23andme warning
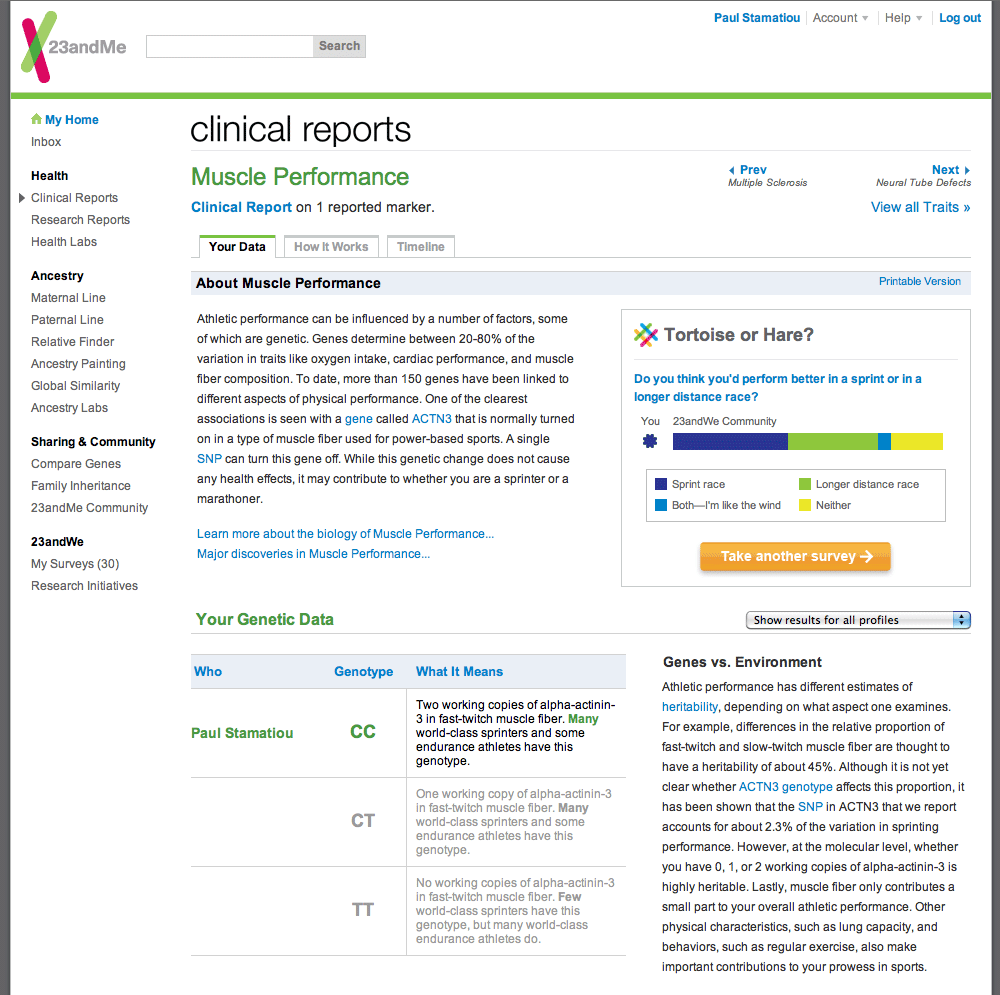
Even though our prediction about your trait may not always match your actual trait, the genetic result underlying that prediction is highly accurate.
Our genotyping platform is a well-established and reliable technology for analyzing DNA, and all samples are processed in CLIA-certified labs.
What are the risks of 23andMe?
What about the risk that DNA testing companies are hacked? Some 6.9 million 23andMe customers had their data compromised after an anonymous hacker accessed user profiles and posted them for sale on the internet earlier this year, the company said Dec.
