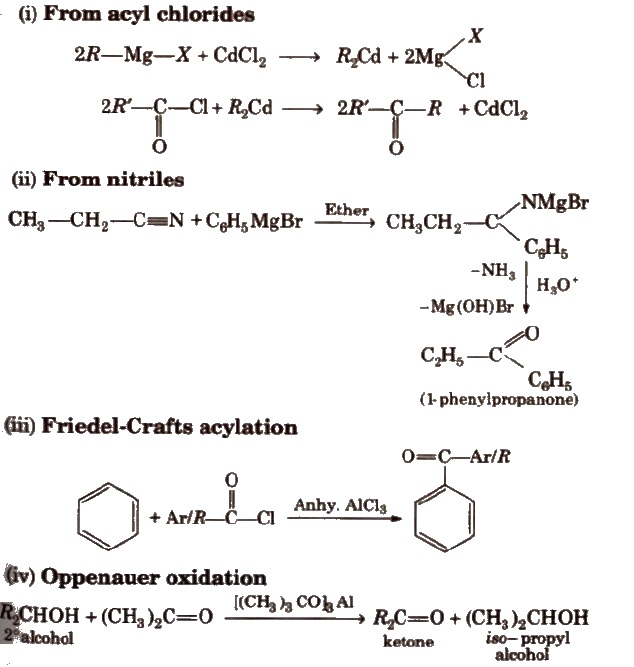
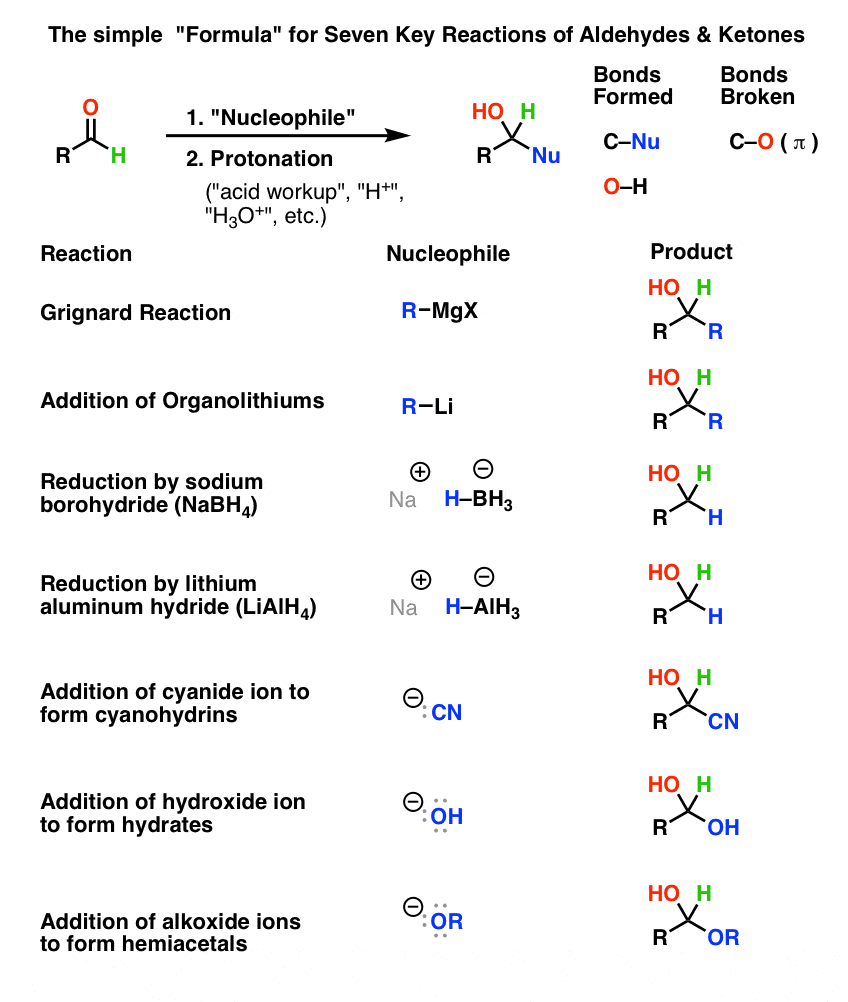
chemical properties between aldehydes and ketones
|
Chapter 5 aldehydes & ketones
The -CH=O group characteristic of aldehydes is often called a formyl group o In ketones the carbonyl carbon atom is connected to two other carbon atoms |
What chemical distinguish between aldehydes and ketones?
The Tollens' test is a reaction that is used to distinguish aldehydes from ketones, as aldehydes are able to be oxidized into a carboxylic acid while ketones cannot.
Tollens' reagent, which is a mixture of silver nitrate and ammonia, oxidizes the aldehyde to a carboxylic acid.How do the chemical structures differ between an aldehyde and a ketone?
