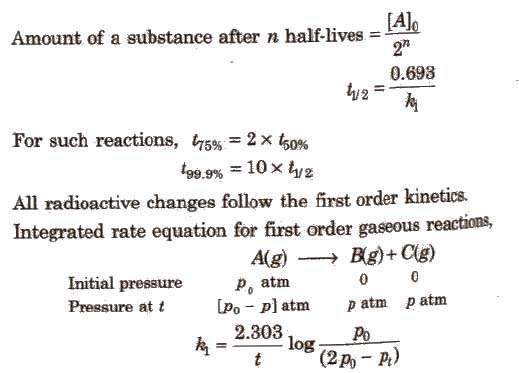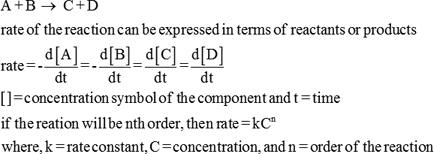chemical kinetics class 12 important formulas
|
Chemical Kinetics Formula Sheet
Chemical Kinetics Formula Sheet Page 2 Zero Order Reaction Reaction rate Page 3 First Order reaction Page 4 Gibbs Free energyhange Second order |
|
Formulae For CHEMICAL KINETICS
CHAPTER 4 - CHEMICAL KINETICS If rate law expression for a reaction is Rate = k [A] [B] Order of reaction = x + y u Important Terms Definitions Formulae |
Chemical kinetics is a difficult topic for many students.
They need a good understanding of the underlying concepts and a firm grasp of mathematics.
What is the main formula for chemical kinetics?
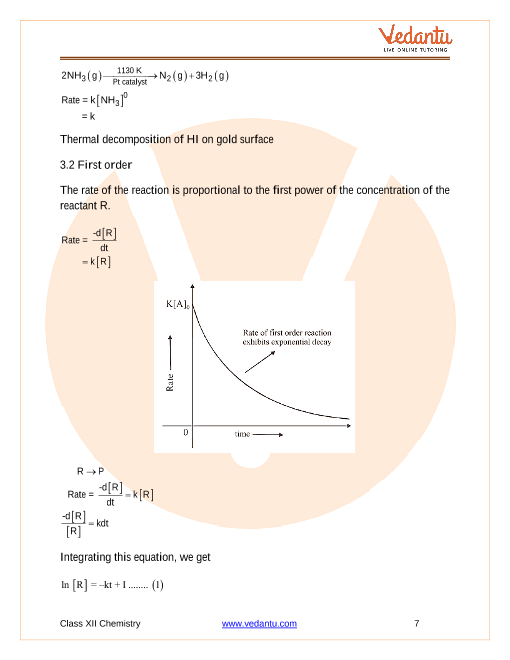
For first-order reactions, the equation ln[A] = -kt + ln[A]0 is similar to that of a straight line (y = mx + c) with slope -k.
This line can be graphically plotted as follows.
Thus, the graph for ln[A] v/s t for a first-order reaction is a straight line with slope -k.
What are the most important formulas in chemistry?
Common Equations Used in Chemistry. Calculation of density or molar mass of gas: d = coefficients in equation] Energy of an electron in the nth state in a hydrogen atom: En = -RH( Dipole moment in terms of charge (Q) and distance of separation (r) between. RT + C. Entropy change of heat flow at constant temperature: ∆S =
Which topics are important in chemical kinetics?
Related topics on Chemical kinetics
Rate of reaction.Rate law and rate constant.Collision theory of chemical reaction.Order and molecularity of reaction.Pseudo First order reaction.Zero order reaction.First order reaction.Second order reaction.- Chemical Kinetics - Formulas.
- All rates written as.
- ∆conc. ∆time or.
- ∆[A] ∆t . Instantaneous rate is the slope of a concentration vs time plot and is. shown by the differential equation: d[A] dt .
- −d[A] dt .
- In general for the overall reation:
- aA + bB → cC + dD.
- overall rxn rate.