|
Sans titre
10 jui. 2010 RE: 23andMe Personal Genome Service™. Dear Ms. Wojcicki: The United States Food and Drug Administration (FDA) has determined that your firm. |
|
Disclosures
*www.genomeweb.com/molecular-diagnostics/oncology-community-sees-potential-patient- harm-fda-ok-23andme-brca-test#.Wuz6ZNPwbVo. FDA. – 2013 FDA warning |
|
Bitter Taste
21 oct. 2015 Of 23andMe research participants with genetics like yours: Bitter Taste. A bitter warning. Scientists believe the bitter taste sense ... |
|
23andMe and the FDA
12 fév. 2014 23andMe a warning letter order- ing it to “immediately discontin- ue marketing the PGS [Saliva. Collection Kit and Personal Ge-. |
|
Overview of 23andMe Phenotype Data Collection
Preceding warning signs. ? Visual changes. ? Family history. ? Medications used (NSAIDS dihydroergotamine |
|
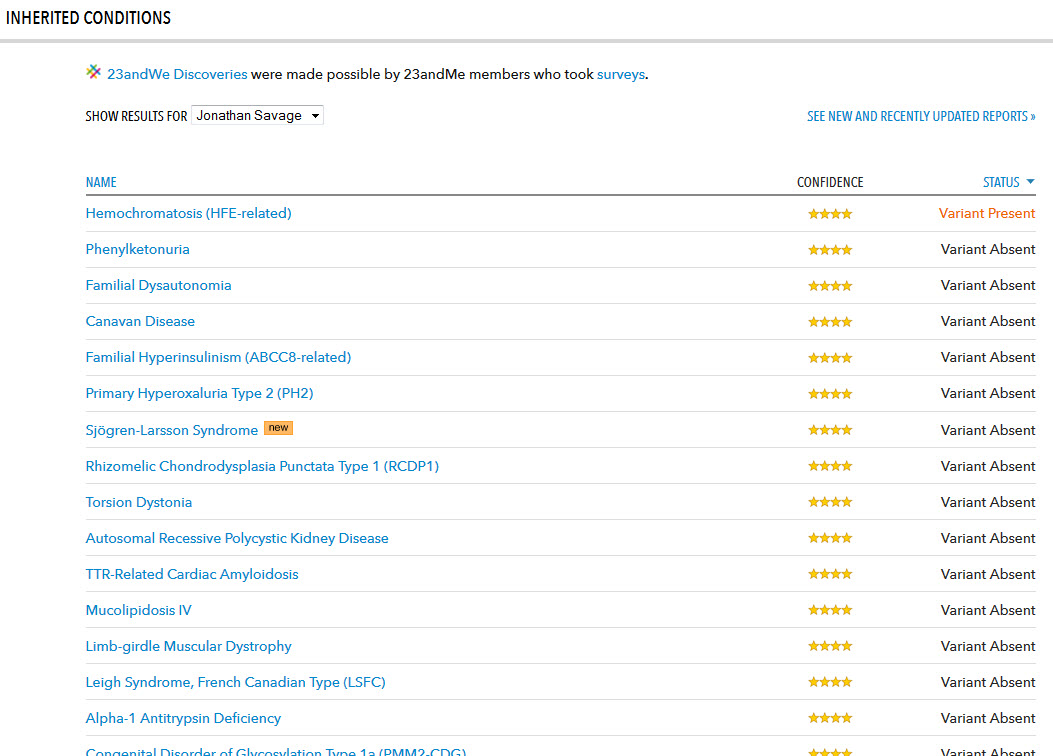
Primary Hyperoxaluria Type 2
Indications for Use. Warnings and Limitations. The 23andMe PGS Carrier Status Test for Primary Hyperoxaluria Type 2 is indicated for the detection of. |
|
23andMe® Personal Genome Service® (PGS) Genetic Health Risk
23andMe® Personal Genome Service® (PGS). Genetic Health Risk Reports Package Insert. Table of contents. • Intended Use. • Important warnings and limitations. |
|
Untitled
17 avr. 2017 Warnings and Limitations. The 23andMe PGS Genetic Health Risk Report for Parkinson's Disease is indicated for reporting of the. |
|
23andMe, the Food and Drug Administration, and the Future - CORE
sending a warning letter to 23andMe, the leading com- pany in the field, directing it to stop providing such testing 1 The FDA acted as the era of widespread, clini |
|
23andMe - CORE
(FDA) halted the sale of genetic health testing, on the grounds that 23andMe was not Warning In: Forbes Pharma Healthcare; 2013 http://www forbes com/ |
|
Case 14: 23andMe (and the FDA) International scientists working on
He also encouraged others to take the test, with the warning that, "the shorter the genetic straw you draw, the more important it is for you to be aware of it "73 He |
|
23andMe and the FDA
12 fév 2014 · 23andMe a warning letter order- ing it to “immediately discontin- ue marketing the PGS [Saliva Collection Kit and Personal Ge- nome Service] |
|
SAFETY DATA SHEET - 23andMe
15 mar 2016 · Warning Hazard statements : H226 - Flammable liquid and vapor H319 - Causes serious eye irritation H336 - May cause drowsiness or |
|
Disclosures - 23andMe for Medical Professionals
2013 FDA warning letter – 2018 Authorized Interim: 3000 to 4000 BRCA+ Experts urge caution: They say: Only tests for 3 of >1000 pathogenic variants |
|
Warning Letters > 23andMe, Inc - Close Out Letter 3/25/14 - FDAnews
25 mar 2014 · Warning Letters > 23andMe, Inc - Close Out Letter 3/25/14 our Warning Letter CMS 415534 dated November 22, 2013 Based on our |
|
Submissions received and TGA response: Review of the regulation
21 nov 2019 · the risks and benefits of 23andMe's DTC genetic health testing product The opt -in page provides specific warnings relevant to each test result |