chemistry form ws10.1.2a redox and electrochemistry answer key
|
Unit 2 Redox and Electrochemistry Notes
Electrochemistry is the interchange of chemical and electrical energy The key to electrochemistry is a redox reaction This unique type of reaction |
|
Unit 6: Redox Reactions and Electrochemistry
Spontaneous Chemical Reaction? yes no Value of E° (positive or negative) positive negative Page 2 Name: ANSWER KEY Thus on the iron bar we want to form |
How do you answer redox reactions?
Some points to remember when balancing redox reactions:
The equation is separated into two half-equations, one for oxidation, and one for reduction.
The equation is balanced by adjusting coefficients and adding H2O, H+, and e- in this order: Balance the atoms in the equation, apart from O and H.How do you solve redox equations in chemistry?
A redox equation can be balanced using the following stepwise procedure: (.
1) Divide the equation into two half-reactions. (.
2) Balance each half-reaction for mass and charge. (.
3) Equalize the number of electrons transferred in each half-reaction. (.
4) Add the half-reactions together.What is a redox reaction in electrochemistry?
A redox reaction can be defined as a chemical reaction in which electrons are transferred between two reactants participating in it.
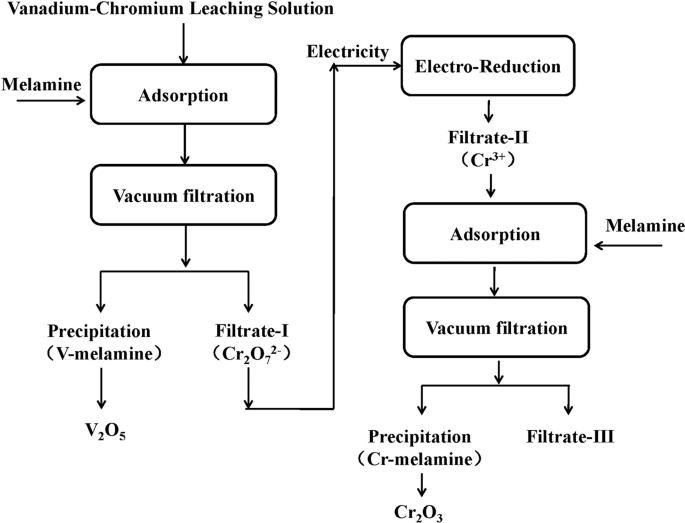
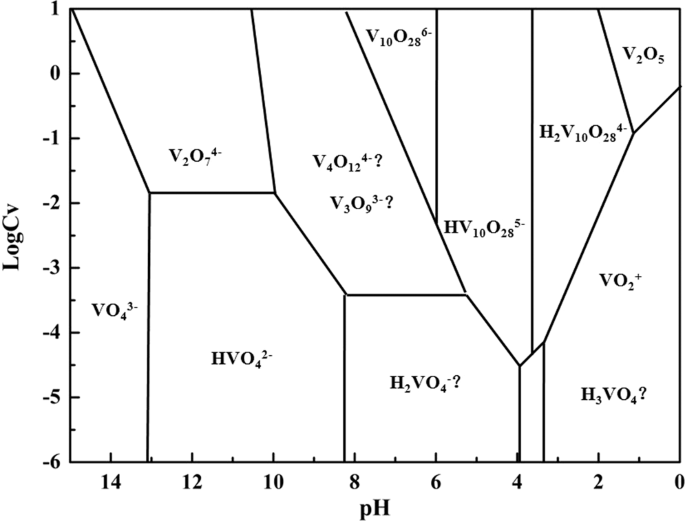
This transfer of electrons can be identified by observing the changes in the oxidation states of the reacting species.The coefficient for H2O(l) when H2O2(aq) Cr2O72−(aq) → O2(g) Cr3(aq) is balanced in acidic aqueous solution is 7.
To balance the given chemical reaction, follow the given steps:First, you need to divide the reaction into half-reactions as shown below.
|
Chemistry form ws10.1.2a redox and electrochemistry worksheet
Chemistry form ws10.1.2a redox and electrochemistry worksheet answers REDOX anti-oxidation reduce decirculation agent Galvanic (Voltaic) Anode Cathode ... |
|
C:Documents and SettingsEvanDesktopChemistryChemistry
Chemistry: Form WS10.1.2A. Name. REDOX AND ELECTROCHEMISTRY When chemical bonds form electrons are either lost |
|
F: Form WS1014A ELECTROCHEMISTRY Name Date Period
ELECTROCHEMISTRY Name Date method In redox reactions, the number of electrons lost is always equal Step 7: Combine ions to form compounds in original equation Chemical reactions often involve the movement of electrons |
|
Analyzing Oxidation-Reduction Reactions - Evans Chemistry Corner
( L Go on to the next page ) Chemistry: Form WS10 1 2A Name REDOX AND ELECTROCHEMISTRY Date When chemical bonds form, electrons are either lost, gained or shared Metals Write your answer in the space provided Reaction |
|
Determining the Voltage of an Electrochemical Cell - Evans
Cu0 E0 = 1 10v Chemistry: Form WS10 3 2A Determining the Voltage of Electrochemical Cells Chemical reactions often involve the movement of electrons |
|
Evans Chemistry Redox And Electrochemistry Answers
Chemistry: Form WS10 2 1A Name _____ REDOX AND ELECTROCHEMISTRY Date _____ Period _____ Applying the Activity Series During a single |
|
Echem Ochem HW
4 jui 2011 · Chemistry: Form WS10 3 1A REDOX AND ELECTROCHEMISTRY Answer questions 5-16 by referring to Table J For each of the electrode |
|
Chemistry Form Ls10 1a - Ruforum
27 nov 2015 · can read or download chemistry form ws 1 4 1a answers in pdf format if or shared, chemistry form ws10 1 1a name redox and electrochemistry date chemistry form ls10 1a redox and electrochemistry page 2 answer the |
|
Chemistry Form Ls10 1a
Maliaeria Cf Chemistry Form Ws10 1 2a Answers No IP FORM LS10 1A REDOX AND ELECTROCHEMISTRY PAGE 2 ANSWER THE QUESTIONS BELOW |
|
Redox Reactions and Electrochemistry
5 Balancing Redox Equations 1 Write the unbalanced equation for the reaction ion ionic form The oxidation of Fe2+ to Fe3+ by Cr 2 O 7 2- in acid solution? |
|
Unit 12: Electrochemistry
source metal into a solution using an external power source and then reducing Regents Chemistry '14-'15 Mr Murdoch Page 6 of 61 Key Notes page: to form Cl2 0 Redox reactions used for electrochemistry are driven by a change in |