more OH groups directly attached with carbon of benzene ring are called Phenols Simplest phenol is Carbolic Acid |
|
62 PHENOL AND ITS ACIDIC PROPERTIES Phenol is used in
6 2 PHENOL AND ITS ACIDIC PROPERTIES Phenol is used in Chloroseptic for its bacteriocidal properties and for its local anesthetic effect which reduces the |
|
Answers PHENOL: ACIDITY - Chemguide
PHENOL: ACIDITY 1 The position of equilibrium is affected by the stability of the negative ion formed In the phenoxide ion, the charge is delocalised over the |
|
Phenols
Nature of the substituent group: a) Electron withdrawing group such as -NO2, -Cl, -CN, -CHO, and -COOH, increases the acidic character of phenol Reason: |