|
Formulae For CHEMICAL KINETICS
It can be experimentally determined. XII Chemistry. CHAPTER 4 - CHEMICAL FORMULAS- CHEMICAL KINETICS. Page 2. 5. Integrated rate law for zero order reaction ... |
|
Chemical Kinetics Formula Sheet
Chemical Kinetics. Formula Sheet. Page 2. Zero Order Reaction. Reaction rate. Page Page 12. Page 13. Page 14. Activated Complex theory or Transition state ... |
|
Chemical Kinetics - Formulas
chemical equation. Half-life (t1/2) : The time it takes for the concentration to drop to one half its current value during the course of the reaction. Note ... |
|
Class - XII Multiple Choice Question Bank [MCQ ] Term – I & Term-II
5. 4. CHEMICAL KINETICS. IMPORTANT POINTS : S.No CONCEPT. DETAILS (THEORY & FORMULA). 1. Rate of. Reaction. Change of concentration of reactants & products per |
|
CHEMISTRY (CLASSES XI –XII)
Mole concept and molar mass; percentage composition and empirical and molecular formula; chemical reactions stoichiometry and calculations based on |
|
Lech103.pdf
In this class of reactions the rate of the reaction is proportional to the Chemical kinetics is the study of chemical reactions with respect to reaction. |
|
Physical Science: Tables & Formulas
basic → strong bases. 0. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10 11 12 13 14. Types of Nuclear Radiation. Radiation Type. Symbol. Charge. Nuclear Equation. Alpha ... |
|
Calculations Formulas Definitions
With these principles you should be able to solve many chemical engineering problems. Good Luck! AIChE would like to thank Professors David Murhammer. Chuck |
|
Revised Syllabus to be followed from JEE (Advanced) 2023
Concept of atoms and molecules; Dalton's atomic theory; Mole concept; Chemical formulae;. Balanced chemical equations; Calculations (based on mole concept and |
|
Chemical K Chemical Kinetics
We shall determine these equations only for zero and first order chemical reactions. In this class of reactions the rate of the reaction is proportional to ... |
|
Chemical Kinetics - Formulas
Half-life (t1/2) : The time it takes for the concentration to drop to one half its current value during the course of the reaction. Note that the “current value |
|
Formulae For CHEMICAL KINETICS
XII Chemistry. CHAPTER 4 - CHEMICAL KINETICS. If rate law expression for a reaction is. Rate = k [A] [B]. Order of reaction = x + y u. Important Terms |
|
Bookmark File PDF Bonding Chemical Formulas 19 2 Answers
students and would equally be useful for the aspirants of medical and engineering entrance examinations. Oswaal CBSE Question Bank Class 11 Physics Chemistry |
|
Chemical Kinetics Formula Sheet
Chemical Kinetics. Formula Sheet. Page 2. Zero Order Reaction. Reaction rate. Page 3 Page 10. Reversible Reaction. Page 11. Page 12. Page 13. Page 14 ... |
|
Intext Question Solution Of Alcohol
17 sept. 2022 Important Formulas Terms & Definitions 10. ... Package Chemistry class 12 with 5 Sample Papers is another ... Chemical Kinetics 5. Surface. |
|
Bonding Chemical Formulas 19 2 Answers
10 in One Study Package for CBSE Chemistry Class 12 with 5 Model Papers Disha Solve Reaction Kinetics study guide PDF with answer key worksheet 25 ... |
|
Read Online Ncert Chemistry Class 12 Solution (PDF) - covid19.gov
12 sept. 2022 Oswaal NCERT Exemplar (Problems - solutions) Class 12 Chemistry Book (For ... of the concepts involved along with Important formulas and. |
|
Chemistry Chemical Names Formulas Assessment Answers
ONE CBSE Study Package Chemistry class 12 with Objective Questions & 3 Sample Papers 3rd Edition has 10 key ingredients that will help you achieve success. |
|
Chemistry Chemical Names Formulas Assessment Answers
20 sept. 2022 10 in One Study Package for CBSE Chemistry Class 12 with Objective Questions & 3 Sample ... 9 Important Formulae Terms and Definitions for. |
|
Online Library Intext Question Solution Of Alcohol ? - covid19.gov.gd
20 sept. 2022 Important Formulas Terms & Definitions 10. ... Chemistry Class 12 Dr. S C Rastogi |
|
Chemical Kinetics
Chemical Kinetics - Formulas Chemical Kinetics - Formulas All rates written as ∆conc ∆time or ∆[A] ∆t Instantaneous rate is the slope of a concentration vs time plot and is shown by the differential equation: d[A] dt −d[A] dt In general for the overall reation: aA + bB → cC + dD overall rxn rate |
|
Formulae For CHEMICAL KINETICS - WordPresscom
CHAPTER 4 - CHEMICAL KINETICS If rate law expression for a reaction is Rate = k [A] [B] Order of reaction = x + y u Important Terms, Definitions Formulae |
|
Chemical Kinetics - NCERT
important to know the rate and the factors controlling the rate of Chemical Kinetics helps us to understand how chemical reactions occur 4 Equations ( 4 1) and (4 2) given above represent the average rate of course of the reaction So, in |
|
CHEMISTRY CLASS-XII - edudel
therefore, formula of the compound is AB2 Some Important Formulae 1 Chemical Kinetics : The branch of physical chemistry which deals with the study |
|
Physical Chemistry 3: — Chemical Kinetics — - Uni Kiel
26 jui 2019 · 5 4 1 Fundamental equation for reactive scattering in crossed molecular beams Figure 1: Physical chemistry courses at CAU Kiel area of “femtochemistry” has grown enormously in importance in the last decade, since |
|
Chemical Kinetics
Chemical Kinetics Kinetics – how fast does a reaction proceed? Important factors which affect rates of reactions: – reactant stoichiometric coefficient in a balanced chemical equation 1 broken and formed during the course of a reaction |
|
Unit-4 CHEMICAL KINETICS - PUE
Ans: Sum of the powers of the concentration of the reactants in the rate equation is called order of reaction Q 9) Calculate the overall order of a reaction which has |
|
Basic Principles of Chemical Kinetics - Wiley-VCH
Chapter 1 Basic Principles of Chemical Kinetics 1 1 Symbols, terminology and abbreviations This book enzyme kinetics (see some of the equations in Chapter 8, Chapter 8 points of view are extreme, of course, but at least in enzyme |
|
Chapter 14 Chemical Kinetics
➢ Explain how the activation energy affects a rate and be able to use the Arrhenius Equation ➢ Predict a rate law for a reaction having multistep mechanism |
|
Chemistry Notes for class 12 Chapter 4 Chemical Kinetics - Ncert Help
The branch of chemistry, which deals with the rate of chemical reactions the factors affecting the rate of reactions and the mechanism of the reaction is called chemical kinetics Chemical Important points about Arrhenius equation (i) If ℜ2 |