chemistry form ws10.1.2a redox and electrochemistry answers
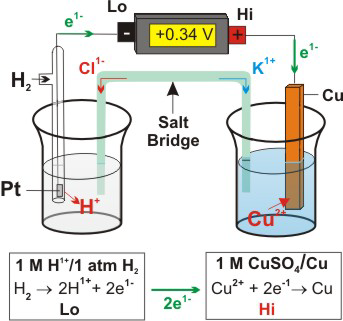
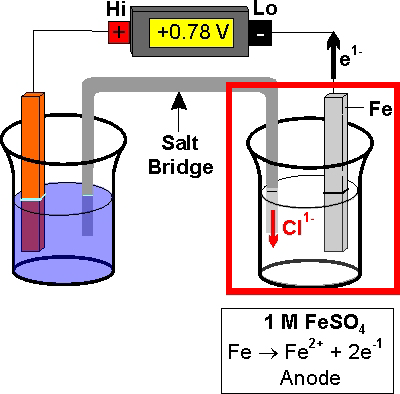
The galvanic cell, or called voltaic cell, is an electrochemical cell that converts the chemical energy to electrical energy from the spontaneous redox reactions taking place in the cell.
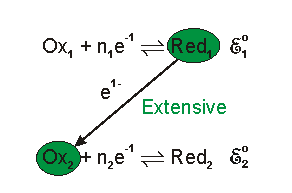
The redox reaction is Faradic reaction, which is defined as reaction involved with electron transfer from/to electrode to/from ions.
How do you answer redox reaction questions?
Example 2: Balancing redox equations in basic solution
1Step 1: Divide the equation into half-reactions.2) Step 2: Balance each half-reaction for mass and charge.
3) Step 3: Equalize the number of electrons transferred.
4) Step 4: Add the half-reactions together.
What is the coefficient for H2O when H2O2 Cr2O72 − → O2 Cr3+ is balanced in acid?
The coefficient for H2O(l) when H2O2(aq) Cr2O72−(aq) → O2(g) Cr3(aq) is balanced in acidic aqueous solution is 7.
To balance the given chemical reaction, follow the given steps:First, you need to divide the reaction into half-reactions as shown below.
|
C:Documents and SettingsEvanDesktopChemistryChemistry
Chemistry: Form WS10.1.2A. Name. REDOX AND ELECTROCHEMISTRY When chemical bonds form electrons are either lost |
|
Chemistry form ws10.1.2a redox and electrochemistry worksheet
Chemistry form ws10.1.2a redox and electrochemistry worksheet answers REDOX anti-oxidation reduce decirculation agent Galvanic (Voltaic) Anode Cathode ... |
|
F: Form WS1014A ELECTROCHEMISTRY Name Date Period
ELECTROCHEMISTRY Name Date method In redox reactions, the number of electrons lost is always equal Step 7: Combine ions to form compounds in original equation Chemical reactions often involve the movement of electrons |
|
Evans Chemistry Redox And Electrochemistry Answers
Chemistry: Form WS10 2 1A Name _____ REDOX AND ELECTROCHEMISTRY Date _____ Period _____ Applying the Activity Series During a single |
|
Evans Chemistry Redox And Electrochemistry Answers - dTV
13 nov 2020 · Chemistry: Form WS10 2 1A Name _____ REDOX AND ELECTROCHEMISTRY Date _____ Period _____ Applying the Activity Series During a |
|
Analyzing Oxidation-Reduction Reactions - Evans Chemistry Corner
( L Go on to the next page ) Chemistry: Form WS10 1 2A Name REDOX AND ELECTROCHEMISTRY Date When chemical bonds form, electrons are either lost, gained or shared Metals Write your answer in the space provided Reaction |
|
A Salt and Battery - Evans Chemistry Corner
Chemistry: Form WS10 3 1A Name REDOX AND ELECTROCHEMISTRY The electricity generated by a battery comes from a chemical reaction known as Answer the questions below based on your reading above and on your knowledge |
|
Echem Ochem HW
4 jui 2011 · Chemistry: Form WS10 3 1A REDOX AND ELECTROCHEMISTRY Answer questions 5-16 by referring to Table J For each of the electrode |
|
Redox Reactions and Electrochemistry
For reactions in basic solutions, add OH- to instead of H+ to balance electronic charges 10 Balance the reaction in the molecular form Page 8 8 |