An aldehyde is an organic compound in which the carbonyl group is attached to a carbon atom at the end of a carbon chain.
A ketone is an organic compound in which the carbonyl group is attached to a carbon atom within the carbon chain.
Aldehydes and ketones generally have lower boiling points than those of alcohols.What is the chemical properties of aldehyde and ketone?
Aldehydes are readily oxidized to carboxylic acids, whereas ketones resist oxidation.
The aldehydes are, in fact, among the most easily oxidized of organic compounds.
They are oxidized by oxygen (O2) in air to carboxylic acids.
The ease of oxidation helps chemists identify aldehydes.15 sept. 2022
|
Subject 3. ANALYSIS OF CHEMICAL PROPERTIES OF
Know structure of aldehydes and ketones. Examine reactivity of carbonyl compounds: aldehydes and ketones in their chemical reactions. Explain reaction |
|
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
correlate physical properties and chemical reactions of aldehydes ketones and carboxylic acids |
|
Identification of Aldehydes and Ketones
Both aldehydes and ketones contain the carbonyl group. C=O that largely determines the chief chemical and physical properties of aldehydes and ketones. Page 3 |
|
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
correlate physical properties and chemical reactions of aldehydes ketones and carboxylic acids |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones
Structure of Aldehydes and Ketones. • The carbonyl carbon of an aldehyde or ketone is sp2-hybridized. • The bond angle is close to 120° (trigonal planar). |
|
Fundamentals of Organic Chemistry CHAPTER 5. ALDEHYDES
2 mai 2020 Know the physical properties of aldehydes and ketones. ? Know how to synthesize an aldehyde or a ketone. ? Know the different nucleophilic ... |
|
Identification of Aldehydes and Ketones - Agroups R and ? may be
and are often referred to collectively as carbonyl compounds. It is this carbonyl group that largely determines the chief chemical and physical properties of |
|
Sahar Mohammed Shakir Sarah Sattar Jabbar
as carbonyl compounds. It is this carbonyl group that largely determines the chief chemical and physical properties of aldehydes and ketones. |
|
Aldehydes and Ketones
How Are Aldehydes and Ketones Named? 12.3. What Are the Physical Properties of Aldehydes and Ketones? 12.4. What Is the Most Common Reaction Theme of. |
|
PHYSICOCHEMICAL PROPERTIES OF ORGANIC MEDICINAL
Principles of Drug Action 1, Spring 2005, Aldehydes and Ketones 2 II Aldehyde and Ketone Solubility The carbonyl function (C=O) of these compounds is a |
|
Carbonyl Chemistry (12 Lectures) Aldehydes and Ketones - chicac
Aldehydes and ketones are hydrogen bond acceptors; this makes them have considerable solubilities in water Ketones such as acetone are good solvents because they dissolve both aqueous and organic compounds Recall that acetone is a polar, aprotic solvent Reactions with acids: – The carbonyl oxygen is weakly basic |
|
ALDEHYDES AND KETONES
CHEMICAL PROPERTIES OF ALDEHYDES AND KETONES Nucleophilic addition reaction In this reaction carbon atom of carbonyl group changes from sp2 to |
|
Aldehydes, Ketones and Carboxylic Acids - NCERT
correlate physical properties and chemical reactions of aldehydes, ketones and carboxylic acids, with their structures; • explain the mechanism of a few selected |
|
Alcohols, Ethers, Aldehydes, and Ketones
Aldehydes and ketones don't form hydrogen bonds to each other, however, they form hydrogen bond with water using the electron lone pairs on oxygen Aldehydes and ketones have lower boiling points compared to alcohols of similar size Aldehydes and ketones with fewer than five carbons are soluble in water |
|
Fundamentals of Organic Chemistry CHAPTER 5 ALDEHYDES
27 jan 2019 · o The polarity of the carbonyl group also affects the solubility properties of aldehydes and ketones o Carbonyl compounds with low molecular |
|
Carbonyl Compounds: Ketones, Aldehydes - UCR Chemistry
13 6 Spectrometric Properties of Carbonyl Compounds However, since most other chemical reactions of aldehydes and ketones are similar, it is likely that |
|
27 ALDEHYDES, KETONES AND CARBOXYLIC ACIDS - NIOS
The physical properties and chemical reactions of aldehydes and ketones are a direct consequence of this polarisation The dipole-dipole attraction between the |
|
Chemistry of Carbonyl Compounds - STET
1 1 Nomenclature - structure of carbonyl compounds - chemical properties - The boiling point of aldehydes and ketones are higher than hydrocarbons and |
|
Properties of Aldehydes and Ketones Introduction Discussion
Aldehydes and Ketones represent an important class of organic molecules containing a carbonyl carbon In this experiment you will study the chemical properties |